Ireland 2019 Lecture 17 and Discussion 11
\newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} }
\newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}}
\newcommand{\id}{\mathrm{id}} \newcommand{\Span}{\mathrm{span}}
( \newcommand{\kernel}{\mathrm{null}\,}\) \newcommand{\range}{\mathrm{range}\,}
\newcommand{\RealPart}{\mathrm{Re}} \newcommand{\ImaginaryPart}{\mathrm{Im}}
\newcommand{\Argument}{\mathrm{Arg}} \newcommand{\norm}[1]{\| #1 \|}
\newcommand{\inner}[2]{\langle #1, #2 \rangle}
\newcommand{\Span}{\mathrm{span}}
\newcommand{\id}{\mathrm{id}}
\newcommand{\Span}{\mathrm{span}}
\newcommand{\kernel}{\mathrm{null}\,}
\newcommand{\range}{\mathrm{range}\,}
\newcommand{\RealPart}{\mathrm{Re}}
\newcommand{\ImaginaryPart}{\mathrm{Im}}
\newcommand{\Argument}{\mathrm{Arg}}
\newcommand{\norm}[1]{\| #1 \|}
\newcommand{\inner}[2]{\langle #1, #2 \rangle}
\newcommand{\Span}{\mathrm{span}} \newcommand{\AA}{\unicode[.8,0]{x212B}}
\newcommand{\vectorA}[1]{\vec{#1}} % arrow
\newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow
\newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} }
\newcommand{\vectorC}[1]{\textbf{#1}}
\newcommand{\vectorD}[1]{\overrightarrow{#1}}
\newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}}
\newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}}
\newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} }
\newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}}
\newcommand{\avec}{\mathbf a} \newcommand{\bvec}{\mathbf b} \newcommand{\cvec}{\mathbf c} \newcommand{\dvec}{\mathbf d} \newcommand{\dtil}{\widetilde{\mathbf d}} \newcommand{\evec}{\mathbf e} \newcommand{\fvec}{\mathbf f} \newcommand{\nvec}{\mathbf n} \newcommand{\pvec}{\mathbf p} \newcommand{\qvec}{\mathbf q} \newcommand{\svec}{\mathbf s} \newcommand{\tvec}{\mathbf t} \newcommand{\uvec}{\mathbf u} \newcommand{\vvec}{\mathbf v} \newcommand{\wvec}{\mathbf w} \newcommand{\xvec}{\mathbf x} \newcommand{\yvec}{\mathbf y} \newcommand{\zvec}{\mathbf z} \newcommand{\rvec}{\mathbf r} \newcommand{\mvec}{\mathbf m} \newcommand{\zerovec}{\mathbf 0} \newcommand{\onevec}{\mathbf 1} \newcommand{\real}{\mathbb R} \newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]} \newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]} \newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]} \newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]} \newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]} \newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]} \newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]} \newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]} \newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]} \newcommand{\laspan}[1]{\text{Span}\{#1\}} \newcommand{\bcal}{\cal B} \newcommand{\ccal}{\cal C} \newcommand{\scal}{\cal S} \newcommand{\wcal}{\cal W} \newcommand{\ecal}{\cal E} \newcommand{\coords}[2]{\left\{#1\right\}_{#2}} \newcommand{\gray}[1]{\color{gray}{#1}} \newcommand{\lgray}[1]{\color{lightgray}{#1}} \newcommand{\rank}{\operatorname{rank}} \newcommand{\row}{\text{Row}} \newcommand{\col}{\text{Col}} \renewcommand{\row}{\text{Row}} \newcommand{\nul}{\text{Nul}} \newcommand{\var}{\text{Var}} \newcommand{\corr}{\text{corr}} \newcommand{\len}[1]{\left|#1\right|} \newcommand{\bbar}{\overline{\bvec}} \newcommand{\bhat}{\widehat{\bvec}} \newcommand{\bperp}{\bvec^\perp} \newcommand{\xhat}{\widehat{\xvec}} \newcommand{\vhat}{\widehat{\vvec}} \newcommand{\uhat}{\widehat{\uvec}} \newcommand{\what}{\widehat{\wvec}} \newcommand{\Sighat}{\widehat{\Sigma}} \newcommand{\lt}{<} \newcommand{\gt}{>} \newcommand{\amp}{&} \definecolor{fillinmathshade}{gray}{0.9}Introduction to cell division
An evolutionary goal of all living systems is to reproduce. Since the basic unit of life is a cell, and we know - thanks at least in part to Francesco Reid - that life begets new life - this means that there must be a process by which to create new cells from parental cells. We also know intuitively that multicellular organisms must somehow increase their number of cells during their growth by creating copies of existing cells. The process by which one cell creates one or more new cells, for both single and multi-celled organisms, requires a parental cell to divide and is called cell division.
From the standpoint of the Design Challenge framework we can stipulate that the big problem of cell division is to make a copy of a cell. If a condition for success requires that the daughter cells be viable then a number of subproblems can be defined:
- The cell must replicate its DNA so that at least two cells have a functional copy after cell division is complete - we have discussed this process already.
- The cell must make sufficient copies of the rest of the cellular content so that daughter cells are viable or it must find a way to ensure that the copied DNA (even without a full replica of cellular content) is viable.
- The cell must divide the replicated cell content and DNA between at least two independently bounded compartments.
- To ensure success, the process must be happen in an evolutionarily competitive time and be accomplished with an evolutionary selection-friendly amount of biochemical resources.
While it is not a strict requirement that this process happen in a coordinated manner, Nature has selected for systems in which all of the steps in the process happen in a highly coordinated way. This helps cells meet requirement number 4 in the list above. The coordinated process and the mechanisms of control are generally referred to as the cell cycle. This term can be used to describe the coordinate process used by any cell that is undergoing cell division. When we observe Nature we find that it has evolved two major modes of reproduction: sexual and asexual. Within each of these modes of reproduction we find several major modes of cell division that occur frequently across all domains of life. We consider three of these modes: binary fission (used primarily by single celled bacteria and archaea), mitosis (used often by eukaryotes in processes of cell division NOT associated with sexual reproduction) and meiosis (a process of cell division tightly linked to sexual reproduction). We discuss these processes in the sections that follow.

Figure. First-year UC Davis students enrolled in a hands-on first-year seminar course examine an agar plate of on which they have "painted" designs with an engineered strain of Escherschia coli. The designs only become apparent after the bacteria multiply through the process of binary fission. Opportunities for getting involved in hands-on research abound on the UC Davis campus - make sure to set time aside to get involved before you graduate. (Photo: by undergraduate student Daniel Oberbauer - 2017)
Cell division in the bacteria and archaea
Bacteria and Archaea
Like all other life forms, bacteria and archaea have one key evolutionary driver: to make more of themselves. Typically, bacterial and archaeal cells grow, duplicate all major cellular constituents, like DNA, ribosomes, etc., distribute this content and then divide into two nearly identical daughter cells. This process is called binary fission and is shown mid-process in the figure below. While some bacterial species are known to use several alternative reproductive strategies including making multiple offspring or budding - and all alternative mechanisms still meet the requirements for cell division stipulated above - binary fission is the most commonly laboratory-observed mechanisms for cell division the bacteria and archaea so we limit our discussion to this mechanisms alone.
(Aside: Those who want to read more about alternatives to binary fission in bacteria should check this link out.)
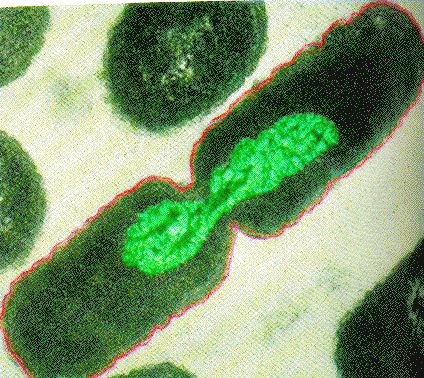
Binary Fission in bacteria starts with DNA replication at the replication origin attached to the cell wall, near the midpoint of the cell. New replication forks can form before the first cell division ends; this phenomenon allows an extremely rapid rate of reproduction. Source: http://biology.kenyon.edu/courses/bi...01/week01.html
Binary Fission
The process of binary fission is the most commonly observed mechanism for cell division in bacteria and archaea (at least the culturable ones studied in the laboratory). The following is a description of a process that happens in some rod-shaped bacteria:
Since we must consider the replication of DNA one structural feature of relevance to DNA replication in the bacteria and archaea is that their genetic material is not enclosed in a nucleus, but instead occupies a specific location, the nucleoid, within the cell. Moreover, the DNA of the nucleoid is associated with numerous proteins that aid in compacting the DNA into a smaller, organized structure. Another organizational feature to note is that the bacterial chromosome is typically attached to the plasma membrane at about the midpoint of the cell. The starting point of replication, the origin, is close to this attachment site. Recall also that replication of the DNA is bidirectional, with replication forks moving away from the origin on both strands of the loop simultaneously. Due to the structural arrangement of the DNA at the midpoint this means that as the new double strands are formed, each origin point moves away from the cell wall attachment toward the opposite ends of the cell.
This process of DNA replication is typically occurring at the same time as a growth in the physical dimensions of the cell. Therefore, as the cell elongates, the growing membrane aids in the transport of the chromosomes towards the two opposite poles of the cells. After the chromosomes have cleared the midpoint of the elongated cell, cytoplasmic separation begins.
The formation of a ring composed of repeating units of a protein called FtsZ (a cytoskeletal protein) directs the formation of a partition between the two new nucleoids. Formation of the FtsZ ring triggers the accumulation of other proteins that work together to recruit new membrane and cell wall materials to the site. Gradually, a septum is formed between the nucleoids, extending from the periphery toward the center of the cell. When the new cell walls are in place, the daughter cells separate.
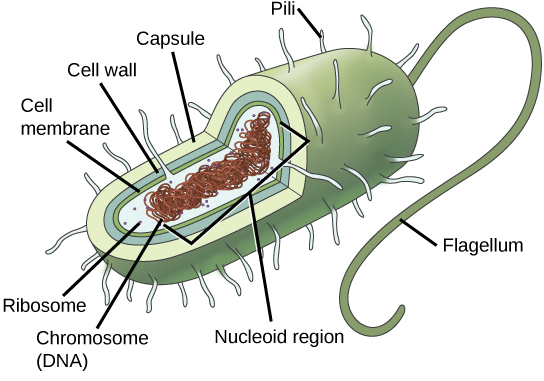
Prokaryotes, including bacteria and archaea, have a single, circular chromosome located in a central region called the nucleoid.
Possible discussion
How does attaching the replicating chromosome to the cell membrane aid in dividing the two chromosomes after replication is complete?
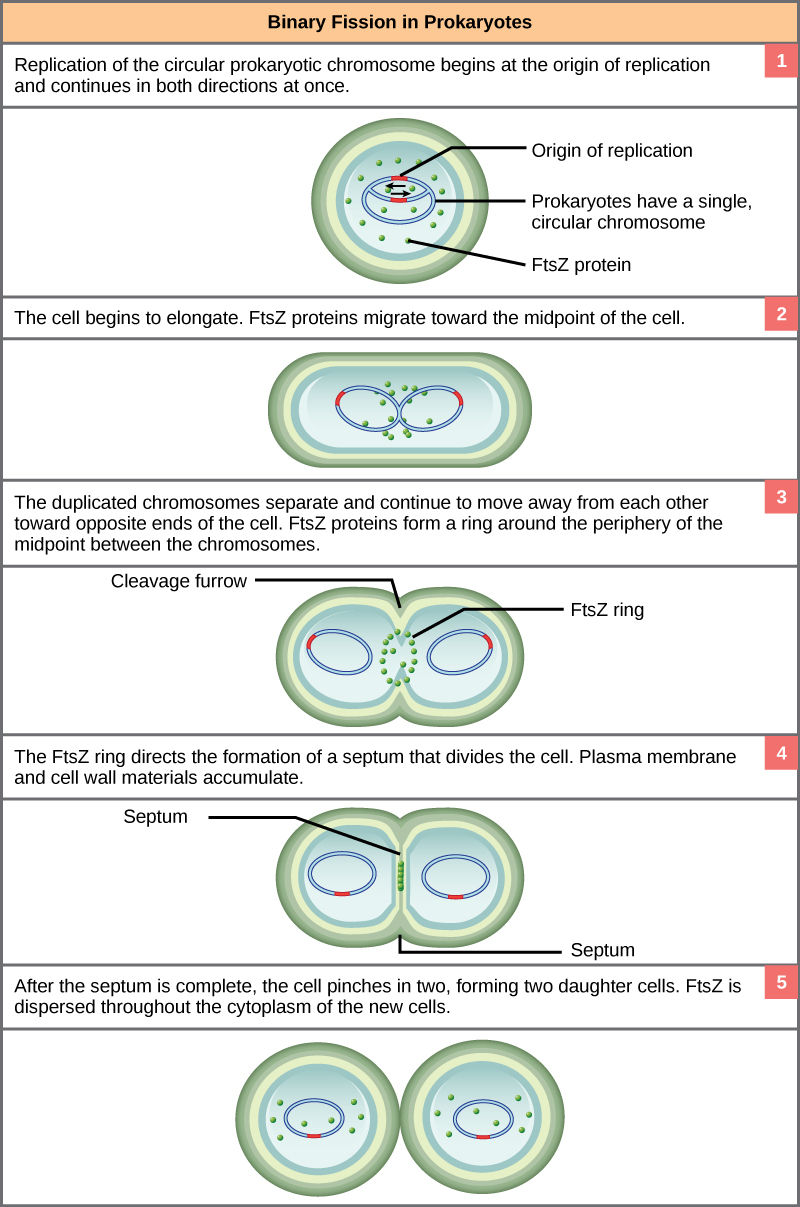
These images show the steps of binary fission in prokaryotes. (credit: modification of work by “Mcstrother”/Wikimedia Commons)
Control of these processes
Not surprisingly, the process of binary fission is strictly controlled in most bacteria and archaea. Somewhat surprisingly, however, while some key molecular players are know, much remains to be discovered and understood about how decisions are made to coordinate the activities.
Eukaryotic Cell Cycle and Mitosis
The cell cycle is an orderly sequence of events used by biological systems to coordinate cell division. In eukaryotes, asexual cell division proceeds via a cell cycle that includes multiple spatially and temporally coordinated events. These include a long preparatory period, called interphase and a mitotic phase called M phase. Interphase is often further divided into distinguishable subphases called G1, S, and G2 phases. Mitosis is the stage in which replicated DNA is distributed to daughter cells and is itself often subdivided into five distinguishable stages: prophase, prometaphase, metaphase, anaphase, and telophase. Mitosis is often accompanied by a process called cytokinesis, during which the cytoplasmic components of the daughter cells are separated either by an actin ring (animal cells) or by cell plate formation (plant cells). The passage through these phases are controlled by checkpoints. There are three major checkpoints in the cell cycle: one near the end of G1, a second at the G2–M transition, and the third during metaphase. These regulatory checks serve to ensure that the processes required to successfully move on to the next phase of the cell cycle have been fully completed and that sufficient resources exist to move on to the next phase of cell division.
Cell Cycle
In asexually reproducing eukaryotic cells, one “turn” of the cell cycle consists of two general phases: interphase, followed by mitosis and cytokinesis. Interphase is the period of the cell cycle during which the cell may either be living and not dividing or in which it is preparing itself to divide. Most of the cells in a fully-developed multicellular organisms are typically found in interphase. Mitosis is the the point in the cell cycle associated with division or distribution of replicated genetic material to two daughter cells. During mitosis the cell nucleus breaks down and two new, fully functional, nuclei are formed. Cytokinesis is the process that divides the cytoplasm into two distinctive cells.
Interphase
G1 Phase
The first stage of interphase is called the G1 phase, or first gap, because little change is visible. However, during the G1 stage, the cell is quite active at the biochemical level. The cell is accumulating the building blocks of chromosomal DNA and the associated proteins, as well as accumulating enough energy reserves to complete the task of replicating each chromosome in the nucleus.
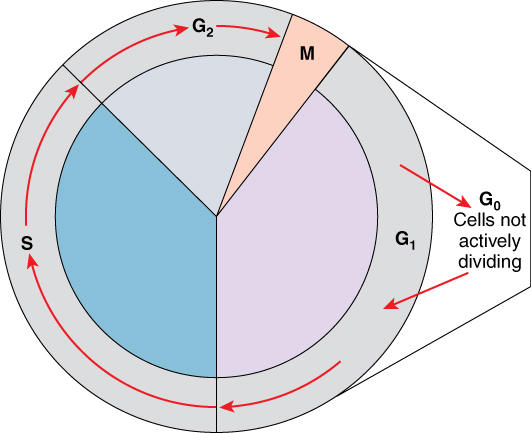
A cell moves through a series of phases in an orderly manner. During interphase, G1 involves cell growth and protein synthesis, the S phase involves DNA replication and the replication of the centrosome, and G2 involves further growth and protein synthesis. The mitotic phase follows interphase. Mitosis is nuclear division during which duplicated chromosomes are segregated and distributed into daughter nuclei. Usually the cell will divide after mitosis in a process called cytokinesis in which the cytoplasm is divided and two daughter cells are formed.
S Phase
Throughout interphase, nuclear DNA remains in a semi-condensed chromatin configuration. In S phase (synthesis phase), DNA replication results in the formation of two identical copies of each chromosome—sister chromatids—that are firmly attached at the centromere region. At the end of this stage, each chromosome has been replicated.
In cells using the organelles called centrosomes, these structures are often duplicated during S phase. Centrosomes consists of a pair of rod-like centrioles composed of tubulin and other proteins that sit at right angles to one another other. The two resulting centrosomes will give rise to the mitotic spindle, the apparatus that orchestrates the movement of chromosomes later during mitosis.
G2 Phase
During the G2 phase, or second gap, the cell replenishes its energy stores and synthesizes the proteins necessary for chromosome manipulation. Some cell organelles are duplicated, and the cytoskeleton is dismantled to provide resources for the mitotic spindle. There may be additional cell growth during G2. The final preparations for the mitotic phase must be completed before the cell is able to enter the first stage of mitosis.
G0 Phase
Not all cells adhere to the classic cell-cycle pattern in which a newly formed daughter cell immediately enters interphase, closely followed by the mitotic phase. Cells in the G0 phase are not actively preparing to divide. The cell is in a quiescent (inactive) stage, having exited the cell cycle. Some cells enter G0 temporarily until an external signal triggers the onset of G1. Other cells that never or rarely divide, such as mature cardiac muscle and nerve cells, remain in G0 permanently
A Quick Aside: Structure of Chromosomes During the Cell Cycle
If the DNA from all 46 chromosomes in a human cell nucleus was laid out end to end, it would measure approximately two meters; however, its diameter would be only 2 nm. Considering that the size of a typical human cell is about 10 µm (100,000 cells lined up to equal one meter), DNA must be tightly packaged to fit in the cell’s nucleus. At the same time, it must also be readily accessible for the genes to be expressed. During some stages of the cell cycle, the long strands of DNA are condensed into compact chromosomes. There are a number of ways that chromosomes are compacted.
Suggested discussion
When should we expect to see highly condensed DNA in the cell (which phases of the cell cycle)? When would the DNA remain un-compacted (during which phases of the cell cycle)?
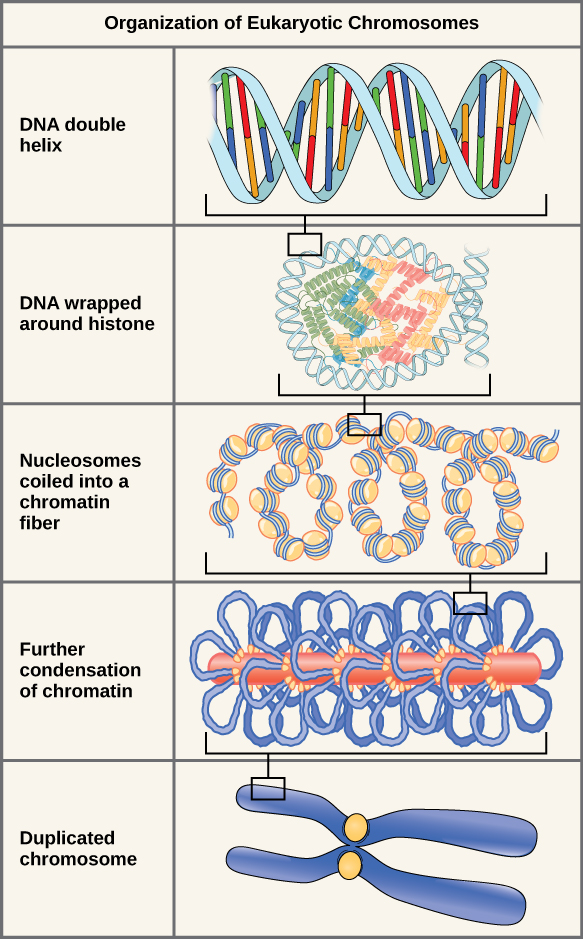
Double-stranded DNA wraps around histone proteins to form nucleosomes that have the appearance of “beads on a string.” The nucleosomes are coiled into a 30-nm chromatin fiber. When a cell undergoes mitosis, the chromosomes condense even further.
Mitosis and Cytokinesis
During the mitotic phase, a cell undergoes two major processes. First, it completes mitosis, during which the contents of the nucleus are equitably pulled apart and distributed between its two halves. Cytokinesis then occurs, dividing the cytoplasm and cell body into two new cells.
Note
The major phases of Mitosis are visually distinct from one another and were originally characterized by what could be seen by viewing dividing cells under a microscope. Some instructors may ask you be able to distinguish each phase be looking at images of cells or more commonly by inspection of cartoon depiction of mitosis. If your instructor is not explicit about this point remember to ask whether this will be expected of you.
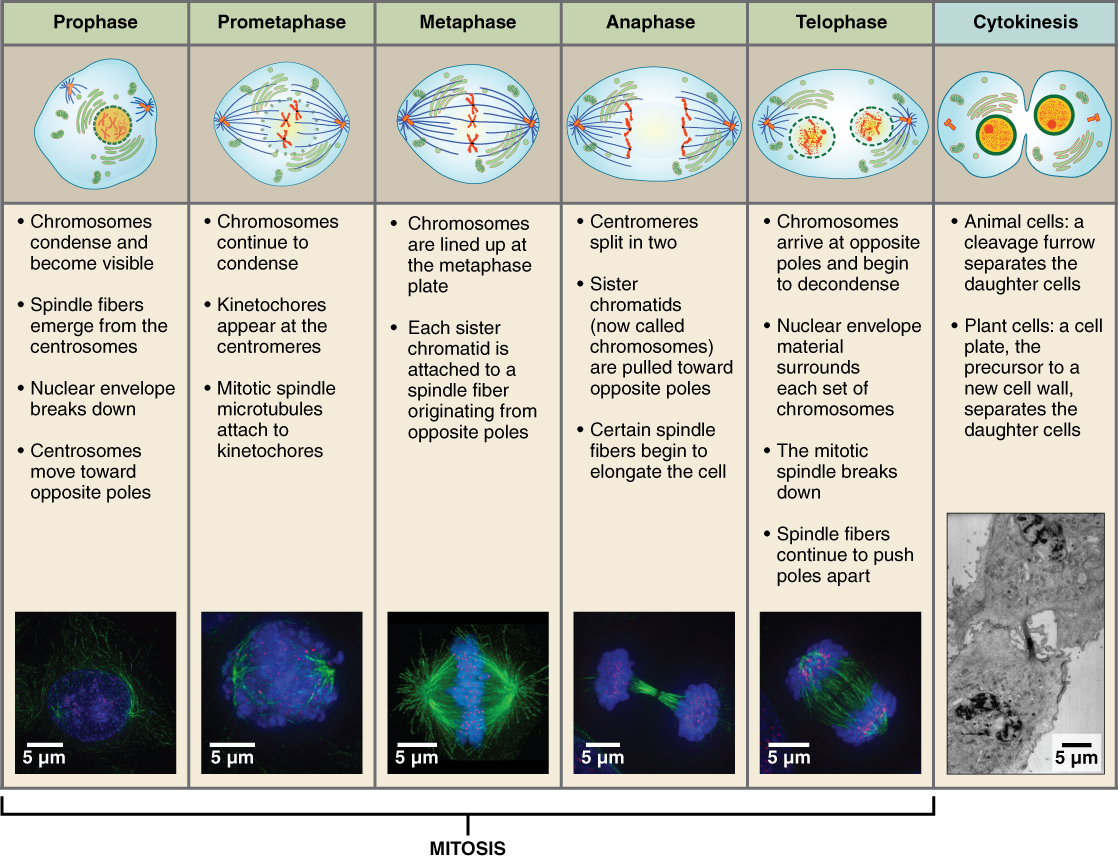
The stages of cell division oversee the separation of identical genetic material into two new nuclei, followed by the division of the cytoplasm. Animal cell mitosis is divided into five stages—prophase, prometaphase, metaphase, anaphase, and telophase—visualized here by light microscopy with fluorescence. Mitosis is usually accompanied by cytokinesis, shown here by a transmission electron microscope. (credit "diagrams": modification of work by Mariana Ruiz Villareal; credit "mitosis micrographs": modification of work by Roy van Heesbeen; credit "cytokinesis micrograph": modification of work by the Wadsworth Center, NY State Department of Health; donated to the Wikimedia foundation; scale-bar data from Matt Russell)
Prophase
Prophase is the first phase of mitosis, during which the loosely packed chromatin coils and condenses into visible chromosomes. During prophase, each chromosome becomes visible with its identical partner (sister chromatid) attached, forming the familiar X-shape of sister chromatids. The nucleolus disappears early during this phase, and the nuclear envelope also disintegrates.
A major occurrence during prophase concerns a very important structure that contains the origin site for microtubule growth. Cellular structures called centrioles that serve as origin points from which microtubules extend. These tiny structures also play a very important role during mitosis. A centrosome is a pair of centrioles together. The cell contains two centrosomes side-by-side, which begin to move apart during prophase. As the centrosomes migrate to two different sides of the cell, microtubules begin to extend from each like long fingers from two hands extending toward each other. The mitotic spindle is the structure composed of the centrosomes and their emerging microtubules.
Near the end of prophase there is an invasion of the nuclear area by microtubules from the mitotic spindle. The nuclear membrane has disintegrated, and the microtubules attach themselves to the centromeres that adjoin pairs of sister chromatids. The kinetochore is a protein structure on the centromere that is the point of attachment between the mitotic spindle and the sister chromatids. This stage is referred to as late prophase or “prometaphase” to indicate the transition between prophase and metaphase.
Metaphase
Metaphase is the second stage of mitosis. During this stage, the sister chromatids, with their attached microtubules, line up along a linear plane in the middle of the cell. A metaphase plate forms between the centrosomes that are now located at either end of the cell. The metaphase plate is the name for the plane through the center of the spindle on which the sister chromatids are positioned. The microtubules are now poised to pull apart the sister chromatids and bring one from each pair to each side of the cell.
Anaphase
Anaphase is the third stage of mitosis. Anaphase takes place over a few minutes, when the pairs of sister chromatids are separated from one another, forming individual chromosomes once again. These chromosomes are pulled to opposite ends of the cell by their kinetochores, as the microtubules shorten. Each end of the cell receives one partner from each pair of sister chromatids, ensuring that the two new daughter cells will contain identical genetic material.
Telophase
Telophase is the final stage of mitosis. Telophase is characterized by the formation of two new daughter nuclei at either end of the dividing cell. These newly formed nuclei surround the genetic material, which uncoils such that the chromosomes return to loosely packed chromatin. Nucleoli also reappear within the new nuclei, and the mitotic spindle breaks apart, each new cell receiving its own complement of DNA, organelles, membranes, and centrioles. At this point, the cell is already beginning to split in half as cytokinesis begins.
Cytokinesis
Cytokinesis is the second part of the mitotic phase during which cell division is completed by the physical separation of the cytoplasmic components into two daughter cells. Although the stages of mitosis are similar for most eukaryotes, the process of cytokinesis is quite different for eukaryotes that have cell walls, such as plant cells.
In cells such as animal cells that lack cell walls, cytokinesis begins following the onset of anaphase. A contractile ring composed of actin filaments forms just inside the plasma membrane at the former metaphase plate. The actin filaments pull the equator of the cell inward, forming a fissure. This fissure, or “crack,” is called the cleavage furrow. The furrow deepens as the actin ring contracts, and eventually the membrane and cell are cleaved in two (see the figure below).
In plant cells, a cleavage furrow is not possible because of the rigid cell walls surrounding the plasma membrane. A new cell wall must form between the daughter cells. During interphase, the Golgi apparatus accumulates enzymes, structural proteins, and glucose molecules prior to breaking up into vesicles and dispersing throughout the dividing cell. During telophase, these Golgi vesicles move on microtubules to collect at the metaphase plate. There, the vesicles fuse from the center toward the cell walls; this structure is called a cell plate. As more vesicles fuse, the cell plate enlarges until it merges with the cell wall at the periphery of the cell. Enzymes use the glucose that has accumulated between the membrane layers to build a new cell wall of cellulose. The Golgi membranes become the plasma membrane on either side of the new cell wall (see panel b in the figure below).
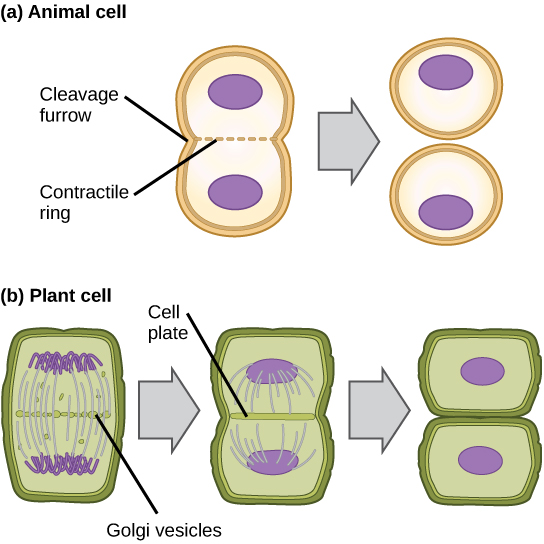
In part (a), a cleavage furrow forms at the former metaphase plate in the animal cell. The plasma membrane is drawn in by a ring of actin fibers contracting just inside the membrane. The cleavage furrow deepens until the cells are pinched in two. In part (b), Golgi vesicles coalesce at the former metaphase plate in a plant cell. The vesicles fuse and form the cell plate. The cell plate grows from the center toward the cell walls. New cell walls are made from the vesicle contents.
Cell Cycle Check Points
It is essential that daughter cells be nearly exact duplicates of the parent cell. Mistakes in the duplication or distribution of the chromosomes lead to mutations that may be passed forward to every new cell produced from the abnormal cell. To prevent a compromised cell from continuing to divide, there are internal control mechanisms that operate at three main cell cycle checkpoints at which the cell cycle can be stopped until conditions are favorable. These checkpoints occur near the end of G1, at the G2–M transition, and during metaphase (see figure below).
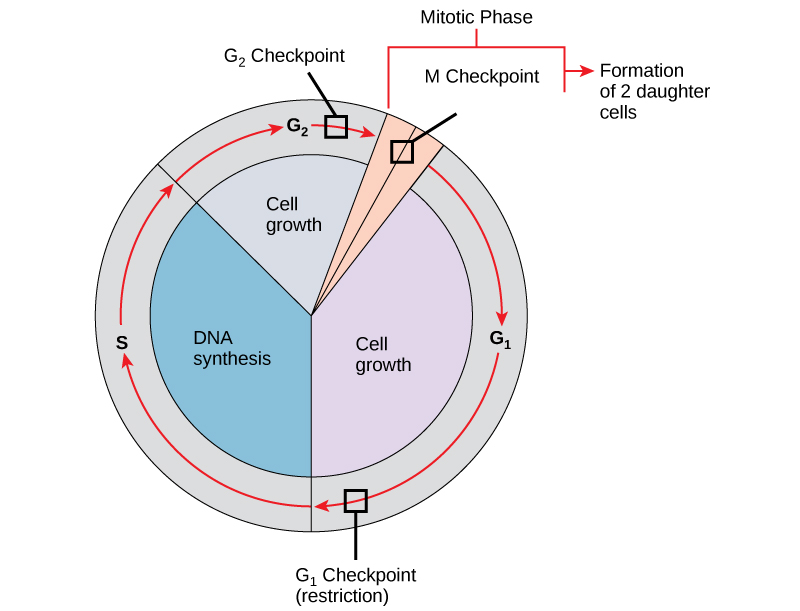
The cell cycle is controlled at three checkpoints. Integrity of the DNA is assessed at the G1 checkpoint. Proper chromosome duplication is assessed at the G2 checkpoint. Attachment of each kinetochore to a spindle fiber is assessed at the M checkpoint.
G1 Checkpoint
The G1 checkpoint determines whether all conditions are favorable for cell division to proceed into S phase where DNA replication occurs. The G1 checkpoint, also called the restriction point, is the point at which the cell irreversibly commits to the cell-division process. In addition to adequate reserves and cell size, there is a check for damage to the genomic DNA at the G1 checkpoint. A cell that does not meet all the requirements will not be released into the S phase.
G2 Checkpoint
The G2 checkpoint bars the entry to the mitotic phase if certain conditions are not met. As in the G1 checkpoint, cell size and protein reserves are assessed. However, the most important role of the G2 checkpoint is to ensure that all of the chromosomes have been replicated and that the replicated DNA is not damaged.
M Checkpoint
The M checkpoint occurs near the end of the metaphase stage of mitosis. The M checkpoint is also known as the spindle checkpoint because it determines if all the sister chromatids are correctly attached to the spindle microtubules. Because the separation of the sister chromatids during anaphase is an irreversible step, the cycle will not proceed until the kinetochores of each pair of sister chromatids are firmly anchored to spindle fibers arising from opposite poles of the cell.
Note
Watch what occurs at the G1, G2, and M checkpoints by visiting this animation of the cell cycle.
When the Cell Cycle gets out of Control
Most people understand that cancer or tumors are caused by abnormal cells that multiply continuously. If the abnormal cells continue to divide unstopped, they can damage the tissues around them, spread to other parts of the body, and eventually result in death. In healthy cells, the tight regulation mechanisms of the cell cycle prevent this from happening, while failures of cell cycle control can cause unwanted and excessive cell division. Failures of control may be caused by inherited genetic abnormalities that compromise the function of certain “stop” and “go” signals. Environmental insult that damages DNA can also cause dysfunction in those signals. Often, a combination of both genetic predisposition and environmental factors lead to cancer.
The process of a cell escaping its normal control system and becoming cancerous may actually happen throughout the body quite frequently. Fortunately, certain cells of the immune system are capable of recognizing cells that have become cancerous and destroying them. However, in certain cases the cancerous cells remain undetected and continue to proliferate. If the resulting tumor does not pose a threat to surrounding tissues, it is said to be benign and can usually be easily removed. If capable of damage, the tumor is considered malignant and the patient is diagnosed with cancer.
Homeostatic Imbalances: Cancer Arises from Homeostatic Imbalances
Cancer is an extremely complex condition, capable of arising from a wide variety of genetic and environmental causes. Typically, mutations or aberrations in a cell’s DNA that compromise normal cell cycle control systems lead to cancerous tumors. Cell cycle control is an example of a homeostatic mechanism that maintains proper cell function and health. While progressing through the phases of the cell cycle, a large variety of intracellular molecules provide stop and go signals to regulate movement forward to the next phase. These signals are maintained in an intricate balance so that the cell only proceeds to the next phase when it is ready. This homeostatic control of the cell cycle can be thought of like a car’s cruise control. Cruise control will continually apply just the right amount of acceleration to maintain a desired speed, unless the driver hits the brakes, in which case the car will slow down. Similarly, the cell includes molecular messengers, such as cyclins, that push the cell forward in its cycle.
In addition to cyclins, a class of proteins that are encoded by genes called proto-oncogenes provide important signals that regulate the cell cycle and move it forward. Examples of proto-oncogene products include cell-surface receptors for growth factors, or cell-signaling molecules, two classes of molecules that can promote DNA replication and cell division. In contrast, a second class of genes known as tumor suppressor genes sends stop signals during a cell cycle. For example, certain protein products of tumor suppressor genes signal potential problems with the DNA and thus stop the cell from dividing, while other proteins signal the cell to die if it is damaged beyond repair. Some tumor suppressor proteins also signal a sufficient surrounding cellular density, which indicates that the cell need not presently divide. The latter function is uniquely important in preventing tumor growth: normal cells exhibit a phenomenon called “contact inhibition;” thus, extensive cellular contact with neighboring cells causes a signal that stops further cell division.
These two contrasting classes of genes, proto-oncogenes and tumor suppressor genes, are like the accelerator and brake pedal of the cell’s own “cruise control system,” respectively. Under normal conditions, these stop and go signals are maintained in a homeostatic balance. Generally speaking, there are two ways that the cell’s cruise control can lose control: a malfunctioning (overactive) accelerator, or a malfunctioning (underactive) brake. When compromised through a mutation, or otherwise altered, proto-oncogenes can be converted to oncogenes, which produce oncoproteins that push a cell forward in its cycle and stimulate cell division even when it is undesirable to do so. For example, a cell that should be programmed to self-destruct (a process called apoptosis) due to extensive DNA damage might instead be triggered to proliferate by an oncoprotein. On the other hand, a dysfunctional tumor suppressor gene may fail to provide the cell with a necessary stop signal, also resulting in unwanted cell division and proliferation.
A delicate homeostatic balance between the many proto-oncogenes and tumor suppressor genes delicately controls the cell cycle and ensures that only healthy cells replicate. Therefore, a disruption of this homeostatic balance can cause aberrant cell division and cancerous growths.
Sexual Reproduction
Sexual reproduction was an early evolutionary innovation after the appearance of eukaryotic cells. The fact that most eukaryotes reproduce sexually is evidence of its evolutionary success. In many animals, it is the only mode of reproduction. And yet, scientists recognize some real disadvantages to sexual reproduction. On the surface, offspring that are genetically identical to the parent may appear to be more advantageous. If the parent organism is successfully occupying a habitat, offspring with the same traits would be similarly successful. There is also the obvious benefit to an organism that can produce offspring by asexual budding, fragmentation, or asexual eggs. These methods of reproduction do not require another organism of the opposite sex. There is no need to expend energy finding or attracting a mate. That energy can be spent on producing more offspring. Indeed, some organisms that lead a solitary lifestyle have retained the ability to reproduce asexually. In addition, in asexual populations every individual is capable of reproduction. In contrast, the males in sexual populations (half the population) are not producing offspring themselves. Because of this, an asexual population can in theory grow twice as fast as a sexual population. This means that in competition, the asexual population would have the advantage. All of these advantages to asexual reproduction, which are also disadvantages to sexual reproduction, should mean that the number of species with asexual reproduction should be more common.
However, multicellular organisms that exclusively depend on asexual reproduction are rare.
So why is sexual reproduction so common?
This is one of the important questions in biology and has been the focus of much research from the latter half of the twentieth century until now. A likely explanation is that the variation that sexual reproduction creates among offspring is very important to the survival and reproduction of those offspring. The only source of genetic variation in asexual organisms is mutation. In sexually reproducing organisms, mutations are continually reshuffled between generations when parents combine their unique genomes, and the genes are mixed into different combinations by the process of meiosis.
The Red Queen Hypothesis
There is no question that sexual reproduction provides evolutionary advantages to organisms that employ this mechanism to produce offspring. The problematic question is why, even in the face of seemingly stable conditions, sexual reproduction persists when it is more difficult and produces fewer offspring for individual organisms? Variation is the outcome of sexual reproduction, but why is ongoing variation necessary? Enter the Red Queen hypothesis, first proposed by Leigh Van Valen in 1973.1 The concept was named in reference to the Red Queen's race in Lewis Carroll's book, Through the Looking-Glass, in which the Red Queen says one must run at full speed just to stay where one is.
All species coevolve with other organisms. For example, predators coevolve with their prey, and parasites coevolve with their hosts. A remarkable example of coevolution between predators and their prey is the unique coadaptation of night flying bats and their moth prey. Bats find their prey by emitting high-pitched clicks, but moths have evolved simple ears to hear these clicks so they can avoid the bats. The moths have also adapted behaviors, such as flying away from the bat when they first hear it, or dropping suddenly to the ground when the bat is upon them. Bats have evolved “quiet” clicks in an attempt to evade the moth’s hearing. Some moths have evolved the ability to respond to the bats’ clicks with their own clicks as a strategy to confuse the bats echolocation abilities.
Each tiny advantage gained by favorable variation gives a species an edge over close competitors, predators, parasites, or even prey. The only method that will allow a coevolving species to keep its own share of the resources is also to continually improve its ability to survive and produce offspring. As one species gains an advantage, other species must also develop an advantage or they will be outcompeted. No single species progresses too far ahead because genetic variation among progeny of sexual reproduction provides all species with a mechanism to produce adapted individuals. Species whose individuals cannot keep up become extinct. The Red Queen’s catchphrase was, “It takes all the running you can do to stay in the same place.” This is an apt description of coevolution between competing species.
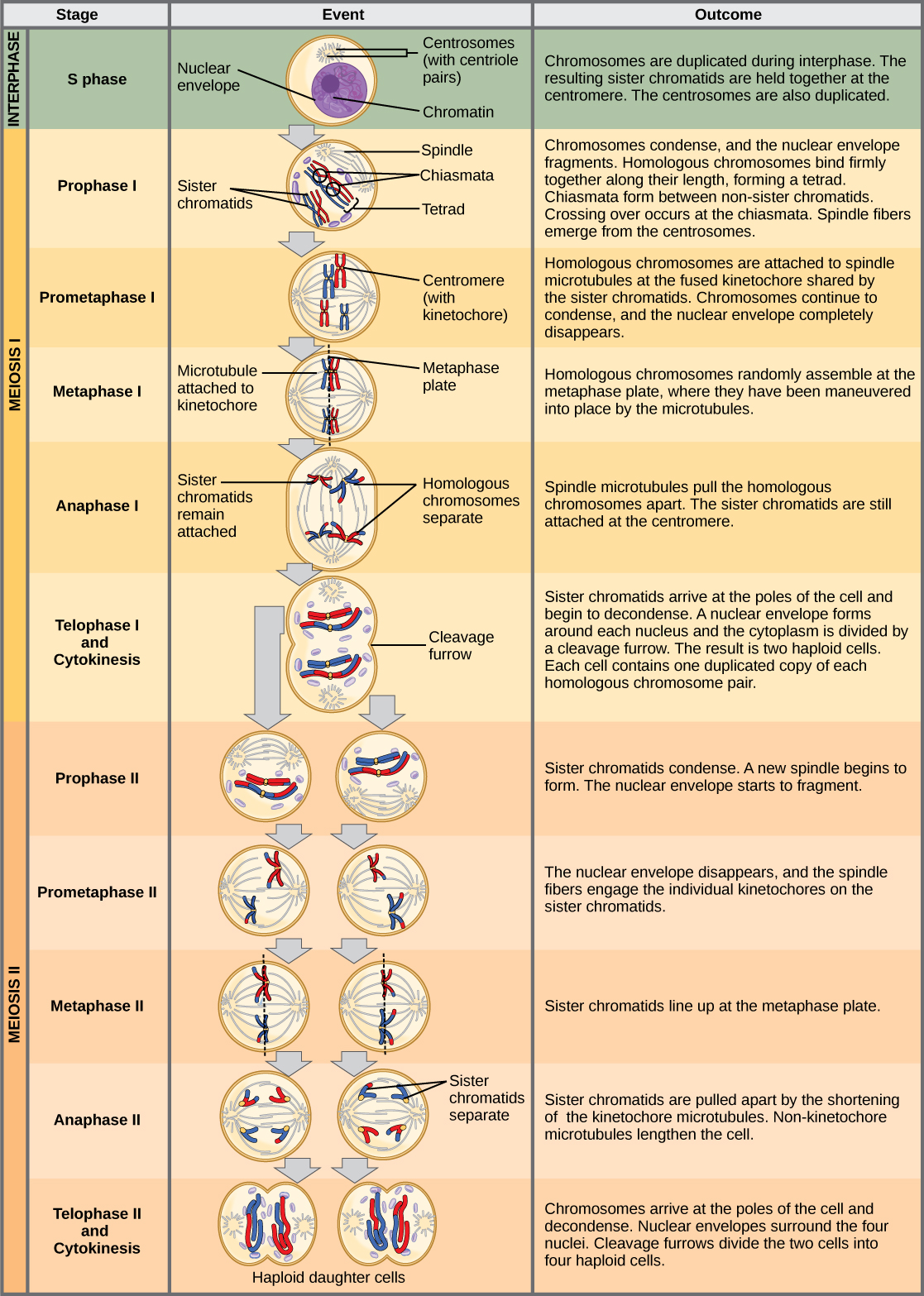
Meiosis
Sexual reproduction requires fertilization, the union of two cells from two individual organisms. If those two cells each contain one set of chromosomes, then the resulting cell contains two sets of chromosomes. Haploid cells contain one set of chromosomes, diploid cells contain two sets of chromosomes. The number of sets of chromosomes in a cell is called its ploidy level. If the reproductive cycle is to continue, then the diploid cell must somehow reduce its number of chromosome sets before fertilization can occur again, or there will be a continual doubling in the number of chromosome sets in every generation. So, in addition to fertilization, sexual reproduction includes a nuclear division that reduces the number of chromosome sets.
The nuclear division that forms haploid cells, which is called meiosis, is related to mitosis. In mitosis, both the parent and the daughter nuclei are at the same ploidy level—diploid for most plants and animals. Meiosis employs many of the same mechanisms as mitosis. However, the starting nucleus is always diploid and the nuclei that result at the end of a meiotic cell division are haploid. To achieve this reduction in chromosome number, meiosis consists of one round of chromosome duplication and two rounds of nuclear division. Because the events that occur during each of the division stages are analogous to the events of mitosis, the same stage names are assigned. However, because there are two rounds of division, the major process and the stages are designated with a “I” or a “II.” Thus, meiosis I is the first round of meiotic division and consists of prophase I, prometaphase I, and so on. Meiosis II, in which the second round of meiotic division takes place, includes prophase II, prometaphase II, and so on.
Meiosis I
Meiosis is preceded by an interphase consisting of the G1, S, and G2 phases, which are nearly identical to the phases preceding mitosis.
Prophase I
Early in prophase I, before the chromosomes can be seen clearly microscopically, homologous chromosomes are attached at their tips to the nuclear envelope by proteins. Homologous chromosomes are similar but not identical chromosomes. For example, chromosome 12 from your mother and chromosome 12 from your father will both be present inside each of your cells. Each chromosome 12 contains the same genes, usually in the same locations, however, each gene can be a different allele. Gene A on chromosome 12 from your mother may be allele R' and gene A on chromosome 12 from your father may be allele r. In species such as humans, even though the X and Y sex chromosomes are not homologous (most of their genes differ), they have a small region of homology that allows the X and Y chromosomes to pair up during prophase I. A partial synaptonemal complex develops only between the regions of homology. It will be very important to understand what homologous chromosomes are when following the process of meiosis.
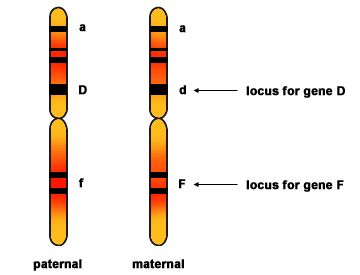
Two homologous chromsomes are shown prior to DNA replication. Each chromosome has three genes with their locus marked. Homologous chromosomes contain the same genes but are not identical. They each can contain different alleles of each gene.
Source: http://mrphome.net/mrp/Homologous_Chromosome.html
As the nuclear envelope begins to break down, the proteins associated with homologous chromosomes bring the pair close to each other. The synaptonemal complex, a lattice of proteins between the homologous chromosomes, first forms at specific locations and then spreads to cover the entire length of the chromosomes. The tight pairing of the homologous chromosomes is called synapsis. In synapsis, the genes on the chromatids of the homologous chromosomes are aligned precisely with each other. The synaptonemal complex supports the exchange of chromosomal segments between non-sister homologous chromatids, a process called crossing over. Crossing over can be observed visually after the exchange as chiasmata (singular = chiasma) (see figure below).
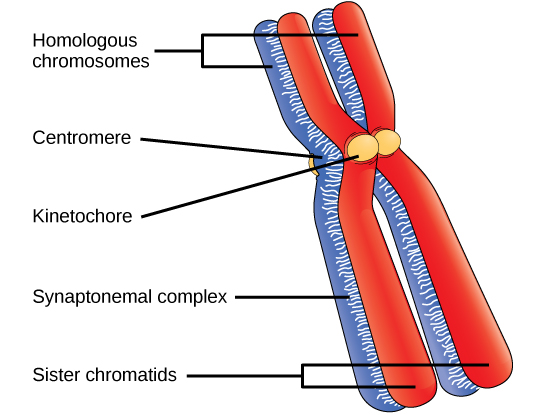
Early in prophase I, homologous chromosomes come together to form a synapse. The chromosomes are bound tightly together and in perfect alignment by a protein lattice called a synaptonemal complex and by cohesin proteins at the centromere.
Located at intervals along the synaptonemal complex are large protein assemblies called recombination nodules. These assemblies mark the points of later chiasmata and mediate the multistep process of crossover—or genetic recombination—between the non-sister chromatids. Near the recombination nodule on each chromatid, the double-stranded DNA is cleaved, the cut ends are modified, and a new connection is made between the non-sister chromatids. As prophase I progresses, the synaptonemal complex begins to break down and the chromosomes begin to condense. When the synaptonemal complex is gone, the homologous chromosomes remain attached to each other at the centromere and at chiasmata. The chiasmata remain until anaphase I. The number of chiasmata varies according to the species and the length of the chromosome. There must be at least one chiasma per chromosome for proper separation of homologous chromosomes during meiosis I, but there may be as many as 25. Following crossover, the synaptonemal complex breaks down and the cohesin connection between homologous pairs is also removed. At the end of prophase I, the pairs are held together only at the chiasmata (figure below) and are called tetrads because the four sister chromatids of each pair of homologous chromosomes are now visible.
The crossover events are the first source of genetic variation in the nuclei produced by meiosis. A single crossover event between homologous non-sister chromatids leads to a reciprocal exchange of equivalent DNA between a maternal chromosome and a paternal chromosome. Now, when that sister chromatid is moved into a gamete cell it will carry some DNA from one parent of the individual and some DNA from the other parent. The sister recombinant chromatid has a combination of maternal and paternal genes that did not exist before the crossover. Multiple crossovers in an arm of the chromosome have the same effect, exchanging segments of DNA to create recombinant chromosomes.
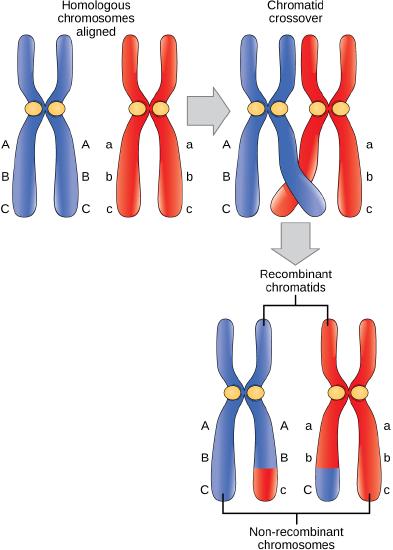
Crossover occurs between non-sister chromatids of homologous chromosomes. The result is an exchange of genetic material between homologous chromosomes.
Possible discussion
What are the major differences between Prophase I of Meiosis and Prophase of Mitosis?
Prometaphase I
The key event in prometaphase I is the attachment of the spindle fiber microtubules to the kinetochore proteins at the centromeres. Kinetochore proteins are multiprotein complexes that bind the centromeres of a chromosome to the microtubules of the mitotic spindle. Microtubules grow from centrosomes placed at opposite poles of the cell. The microtubules move toward the middle of the cell and attach to one of the two fused homologous chromosomes. The microtubules attach at each chromosomes' kinetochores. With each member of the homologous pair attached to opposite poles of the cell, in the next phase, the microtubules can pull the homologous pair apart. A spindle fiber that has attached to a kinetochore is called a kinetochore microtubule. At the end of prometaphase I, each tetrad is attached to microtubules from both poles, with one homologous chromosome facing each pole. The homologous chromosomes are still held together at chiasmata. In addition, the nuclear membrane has broken down entirely.
Metaphase I
During metaphase I, the homologous chromosomes are arranged in the center of the cell with the kinetochores facing opposite poles. The homologous pairs orient themselves randomly at the equator. For example, if the two homologous members of chromosome 1 are labeled a and b, then the chromosomes could line up a-b, or b-a. This is important in determining the genes carried by a gamete, as each will only receive one of the two homologous chromosomes. This is called Independent Assortment. Recall that homologous chromosomes are not identical, they contain slight differences in their genetic information, causing each gamete to have a unique genetic makeup.
This randomness is the physical basis for the creation of the second form of genetic variation in offspring. Consider that the homologous chromosomes of a sexually reproducing organism are originally inherited as two separate sets, one from each parent. Using humans as an example, one set of 23 chromosomes is present in the egg donated by the mother. The father provides the other set of 23 chromosomes in the sperm that fertilizes the egg. Every cell of the multicellular offspring has copies of the original two sets of homologous chromosomes. In prophase I of meiosis, the homologous chromosomes form the tetrads. In metaphase I, these pairs line up at the midway point between the two poles of the cell to form the metaphase plate. Because there is an equal chance that a microtubule fiber will encounter a maternally or paternally inherited chromosome, the arrangement of the tetrads at the metaphase plate is random. Any maternally inherited chromosome may face either pole. Any paternally inherited chromosome may also face either pole. The orientation of each tetrad is independent of the orientation of the other 22 tetrads.
This event—the random (or independent) assortment of homologous chromosomes at the metaphase plate—is the second mechanism that introduces variation into the gametes or spores. In each cell that undergoes meiosis, the arrangement of the tetrads is different. The number of variations is dependent on the number of chromosomes making up a set. There are two possibilities for orientation at the metaphase plate; the possible number of alignments therefore equals 2n, where n is the number of chromosomes per set. Humans have 23 chromosome pairs, which results in over eight million (223) possible genetically-distinct gametes. This number does not include the variability that was previously created in the sister chromatids by crossover. Given these two mechanisms, it is highly unlikely that any two haploid cells resulting from meiosis will have the same genetic composition (see figure below).
To summarize the genetic consequences of meiosis I, the maternal and paternal genes are recombined by crossover events that occur between each homologous pair during prophase I. In addition, the random assortment of tetrads on the metaphase plate produces a unique combination of maternal and paternal chromosomes that will make their way into the gametes.

Random, independent assortment during metaphase I can be demonstrated by considering a cell with a set of two chromosomes (n = 2). In this case, there are two possible arrangements at the equatorial plane in metaphase I. The total possible number of different gametes is 2n, where n equals the number of chromosomes in a set. In this example, there are four possible genetic combinations for the gametes. With n = 23 in human cells, there are over 8 million possible combinations of paternal and maternal chromosomes.
Anaphase I
In anaphase I, the microtubules pull the linked chromosomes apart. The sister chromatids remain tightly bound together at the centromere. The chiasmata are broken in anaphase I as the microtubules attached to the fused kinetochores pull the homologous chromosomes apart.
Possible discussion
What major difference occurs in Anaphase I of Meiosis compared to Anaphase of Mitosis?
Telophase I and Cytokinesis
In telophase, the separated chromosomes arrive at opposite poles. The remainder of the typical telophase events may or may not occur, depending on the species. In some organisms, the chromosomes decondense and nuclear envelopes form around the chromatids in telophase I. In other organisms, cytokinesis—the physical separation of the cytoplasmic components into two daughter cells—occurs without reformation of the nuclei. In nearly all species of animals and some fungi, cytokinesis separates the cell contents via a cleavage furrow (constriction of the actin ring that leads to cytoplasmic division). In plants, a cell plate is formed during cell cytokinesis by Golgi vesicles fusing at the metaphase plate. This cell plate will ultimately lead to the formation of cell walls that separate the two daughter cells.
Two haploid cells are the end result of the first meiotic division. The cells are haploid because at each pole, there is just one of each pair of the homologous chromosomes. Therefore, only one full set of the chromosomes is present. This is why the cells are considered haploid—there is only one chromosome set, even though each homolog still consists of two sister chromatids. Recall that sister chromatids are merely duplicates of one of the two homologous chromosomes (except for changes that occurred during crossing over). In meiosis II, these two sister chromatids will separate, creating four haploid daughter cells.
Meiosis II
In some species, cells enter a brief interphase, or interkinesis, before entering meiosis II. Interkinesis lacks an S phase, so chromosomes are not duplicated. The two cells produced in meiosis I go through the events of meiosis II in synchrony. During meiosis II, the sister chromatids within the two daughter cells separate, forming four new haploid gametes. The mechanics of meiosis II is similar to mitosis, except that each dividing cell has only one set of homologous chromosomes. Therefore, each cell has half the number of sister chromatids to separate out as a diploid cell undergoing mitosis.
Prophase II
If the chromosomes decondensed in telophase I, they condense again. If nuclear envelopes were formed, they fragment into vesicles. The centrosomes that were duplicated during interkinesis move away from each other toward opposite poles, and new spindles are formed.
Prometaphase II
The nuclear envelopes are completely broken down, and the spindle is fully formed. Each sister chromatid forms an individual kinetochore that attaches to microtubules from opposite poles.
Metaphase II
The sister chromatids are maximally condensed and aligned at the equator of the cell.
Anaphase II
The sister chromatids are pulled apart by the kinetochore microtubules and move toward opposite poles. Non-kinetochore microtubules elongate the cell.
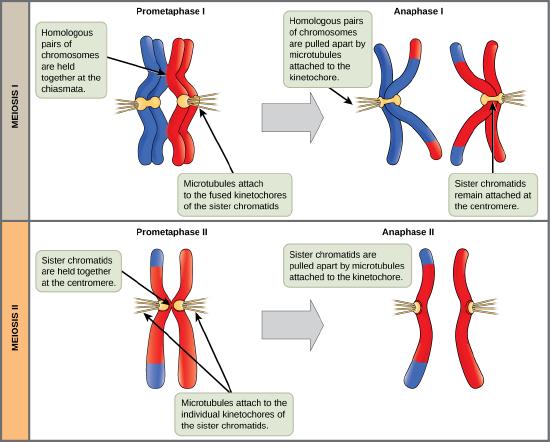
The process of chromosome alignment differs between meiosis I and meiosis II. In prometaphase I, microtubules attach to the fused kinetochores of homologous chromosomes, and the homologous chromosomes are arranged at the midpoint of the cell in metaphase I. In anaphase I, the homologous chromosomes are separated. In prometaphase II, microtubules attach to the kinetochores of sister chromatids, and the sister chromatids are arranged at the midpoint of the cells in metaphase II. In anaphase II, the sister chromatids are separated.
Telophase II and Cytokinesis
The chromosomes arrive at opposite poles and begin to decondense. Nuclear envelopes form around the chromosomes. Cytokinesis separates the two cells into four unique haploid cells. At this point, the newly formed nuclei are both haploid. The cells produced are genetically unique because of the random assortment of paternal and maternal homologs and because of the recombining of maternal and paternal segments of chromosomes (with their sets of genes) that occurs during crossover. The entire process of meiosis is outlined in the figure below.
An animal cell with a diploid number of four (2n = 4) proceeds through the stages of meiosis to form four haploid daughter cells.
Comparing Mitosis and Meiosis
Mitosis and meiosis are both forms of division of the nucleus in eukaryotic cells. They share some similarities, but also exhibit distinct differences that lead to very different outcomes. Mitosis is a single nuclear division that results in two nuclei that are usually partitioned into two new cells. The nuclei resulting from a mitotic division are genetically identical to the original nucleus. They have the same number of sets of chromosomes, one set in the case of haploid cells and two sets in the case of diploid cells. In most plants and all animal species, it is typically diploid cells that undergo mitosis to form new diploid cells. In contrast, meiosis consists of two nuclear divisions resulting in four nuclei that are usually partitioned into four new cells. The nuclei resulting from meiosis are not genetically identical and they contain one chromosome set only. This is half the number of chromosome sets in the original cell, which is diploid.
The main differences between mitosis and meiosis occur in meiosis I, which is a very different nuclear division than mitosis. In meiosis I, the homologous chromosome pairs become associated with each other, are bound together with the synaptonemal complex, develop chiasmata and undergo crossover between sister chromatids, and line up along the metaphase plate in tetrads with kinetochore fibers from opposite spindle poles attached to each kinetochore of a homolog in a tetrad. All of these events occur only in meiosis I.
When the chiasmata resolve and the tetrad is broken up with the homologs moving to one pole or another, the ploidy level—the number of sets of chromosomes in each future nucleus—has been reduced from two to one. For this reason, meiosis I is referred to as a reduction division. There is no such reduction in ploidy level during mitosis.
Meiosis II is much more analogous to a mitotic division. In this case, the duplicated chromosomes (only one set of them) line up on the metaphase plate with divided kinetochores attached to kinetochore fibers from opposite poles. During anaphase II, as in mitotic anaphase, the kinetochores divide and one sister chromatid—now referred to as a chromosome—is pulled to one pole while the other sister chromatid is pulled to the other pole. If it were not for the fact that there had been crossover, the two products of each individual meiosis II division would be identical (like in mitosis). Instead, they are different because there has always been at least one crossover per chromosome. Meiosis II is not a reduction division because although there are fewer copies of the genome in the resulting cells, there is still one set of chromosomes, as there was at the end of meiosis I.

Meiosis and mitosis are both preceded by one round of DNA replication; however, meiosis includes two nuclear divisions. The four daughter cells resulting from meiosis are haploid and genetically distinct. The daughter cells resulting from mitosis are diploid and identical to the parent cell.
The Mystery of the Evolution of Meiosis
Some characteristics of organisms are so widespread and fundamental that it is sometimes difficult to remember that they evolved like other simpler traits. Meiosis is such an extraordinarily complex series of cellular events that biologists have had trouble hypothesizing and testing how it may have evolved. Although meiosis is inextricably entwined with sexual reproduction and its advantages and disadvantages, it is important to separate the questions of the evolution of meiosis and the evolution of sex, because early meiosis may have been advantageous for different reasons than it is now. Thinking outside the box and imagining what the early benefits from meiosis might have been is one approach to uncovering how it may have evolved.
Meiosis and mitosis share obvious cellular processes and it makes sense that meiosis evolved from mitosis. The difficulty lies in the clear differences between meiosis I and mitosis. Adam Wilkins and Robin Holliday2 summarized the unique events that needed to occur for the evolution of meiosis from mitosis. These steps are homologous chromosome pairing, crossover exchanges, sister chromatids remaining attached during anaphase, and suppression of DNA replication in interphase. They argue that the first step is the hardest and most important, and that understanding how it evolved would make the evolutionary process clearer. They suggest genetic experiments that might shed light on the evolution of synapsis.
There are other approaches to understanding the evolution of meiosis in progress. Different forms of meiosis exist in single-celled protists. Some appear to be simpler or more “primitive” forms of meiosis. Comparing the meiotic divisions of different protists may shed light on the evolution of meiosis. Marilee Ramesh and colleagues 3 compared the genes involved in meiosis in protists to understand when and where meiosis might have evolved. Although research is still ongoing, recent scholarship into meiosis in protists suggests that some aspects of meiosis may have evolved later than others. This kind of genetic comparison can tell us what aspects of meiosis are the oldest and what cellular processes they may have borrowed from in earlier cells.
Link to Learning
Click through the steps of this interactive animation to compare the meiotic process of cell division to that of mitosis: How Cells Divide.
FOOTNOTES
- Leigh Van Valen, “A new evolutionary law,” Evolutionary Theory 1 (1973): 1–30.
- Adam S. Wilkins and Robin Holliday, “The Evolution of Meiosis from Mitosis,” Genetics 181 (2009): 3–12.
- Marilee A. Ramesh, Shehre-Banoo Malik and John M. Logsdon, Jr, “A Phylogenetic Inventory of Meiotic Genes: Evidence for Sex in Giardia and an Early Eukaryotic Origin of Meiosis,” Current Biology 15 (2005):185–91.