You will study a signal transduction protein and their interaction domains using a variety of web-based proteomics programs. For most of these programs you will need to input the amino acid sequence in FASTA format. Select a PDB code for a protein from the table at the end of this section. You could also use these programs to study any protein in the PDB.
Getting the FASTA sequence
1. First go to the PDB. Input the name of your protein (which has an interaction domain) in the search box. Limit the search to homo sapiens. Pick from the list of protein structure files the most appropriate one. The example below is for the 2YYN pdb code.
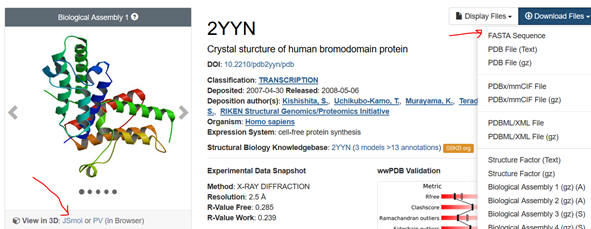
2. Select the Download Files dropdown and save the FASTA sequence to your home directory. Download the file as a Wordpad. You might have to remove recurring sections that don’t correspond to the single letter amino acid sequence or identical sequences if the structure consists of identical subunits To see if that might be the case, select JSmol (see figure above), rotate the structure with your mouse to see if there are multiple chains, and hover the mouse over the chains to see how the amino acids in that chain are labeled. You might see [TRP]33A: for example, where A indicates a separate A chain. Move to other chains. Then go to the Wordpad version of the FASTA sequences. You can examine the chains to see if the chains are identical. If so delete all but the first. See the above FASTA link for help.
I. Prediction of Protein Properties from Sequence Data
Use the following programs to gain information about your protein. Snip (with snipping tool for example) and paste a bit of relevant info from each program (using Snipping Tool) into this DOCX file and save it into the folder and upload it into Sharepoint. Name the file Lastname_LastName_FirstInitial_WebInteraction. If you have any problem with any of the programs (lots of error messages), skip that particular program. Several of them do the same type of analyzes. Compare the result. Snip and paste sufficient content to show that you complete the question. Write answers when asked to interpret the output.
a. Sequence Manipulation Suite: Determine the molecular weight of the protein.
b. Eukaryotic Linear Motif: Linear motifs are short, evolutionarily intrinsically disordered section of regulatory proteins and provide low-affinity interaction interfaces. These compact modules play central roles in mediating every aspect of the regulatory functionality of the cell. They are particularly prominent in mediating cell signaling, controlling protein turnover and directing protein localization. The Eukaryotic Linear Motif (ELM) provides the biological community with a comprehensive database of known experimentally validated motifs, and an exploratory tool to discover putative linear motifs in user-submitted protein sequences. Snip and paste the top of the output that shows the IUPRED showing the disorder/order graph.
c. TargetP 1.1 : predicts the subcellular location of eukaryotic protein. Snip and paste the results. Interpret them based on this link. Where is your protein likely found?
d. NET-NES 1.1 Server:: predicts leucine-rich nuclear export signals (NES) in eukaryotic protein This link will help you explain the output. Does yours?
e. NLSdb -- Database of nuclear localization signals: Search for information on nuclear localization signals (NLSs) and nuclear proteins. Select Query. Input the PDB code and select NL. Does yours?
f. NetPhos 2.0 server: produces neural network predictions for serine, threonine and tyrosine phosphorylation sites in eukaryotic proteins. (other cool prediction programs from this site)
g. TMPRED: The TMpred program makes a prediction of membrane-spanning regions and their orientation. The algorithm is based on the statistical analysis of TMbase, a database of naturally occurring transmembrane proteins. The prediction is made using a combination of several weight-matrices for scoring. Paste in your FASTA sequence but remove the header before running. Does it have a transmembrane helix?
h. TopPred 1.1 – Topology predictor for membrane proteins at the Pasteur Institute. You will have to input your email address. Paste in the entire FASTA file. Does it have transmembrane helices? (Part of Mobyle)
Save the first graph (PNG graphic file) of the output, open it with Adobe Photoshop, and paste the image into your report. Does the graph show alternating hydrophobic (+ values)/hydrophilic (- values) sections consistent with transmembrane helices (for example you would expect to see 7 hydrophobic stretches for GPCR)?
i. PFAM – multiple analyses of Protein FAMilies. This program looks at the domain organization of a protein sequence. Input the pdb code. When finished, select “sequences” in the list below.

Then select the human sequence. Snip the resulting diagram and legend showing the domain structure of the protein. You can also click on each domain in the diagram to get more info on the domain. Does the protein have the domain suggested in the beginning table?
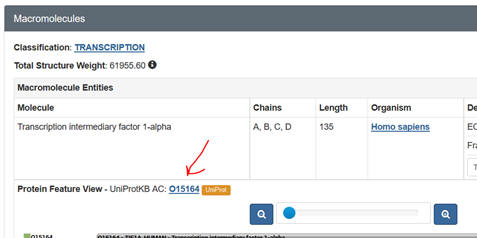
j. Prosite: Input your FASTA sequence in the Quick Scan mode. Select Exclude motifs with a high probability of occurrence from the scan. Snip and Paste the Hits by Proifle domain structure. Sometimes you might need a different code number, the UniProtKB: Accession number. Get this from the PDB web page as shown below:
k. eFindSite: is a ligand binding site prediction and virtual screening algorithm that detects common ligand binding sites. Put in the PDB code and then the pdb file you downloaded.
l. eFindSitePPI: detects protein binding sites and residues using meta-threading. It also predicts interfacial geometry and specific interactions stabilizing protein-protein complexes, such as hydrogen bonds, salt bridges, aromatic and hydrophobic interactions
m. NCBI Standard Protein BLAST: Input the FASTA file. The output shows the domain and domain superfamily followed by other protein sequences nearly identical to your protein. The results are graphical followed by descriptive. Snip domain structure with the closest aligned sequences. Then select under PDB structures the pdb code (example below 1xww).
You will see a window similar to below. Select under domain 1xwwwA00 (as an example). :
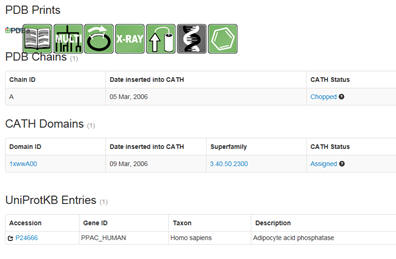
Then select the UniProtKB accession number. Confirm the many of the predictions you made above.
n. Predict Protein Open: Physiochemical properties of your protein. You will have to provide your email address. When complete you can access much of what you learned above by the links to the left under the Dashboard.
II. Visualizing Protein Interactions
It is important to be able to visualize the binding interactions between the targeted domain and the ligand (small molecule, PTM modified protein, protein or DNA). Here are some programs that allow that. Note: which programa you select will depend on if your protein is bound to a small ligand or to another protein or other macromolecule, in which case you need to explore protein interaction interfaces.
LIGAND:PROTEIN INTERACTIONS
Assignment: You will study the interaction of Protein Kinase C (PKC) with the ligand phorbal ester, which is a mimic of the 2nd messenger diacylglyceride with two programs: Ligand Explorer and Protein-Ligand Interaction Profilers
a. Ligand Explorer is a Java-based program. It probably will NOT work on a Mac running Safari. You will need the latest version of Java to run it. Try the various computer labs around campus as well. Go to the PDB page for your protein. After you input the pdb code, scroll down to Small Molecules section in the middle of the displayed page for that complex. There are links for both 2D and 3D visualization of the interactions. .
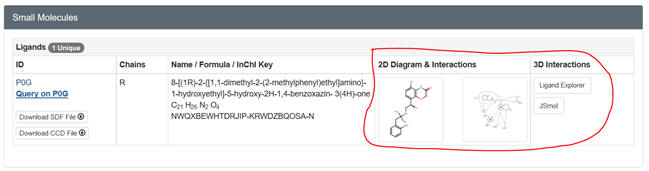
Select the 2D plot showing the interactions. The select Jsmol to see the ligand with a binding surface) interacting with contact residues in the protein. You can select a white background and toggle on and off H bonds. SNIP and PASTE.
Now select Ligand Explorer for a more detailed view. Make sure to select the correct ligand (see table below). You may be prompted to allow pop-ups form the site. If so, allow it. You may have to reselect Ligand Explorer again to start the program. Keep giving permissions and following prompts until Ligand Explorer is open. Once launched, select open this link in a new tab or window and instructions will open in a browser. Use the mouse to help find the best view of the interactions.
Select in turn hydrogen bond, hydrophobic, bridged H bond (mediated by a water molecule) and metal interaction (shown on the left hand side. Select Label Interactions by Distance. Take a cropped screenshot of each interaction (see instructions below).
For the final rendering, move the toggle on the Select Surfaces to opaque. Then change the distance in the best way to show the cavity in which the ligand binds. Color by hydrophobicity which gives two colors representing nonpolar and polar parts of cavity. Select solid surfaces
b. Protein-Ligand Interaction Profilers: Input the name of your PDB file. After the run is complete, select SMALL MOLECULE and then the appropriate ligand. You will get a 2D representation you can snip and paste. Then select Pymol 3D view (first 5 computers in ASC 135). You will see an interactive rendering of a small bound ligand and the protein residues it contacts in the complex. You can get a free student download of Pymol for your own computer. Snip and Paste relevant info.
PROTEIN:MACROMOLECULE SURFACE INTERACTIONS
You will study protein:protein interactions between a Src domain and small phospho-Tyr peptide using InterProSurf and COCOMAPS.
a. InterProSurf: Reports numbers of surface and buried atoms for each chain, and areas for each residue deemed to be in the interface. Select PDB Complex in the top menu tabs and input your pdb file. This gives numerical data only. Snip and Paste relevant info.
b. COCOMAPS: analyzes and visualizes interfaces in biological complexes (such as protein-protein, protein-DNA and protein-RNA complexes). Input the PDB file name and then the chains within the PDB file that you wish to see the interaction surface. Put in the letter for one of the interacting chains you selected into the first input box and the second letter into the second box. Detailed results will appear in graphical and tabular form.

A great way to visualize the binding interface is to download the new .pdb and .pml files and open the pdb file in Pymol . Once the PDB file is opened in Pymol, select file -> run -> script_name.pml. Snip and Paste relevant info.
Table: Signaling Proteins for Analysis
| Domains in Signaling Molecules |
| Domain | Binding Target | Cellular Process | Example protein | Pdb file |
| Bromo | Acetyl-Lys | Chromatin reg. | BRD4 | 2YYN |
| C1 | diacylglycerol | Plasma memb recruitment | Raf-1 | 3OMV |
| C2 | Phospholipid (Ca dependent) | Membrane targeting, vesicle trafficking | PRKCA | 3IW4 |
| CARD | Homotypic interactins | apoptosis | CRADD | 3CRD |
| Chromo | Methyl-Lys | Chromo reg, gene txn | CBX1 | 3F2U |
| Death (DD) | Homotypic inter. | Apoptosis | Fas | 3EZQ |
| DED | Homotypic inter. | Apoptosis | Caspase 8 | 1F9E |
| DEP | Memb, GPCRs | Sig trans, prot trafficking | Dsh human dishevelled 2 | 2REY |
| GRIP | Arf/Art G prot | Golgi traffic | Golgin-97 (Golga5) | 1R4A |
| PDZ | C-term peptide motifs | Diverse, scaffolding | PSD-95 Or discs large homolog 4 | 1L6O |
| PH | Phospholipids | Membrane recuirt | Akt | 1O6L 3CQW |
| PTB | Phosphor-Y | Y kinase signaling | Shc 1 SHC-transforming protein 1 | 1UEF 1irs europe |
| RGS | GTP binding pocket of Galpha | Sig trans | RGS4 | 1EZT |
| SH2 | phosphoY | pY-signaling | Src | 4U5W |
| SH3 | Pro-rich sequence | Diverse, cytoskelet | Src | 2PTK |
| TIR | Homo/Heterotypic | Cytokine and immune | TLR4 | 3VQ2 |
| TRAF | TNF signaling | Cell survival | TRAF-1 | 3ZJB |
| VHL | hydroxyPro | ubiquitinylation | VHL | 1VCB |
| Protein Ligand and Protein Protein Interactions |
| Protein (PKC) :Ligand (phorbal ester mimic of 2nd messenger diacylglyceride with Ligand Explorer and
Protein-Ligand Interaction Profilers | 1PTR |
| Protein (Chain E-Src fragment) : Protein (Chain I – phospho-peptide) with COCOMAPS
| 1QG1 |
| H-Ras-GppNHp bound to the Ras binding domain (RBD) of Raf Kinase GppNHp binding with Ligand Explorer and Protein-Ligand Interaction Profilers Protein (Ras, chain A):Protein (RBD-Raf, Chain B) interactions with COCOMAPS | 4G0N |
Contributors and Attributions








