12.1.1: Carbonates
- Page ID
- 131982
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Carbonates are a group of minerals that include the carbonate ion \(\left(\text{CO}_{3}^{2-}\right)\) as an essential structural unit (Deer et al., 1992). Among the most common are calcite \(\left(\text{CaCO}_{3}\right)\), aragonite \(\left(\text{CaCO}_{3}\right)\), magnesite \(\left(\text{MgCO}_{3}\right)\), dolomite \(\left(\text{CaMg} \left(\text{CO}_{3}\right)_{2}\right)\), siderite \(\left(\text{FeCO}_{3}\right)\), rhodochrosite \(\left(\text{MnCO}_{3}\right)\), strontionite \(\left(\text{SrCO}_{3}\right)\), and witherite \(\left(\text{BaCO}_{3}\right)\) (Deer et al., 1992). These minerals function as important pH buffers in geochemical systems and major carbon reservoirs in the global carbon cycle. They function as cements for sedimentary rocks. Carbonate biominerals also serve as a marker for the emergence of life on Earth. Specifically, stromatolites are among the earliest evidence of life in the rock record and their layered structures form by sediment trapping and carbonate biomineralization on microbial mats (Grotzinger and Knoll, 1999) (Fig. \(12.2\text{A, B}\)).

Image sources: (A) Paul Harrison, https://commons.wikimedia.org/w/index.php?curid=714512; (B) Michael C. Rygel, https://commons.wikimedia.org/wiki/File:Stromatolites_hoyt_mcr1.JPG; (C), https://commons.wikimedia.org/wiki/File:Gephyrocapsa_oceanica.jpg; (D - F) Thomas et al. (2000)
Microorganisms can help form carbonate minerals by generating surfaces that promote nucleation (Fig. \(11.2\)). In the microbial mats of modern carbonate stromatolites, for example, extracellular polymeric substances (EPS) produced by cyanobacteria and other microorganisms can serve as sites for mineral nucleation. Negatively charged functional groups on the EPS can strongly bind calcium and other cations from the seawater. This binding of calcium ions can actually inhibit precipitation by decreasing the availability of calcium for precipitation. However, if the EPS becomes saturated with calcium or if the EPS is altered and reorganized, it can then serve as a template for carbonate precipitation (Dupraz and Visscher, 2005).
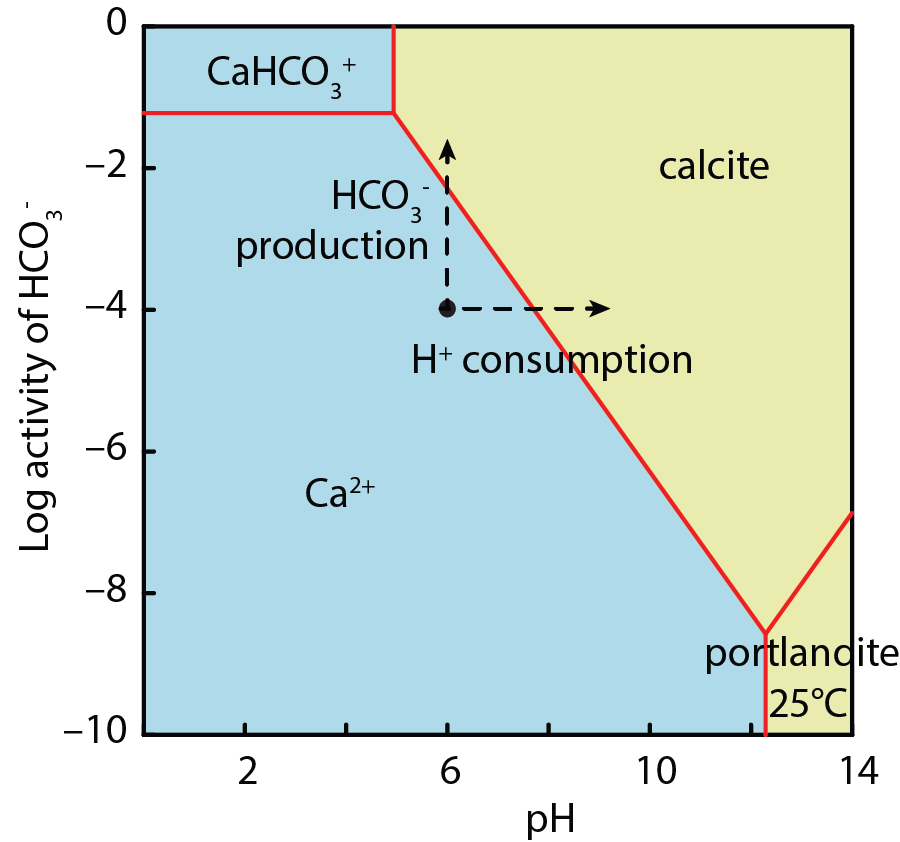
Coupled with these nucleation effects, microorganisms can also induce carbonate mineralization by altering the chemistry of their environment in ways that cause carbonate minerals to become supersaturated. They may, for example, cause supersaturation by producing bicarbonate or consuming hydrogen ions (i.e., increasing pH) (Fig. \(12.3\)), as discussed in Section 11.3. In addition, microbial reactions can also help supply cations that precipitate in carbonate minerals. For example, microorganisms can supply divalent metal cations by reducing (oxyhydr)oxide minerals and by degrading EPS that has inhibited carbonate precipitation by sequestering calcium and magnesium ions (Dupraz and Visscher, 2005).
Many microbial reactions include hydrogen ions, either as a reactant or product. Recall from Chapter 4 that we often add hydrogen ions when we balance redox reactions. Moreover, many microorganisms generate bicarbonate by oxidizing organic carbon, and most are thought to live within biofilms (Costerton et al., 1995; Flemming and Wuertz, 2019). Taken together, these observations indicate that the ability to induce carbonate mineralization is widespread across microbial life. Indeed, this possibility has been recognized for many years based on observations from cultures (Boquet et al., 1973). In addition to induced carbonate mineralization, some microorganisms intentionally form calcium carbonate phases. Notable examples of biologically controlled carbonate mineralization include coccolithophores and foraminifera, groups that commonly construct their shells (or tests) out of calcium
https://commons.wikimedia.org/wiki/File:Calcium_speciation.png


