12.6: Transport on the Cytoskeleton
- Page ID
- 16168
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
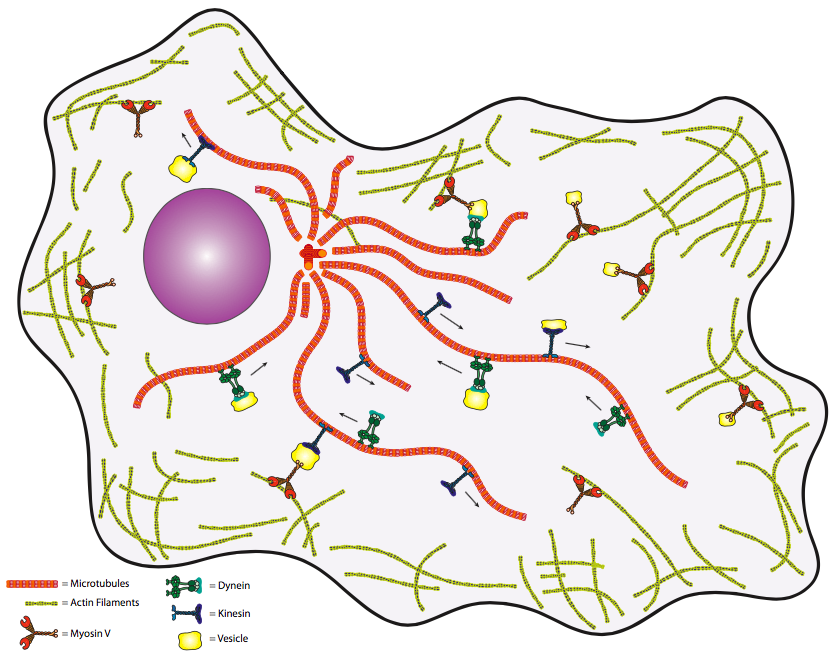
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)While it can be useful to think of these cytoskeletal structures as analogous to an animal skeleton, perhaps a better way to remember the relative placement of the microtubules and microfilaments is by their function in transporting intracellular cargo from one part of the cell to another. By that analogy, we might consider the microtubules to be a railroad track system, while the microfilaments are more like the streets. By the same analogy, we can suggest that the microtubule network and microfilament network are connected at certain points so that when cargo reaches its general destination by microtubule (rail), then it can be taken to its specific address by microfilament. Let’s extend this analogy a bit further. If the microtubules and microfilaments are the tracks and streets, then what are the trains and trucks? Ah, an astute question, Grasshopper. On the microtubules, the “trains” are one of two families of molecular motors: the kinesins and the dyneins.
We can generalize somewhat and say that the kinesins drive towards the (+) end (toward periphery of cell) while the dyneins go toward the (-) end (toward the MTOC). On actin microfilaments, the molecular motors are proteins of the myosin family. At this point, the analogies end, as the functioning of these molecular motors is very different from locomotion by train or truck. Finally, one might question the biological need for such a transport system. Again, if we analogize to human transport, then we could say that transport via simple diffusion is akin to people carrying packages randomly about the cell. That is to say, the deliveries will eventually be made, but you wouldn’t want to count on this method for time-critical materials. Thus a directed, high-speed system is needed to keep cells (particularly larger, eukaryotic cells) alive.
Although this type of transport occurs in all eukaryotic cells, a particularly well-studied case is axonal transport (also called axoplasmic transport) in neurons. Here, the transport of materials from the cell body (soma) to the tips of the axons can sometimes traverse very long distances up to several meters in larger animals, and must do so in a timely manner. Axonal transport is generally classified as anterograde (from soma to axon terminal) or retrograde (from terminals back). The types of material transported in these two directions is very different: much of the anterograde transport is protein building blocks for extending the axon or synaptic vesicles containing neurotransmitters; retrograde transport is mostly endocytic vesicles and signaling molecules. Axonal transport is also categorized as fast and slow. Slow transport is primarily the movement of proteins directly bound to the motors, and they can move from from 100 mm per day (SCa, slow component a) up to 3mm/day (SCb). In comparison, fast transport is generally movement of vesicles, and can vary from 50 to 400 mm/day. The mechanism of slow transport had been debated for over a decade until 2000, when direct visualization of fluorescently labeled neurofilaments in transport showed that the actual movement of the proteins was very similar to the movement in fast axonal transport, but there were many pauses in the transport, a “stop and go” mechanism rather moving from source to destination continuously.
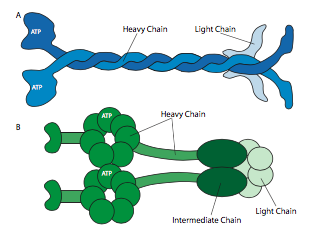
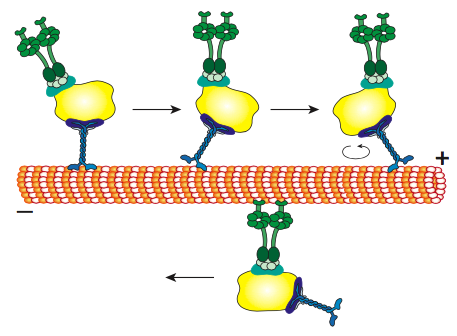
All of the kinesins and dyneins have a few key commonalities. There is a catalytic energy-releasing “head” connected to a hinge or neck region allowing the molecule to flex or “step”, and there is a cargo-carrying tail beyond that (Figure \(\PageIndex{8}\)). The head of a kinesin or dynein catalyzes the hydrolysis of ATP, releasing energy to change its conformation relative to the neck and tail of the molecule, allowing it to temporarily release its grip on the microtubule, swivel its “hips” around to plant itself a “step” away, and rebind to the microtubule (Figure \(\PageIndex{9}\)). On the actin microfilaments, the myosins, of which there are also many types (some depicted in Figure \(\PageIndex{10}\)) are the molecular motors. Their movement is different from dyneins and kinesins, as will be described in the next section, but also uses the energy of ATP hydrolysis to provide energy for the conformational changes needed for movement. We have introduced the motors, but considering the enormous diversity in the molecules that need to be transported around a cell, it would be impossible for the motors to directly bind to all of them. In fact, the motors bind to their cargo via adapter molecules that bind the motor on one side, and a cargo molecule or vesicle on the other. Further examination of the cargo and the routing of the cargo by address markers (SNAREs) was discussed in the vesicular transport chapter.