6.6: The Krebs/TCA/Citric acid cycle
- Page ID
- 16448
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Glycolysis through fermentative reactions produces ATP anaerobically. The evolution of respiration (the aerobic use of oxygen to efficiently burn nutrient fuels) had to wait until photosynthesis created the oxygenic atmosphere we live in now. Read more about the source of our oxygenic atmosphere in Dismukes GC et al. [(2001) The origin of atmospheric oxygen on earth: the innovation of oxygenic photosynthesis. Proc. Nat. Acad. Sci. USA 98:2170-2175].
The Krebs cycle is the first pathway of oxygenic respiration. Evolution of this respiration and the chemical bridge from glycolysis to the Krebs cycle, no doubt occurred a few reactions at a time, perhaps at first as a means of protecting anaerobic cells from the ‘poisonous’ effects of oxygen. Later, natural selection fleshed out the aerobic Krebs cycle, electron transport and oxidative phosphorylation pathways we see today.
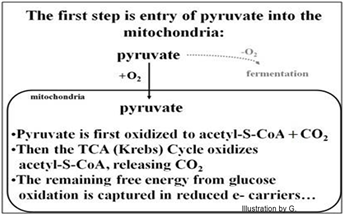
Whatever its initial utility, these reactions were an adaptive response to the increase in oxygen in the earth’s atmosphere. As a pathway for getting energy out of nutrients, respiration is much more efficient than glycolysis. Animals rely on it, but even plants and photosynthetic algae use the respiratory pathway when sunlight is not available! Here we focus on oxidative reactions in mitochondria, beginning with pyruvate oxidation and continuing to the redox reactions of the Krebs cycle.
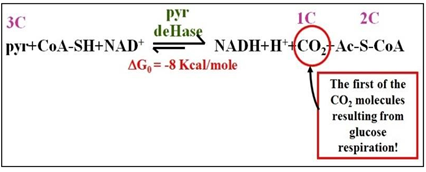
After entering the mitochondria, pyruvate dehydrogenase catalyzes pyruvate oxidation to Acetyl-S-Coenzyme A (Ac-S-CoA). Then the Krebs cycle completely oxidizes the Ac-S- CoA. These mitochondrial redox reactions generate CO2 and lot of reduced electron carriers (NADH, FADH2). The free energy released in these redox reactions is coupled to the synthesis of only one ATP per pyruvate oxidized (i.e., two per the glucose we started with!). It is the NADH and FADH2 molecules have captured most of the free energy in the original glucose molecules. These entry of pyruvate into the mitochondrion and its oxidation are summarized below.
Pyruvate oxidation converts a 3C carbohydrate into acetate, a 2C molecule, releasing a molecule of CO2. In this highly exergonic reaction, CoA-SH forms a high-energy thioester linkage with the acetate in Ac-S-CoA. The oxidation of pyruvic acid results in the reduction of NAD+, production of Ac-S-CoA and a molecule of CO2, as shown below.
The Krebs cycle functions during respiration to oxidize Ac-S-CoA and to reduce NAD+ and FAD to NADH and FADH2 (respectively). Intermediates of the Krebs cycle also function in amino acid metabolism and interconversions. All aerobic organisms alive today share the Krebs cycle we see in humans. This is consistent with its spread early in the evolution of our oxygen environment. Because of the central role of Krebs cycle intermediates in other biochemical pathways, parts of the pathway may even have pre- dated the complete respiratory pathway. The Krebs cycle takes place in mitochondria of eukaryotic cells.
After the oxidation of pyruvate, the Ac-S-CoA enters the Krebs cycle, condensing with oxaloacetate in the cycle to form citrate. There are four redox reactions in the Krebs cycle. As we discuss the Krebs cycle, look for the accumulation of reduced electron carriers (FADH2, NADH) and a small amount of ATP synthesis by substrate-level phosphorylation. Also, follow the carbons in pyruvate into CO2. The Krebs Cycle as it occurs in animals is summarized below.
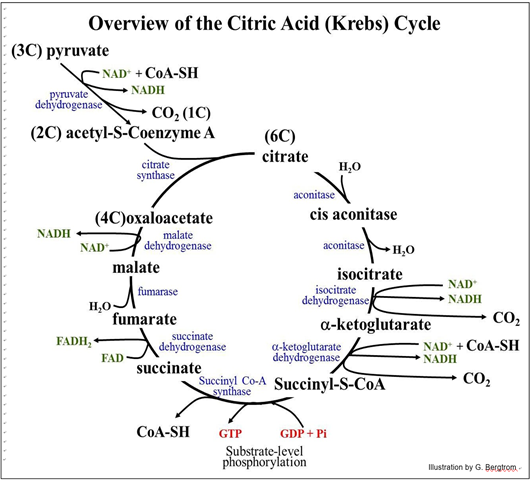
To help you understand the events of the cycle,
1. find the two molecules of CO2 produced in the Krebs cycle itself.
2. find GTP (which quickly transfers its phosphate to ADP to make ATP). Note that in bacteria, ATP is made directly at this step.
3. count all of the reduced electron carriers (NADH, FADH2). Both of these electron carriers carry a pair of electrons. If you include the electrons on each of the NADH molecules made in glycolysis, how many electrons have been removed from glucose during its complete oxidation?
Remember that glycolysis produces two pyruvates per glucose, and thus two molecules of Ac-S-CoA. Thus, the Krebs cycle turns twice for each glucose entering the glycolytic pathway. The high-energy thioester bonds formed in the Krebs cycle fuel ATP synthesis as well as the condensation of oxaloacetate and acetate to form citrate in the first reaction. Each NADH carries about 50 Kcal of the 687 Kcal of free energy originally available in a mole of glucose; each FADH2 carries about 45 Kcal of this free energy. This energy will fuel ATP production during electron transport and oxidative phosphorylation.
159 Highlights of the Krebs Cycle
Finally, the story of the discovery of the Krebs cycle is as interesting as the cycle itself! Albert Szent-Györgyi won a Nobel Prize in 1937 for discovering some organic acid oxidation reactions initially thought to be part of a linear pathway. Hans Krebs did the elegant experiments showing that the reactions were part of a cyclic pathway. He proposed (correctly!) that the cycle would be a supercatalyst that would catalyze the oxidation of yet another organic acid. Some of the experiments are described by Krebs and his coworkers in their classic paper: Krebs HA, et al. [(1938) The formation of citric and α-ketoglutaric acids in the mammalian body. Biochem. J. 32: 113–117]. Hans Krebs and Fritz Lipmann shared the 1953 Nobel Prize in Physiology or Medicine. Krebs was recognized for his elucidation of the TCA cycle, which now more commonly carries his name. Lipmann was recognized for proposing ATP as the mediator between food (nutrient) energy and intracellular work energy, and for discovering the reactions that oxidize pyruvate and synthesize Ac-S-CoA, bridging the Krebs Cycle and oxidative phosphorylation (to be considered iin the next chapter).
160 Discovery of the Krebs Cycle
You can read Krebs’ review of his own research in Krebs HA [(1970) The history of the tricarboxylic acid cycle. Perspect. Biol. Med. 14:154-170]. For a classic read on how Krebs described his supercatalyst suggestion, click Hans Krebs Autobiographical Comments. For more about the life of Lipmann, check out the brief Nobel note on the Fritz Lipmann Biography.


