Bond Types - Ionic and Covalent
- Page ID
- 8155
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Bond Types
In Bis2a we focus primarily on three different bond types: ionic bonds, covalent bonds, and hydrogen bonds. We expect students to be able to recognize each different bond type in molecular models. In addition, for commonly seen bonds in biology, we expect student to provide an chemical explanation, rooted in ideas like electronegativity, for how these bonds contribute to the chemistry of biological molecules.
Ionic Bonds
Ionic bonds are electrostatic interactions formed between ions of opposite charges. For instance, most of us appreciate that sodium chloride (NaCl)positively charged sodium ions and negatively charged chloride ions associate via electrostatic (+ attracts -) interactions to make crystals of sodium chloride, or table salt, creating a crystalline molecule with zero net charge. These origins of these interactions may arise from the association of neutral atoms whose difference in electronegativities is sufficiently high. Take the example above. If we imagine that a neutral sodium atom and a neutral chlorine atom approached one another it is possible that at close distances, due to the relatively large difference in electronegative between the two atoms that an electron from the neutral sodium atom was transferred to the neutral chloride atom, resulting in a negatively charged chloride ion and a positively charged sodium ions. These ions can now interact via an ionic bond.
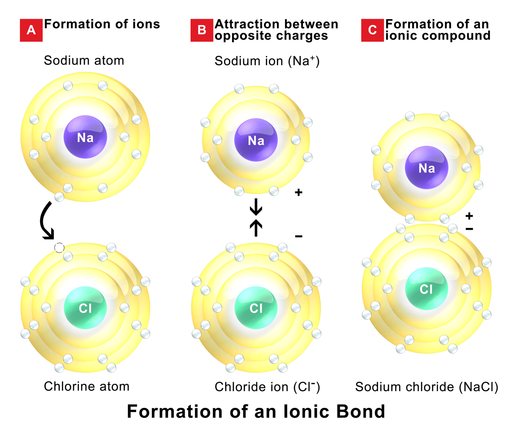
Figure 1: The formation of an ionic bond between sodium and chloride is depicted. In panel A, a sufficient difference in electronegativity between sodium and chloride induce a transfer of electron from the sodium to the chloride forming two ions illustrated in panel b. In panel c the two ions associate via an electrostatic interaction.
Attribution: By BruceBlaus (Own work) [CC BY-SA 4.0 (http://creativecommons.org/licenses/by-sa/4.0)], via Wikimedia Commons
This movement of electrons from one atom to another is referred to as electron transfer. In the example above, when sodium loses an electron, it now has 11 protons, 11 neutrons, and 10 electrons, leaving it with an overall charge of +1 (summing charges: 11 protons at +1 charge each and 10 electrons at -1 charge each = +1). Once charged the the sodium atom is referred to as a sodium ion. Likewise, based on its electronegativity, a neutral chlorine (Cl) atom tends to gain an electron to create an ion with 17 protons, 17 neutrons, and 18 electrons, giving it a net negative (–1) charge. It is now referred to as a chloride ion.
We can interpret the electron transfer above using the concept of electronegativity. Begin by comparing the electronegativities of sodium and chlorine by examining the periodic table of elements below. We see that chlorine is located on the upper right hand corner of the table while sodium is on the upper left. Comparing the electronegativity values of chlorine and sodium directly we see that the chlorine atom is more electronegative than its sodium counterpart. The difference in the electronegativity of chlorine (3.16) and sodium (0.93) is 2.23 (using the scale in the table below). Given that we know an electron transfer will take place between these two element, we can conclude that differences in electronegativities of of ~2.2 are large enough to cause an electron transfer between two atoms and that interactions between such elements are likely through ionic bonds.
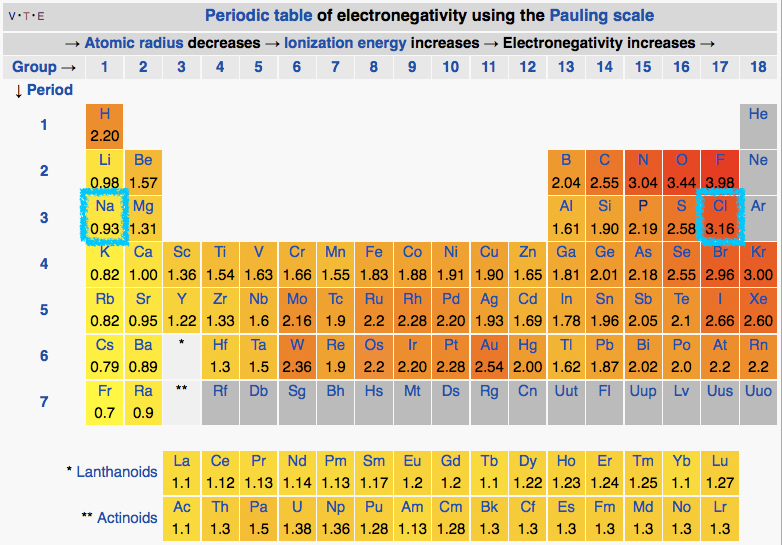
Figure 2: The periodic table of the elements listing electronegativity values for each element. The elements sodium and chlorine are boxed with a teal boundary. Attribution: By DMacks (https://en.wikipedia.org/wiki/Electronegativity) [CC BY-SA 3.0 (http://creativecommons.org/licenses/by-sa/3.0)], via Wikimedia Commons - Modified by Marc T. Facciotti
Note: Possible Discussion
The atoms in a 5 in. x 5 in. brick of table salt (NaCl) sitting on your kitchen counter are held together almost entirely by ionic bonds. Based on that observation how would you characterize the strength of ionic bonds?
Now consider that same brick of table salt after having been thrown into an average backyard swimming pool. After a couple of hours the brick would be completely dissolved and the sodium and chloride ions would be uniformly distributed throughout the pool. What might you conclude about the strength of ionic bonds from this observation?
Propose a reason why the ionic bonds in air might be behaving differently than those in water? What is the significance of this to biology?
For additional information:
Check out the link from the Khan academy on ionic bonds.
Covalent Bonds
We can also invoke the concept of electronegativity to help describe the interactions between atoms of similar electronegativity, differences that are smaller than required to form an ionic bond. These types of associations often result in a bond called a covalent bond. In these bonds, electrons are shared between two atoms - in contrast to an ionic interaction in which electrons remain on each atom of an ion or are transferred between species of highly different electronegativities.
We start by exploring the covalent bond by looking at an example where the difference in electronegativity is zero. Consider a very common interaction in biology, the interaction between two carbon atoms. In this case each atom has the same electronegativity, 2.55; the difference in electronegativity is therefore zero. If we build our mental model of this interaction using the concept of electronegativity we realize that each atom in the carbon carbon pair has the same tendency to "pull" electrons to it. In this case when a bond is formed neither of the two atoms will tend to "pull" (a good anthropomorphism) electrons from the other, they will "share" (another anthropomorphism) the electrons equally, instead.
Aside: Bounding Example
The two examples above (1) the interaction of sodium and chlorine and (2) the interaction between two carbon atoms is an example of framing a discussion by bounding or asymptotic analysis (see earlier reading). We examined what happens to a physical system when considering two extremes. In this case the extremes were in electronegativity differences between interacting atoms. The interaction of sodium and chlorine illustrated what happens when two atoms have a large difference in electronegativities and the carbon carbon example illustrated what happens when that difference is zero. Once we have those mental goal posts describing what happens at the extremes it is then easier to imagine what might happen in between - in this case what happens at differences of electronegativity that are in between 0 and 2.2. We do that next.
When the sharing of electrons between two covalently bonded atoms if nearly equal we call these bonds non-polar covalent bonds. If by contrast, the sharing of electrons is not equal between the two atoms (likely due to a difference in electronegativities between atoms) we call these bonds polar covalent bonds.
In a polar covalent bonds, the electrons are unequally shared by the atoms and are attracted to one nucleus more than to the other. Because of the unequal distribution of electrons between atoms in a polar covalent bond, a slightly positive (indicated δ+) or slightly negative (indicated δ–) charge develops on each pole of the bond. This slightly positive (δ+) charge will develop on the less electronegative atom as electrons get pulled more towards the slightly more electronegative atom. A slight negative charge (indicated δ–) charge will develop on the more electronegative atom. Since there are two poles (the positive and negative poles) the bond is said to possess a dipole.
Examples of Nonpolar Covalent and Polar Covalent Bonds in Biologically Relevant Molecules
Nonpolar covalent bonds
Molecular Oxygen
Molecular oxygen (O2) is an association between two atoms of oxygen. Since the two atoms share the same electronegativity the bonds in molecular oxygen non-polar covalent.
Methane
Another example of a nonpolar covalent bond is the C-H bond found in gas methane (CH4). Unlike the case of molecular oxygen where the two bonded atoms share the same electronegativity, carbon and hydrogen do not have the same electronegativity; C = 2.55 and H = 2.20 - the difference in electronegativity is 0.35.
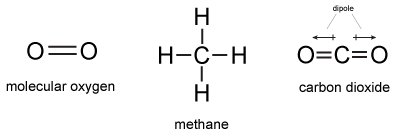
Figure 3: Molecular line drawings of molecular oxygen, methane and carbon dioxide.
Attribution: Marc T. Facciotti (own work)
Some of you may now be confused. If there is a difference in electronegativity between the two atoms is the bond not by definition polar? The answer is both yes and no and depends on the definition of polar that the speaker/writer is using. Since this is an example of how shortcuts in the use of specific vocabulary can sometimes lead to confusion we take a moment to discuss this here. See the mock exchange between student and instructor below for clarification:
1. Instructor: "In biology we often that the C-H bond is nonpolar."
2. Student: "But there is an electronegativity difference between C and H and therefore it would appear that C should have a little bit more tendency to attract electrons. This difference should create a small negative charge around the carbon and a small positive charge around the hydrogen."
3. Student: "Since there is a differential distribution of charge across the bond it would seem that, by definition, this should be considered a polar bond."
4. Instructor: "In fact, the bond does have some small polar character." 5. Student: "So, then it's polar? I'm confused."
6. Instructor: "It has some small amount of polar character but it turns out that for most of the common chemistry that we will encounter that this small amount of polar character is insufficient to lead to "interesting" chemistry. So, the while the bond is strictly slightly polar, from a practical standpoint it is effectively nonpolar. We therefore call it non polar."
7. Student: "That's needlessly confusing, how am I supposed to know when you mean strictly 100% nonpolar, slightly polar, or functionally polar when you use the same word to describe two of those three things?"
8. Instructor: "Yup, it sucks. The fix is that I need to be as clear as I can when I talk with you about "polarity" about how I am using the term. I also need to inform you that you will find this (and other) shortcut used when you go out into the field and encourage you to start learning to recognize what is intended by the context of the conversation.
An real-world analogy of this same problem might be the use of the word "newspaper". It can be used in a sentence to refer to the company that publishes some news OR it can refer to the actual item that the company produces. In this case the disambiguation is easily made by native English speakers who can read meaning from the context; new learners may be more confused. Don't worry, as you see more examples of technical word use in science you'll learn to read the meaning from context too."
A side:
How large should the difference in electronegativity be in order to create a bond that has sufficient polar character that we decide to call it polar in biology? Of course, the exact value depends on a number of factors but as a loose rule of thumb we sometimes use a difference of 0.4 as a guesstimate.
This extra information is purely for your information. You will not be asked to assign polarity based on this criteria in Bis2a. You should however appreciate the concept of how polarity can be determined by using the concept of electronegativity, the functional consequences of polarity (more on this in other sections), and the nuance associated with these terms (the discussion above).
Polar covalent bonds
The polar covalent bond can be illustrated by examining the association between O and H in water (H2O). Oxygen has an electronegativity of 3.44 while hydrogen has an electronegativity of 2.20. The difference of electronegativity is 1.24. It turns out that this size of electronegativity difference is large enough that the dipole across the bond contributes to chemical phenomena that we will be interested in.
This is also a good point to mention another common source of student confusion regarding the use of the term polar. Water has polar bonds. This statement refers specifically to the individual O-H bonds. Each of these bonds has a dipole. However, students will also hear that water is a polar molecule. This is also true. This latter statement is referring to the fact that the sum of the two bond dipoles creates a dipole across the whole molecule. A molecule may be nonpolar but still have some polar bonds.
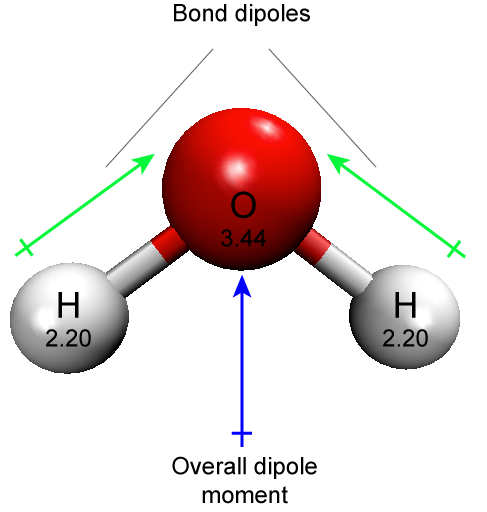
Figure 4: A water molecule has two polar O-H bonds. Since the distribution of charge in the molecule is asymmetric (due to the number and relative orientation of the bond dipoles) the molecule is also polar. The element name and electronegativities are reported in the respective sphere.
Attribution: Marc T. Facciotti (own work)
For additional information: View this short video to see an animation of ionic and covalent bonding.
The continuum of bonds between covalent and ionic
The discussion of bond types above highlights that in nature you will see bonds on a continuum from completely non-polar covalent to purely ionic depending on the atoms that are interacting. As you proceed through your studies you will further discover that in larger, multi-atom molecules, the localization of electrons around an atom is also influenced by multiple factors. For instance, other atoms that are also bonded nearby will exert an influence on the electron distribution around a nucleus in a way that is not easily accounted for by invoking simple arguments of pairwise comparisons of electronegativity. Local electrostatic fields produced by other non-bonded atoms may also have an influence. Recall, reality is always more complicated than our models. However, if the models allow us to reason and predict with "good enough" precision or to understand some key underlying concepts that can be extend later they are quite useful.
Key Bonds in Bis2a
Fortunately, in Bis2A we are largely concerned by the chemical behavior of and bonds between atoms in biomolecules. Since biological systems are composed by a relatively small number of common elements (e.g. C, H, N, O, P, S) and some key ions (e.g. Na+, Cl-, Ca2+, K+, etc.). Start recognizing commonly occurring bonds and the chemical properties that we often see them showing. Some common bonds include C-C, C-O, C-H, N-H, C=O, C-N, P-O, O-H, S-H, and some variants. These will be discussed further in the context of functional groups. The task is not as daunting as it seems.

