8.5: Receptor Tyrosine Kinases (RTKs)
- Page ID
- 3070
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Receptor tyrosine kinases mediate responses to a large number of signals, including peptide hormones like insulin and growth factors like epidermal growth factor. Like the GPCRs, receptor tyrosine kinases bind a signal, then pass the message on through a series of intracellular molecules, the last of which acts on target proteins to change the state of the cell.
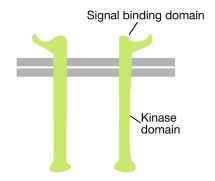
As the name suggests, a receptor tyrosine kinase is a cell surface receptor that also has a tyrosine kinase activity. The signal binding domain of the receptor tyrosine kinase is on the cell surface, while the tyrosine kinase enzymatic activity resides in the cytoplasmic part of the protein (see figure above). A transmembrane alpha helix connects these two regions of the receptor.
What happens when signal molecules bind to receptor tyrosine kinases?
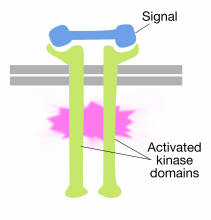
Binding of signal molecules to the extracellular domains of receptor tyrosine kinase molecules causes two receptor molecules to dimerize (come together and associate). This brings the cytoplasmic tails of the receptors close to each other and causes the tyrosine kinase activity of these tails to be turned on. The activated tails then phosphorylate each other on several tyrosine residues. This is called autophosphorylation.
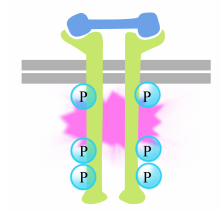
The phosphorylation of tyrosines on the receptor tails triggers the assembly of an intracellular signaling complex on the tails. The newly phosphorylated tyrosines serve as binding sites for signaling proteins that then pass the message on to yet other proteins. An important protein that is subsequently activated by the signaling complexes on the receptor tyrosine kinases is called Ras.
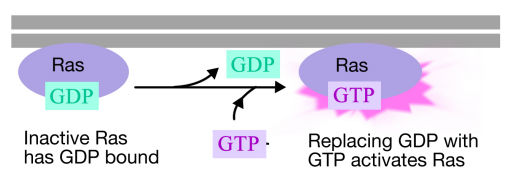
The Ras protein is a monomeric guanine nucleotide binding protein that is associated with the cytosolic face of the plasma membrane (in fact, it is a lot like the alpha subunit of trimeric G-proteins). Just like the alpha subunit of a G- protein, Ras is active when GTP is bound to it and inactive when GDP is bound to it.Also, like the alpha subunit, Ras can hydrolyze the GTP to GDP.
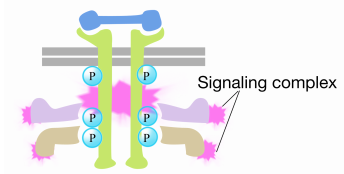
When a signal arrives at the receptor tyrosine kinase, the receptor monomers come together and phosphorylate each others' tyrosines, triggering the assembly of a complex of proteins on the cytoplasmic tail of the receptor. One of the proteins in this complex interacts with Ras and stimulates the exchange of the GDP bound to the inactive Ras for a GTP. This activates the Ras.
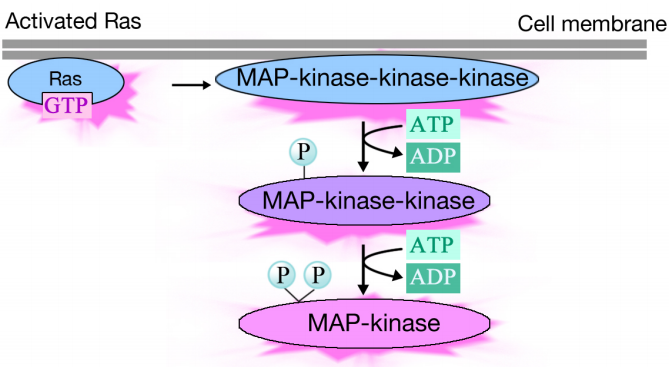
Activated Ras triggers a phosphorylation cascade of three protein kinases, which relay and distribute the signal. These protein kinases are members of a group called the MAP kinases (Mitogen Activated Protein Kinases). The final kinase in this cascade phosphorylates various target proteins, including enzymes and transcriptional activators that regulate gene expression.
The phosphorylation of various enzymes can alter their activities, and set off new chemical reactions in the cell, while the phosphorylation of transcriptional activators can change which genes are expressed. The combined effect of changes in gene expression and protein activity alter the cell's physiological state.
Once again, in following the path of signal transduction mediated by RTKs, it is possible to discern the same basic pattern of events: a signal is bound by the extracellular domains of receptor tyrosine kinases, resulting in receptor dimerization and autophosphorylation of the cytosolic tails, thus conveying the message to the interior of the cell.
The message is passed on via a signalling complex to Ras which then stimulates a series of kinases. The terminal kinase in the cascade acts on target proteins and brings about in changes in protein activities and gene expression.
The descriptions above provide a very simple sketch of some of the major classes of receptors and deal primarily with the mechanistic details of the steps by which signals received by various types of receptors bring about changes in cells. A major take-home lesson is the essential similarity of the different pathways.
Another point to keep in mind is that while we have looked at each individual pathway in isolation, a cell, at any given time receives multiple signals that set off a variety of different responses at once. The pathways described above show a considerable degree of "cross-talk" and the response to any given signal is affected by the other signals that the cell receives simultaneously. The multitude of different receptors, signals and the combinations thereof are the means by which cells are able to respond to an enormous variety of different circumstances.


