Homework Problems - Literature Learning Module: Transpeptidase Inhibition - KEY
- Page ID
- 9981
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Research Paper: A Novel, Species-specific Class of Uncompetitive Inhibitors of gamma-Glutamyl Transpeptidase. Jarrod B. King, Matthew B. West , Paul F. Cook , and Marie H. Hanigan. J. Biol. Chem., 284, NO. 14, pp. 9059–9065, April 3, 2009.
From Abstract: Expression of gamma -glutamyl transpeptidase (GGT) in tumors contributes to resistance to radiation and chemotherapy. GGT is inhibited by glutamine analogues that compete with the sub- strate for the gamma -glutamyl binding site. However, the glutamine analogues that have been evaluated in clinical trials are too toxic for use in humans. We have used high throughput screening to evaluate small molecules for their ability to inhibit GGT and have identified a novel class of inhibitors that are not glutamine analogues.
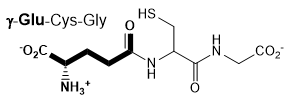
The enzyme γ-glutamyl transpeptidase (GGT), which is localized to the cell surface, cleaves the γ-glutamyl amide bond of extracellular glutathione (right), and transfers the γ-glutamyl group to an acceptor substrate.
a. A compound, OU749 was discovered that inhibited the enzyme. Shown are the structure of OU749 (left) and substrate velocity curves (right) for the inhibition of human kidney GGT by 0 μm (▪), and various concentration of OU749 (15.2 μm (next curve down), then 31.3 μm, 62.5 μm, and 125 μm) in the presence of 40 mm glygly. From inspection of the graph, do you think the inhibition is competitive? Explain?
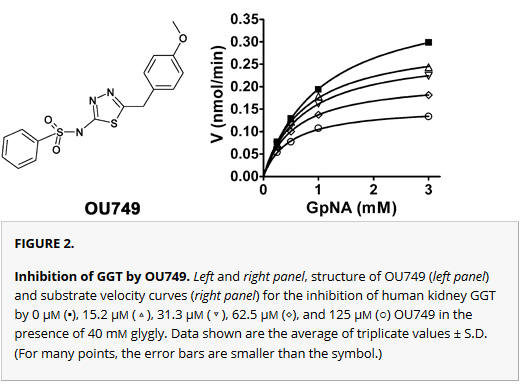
It can't be competitive since the apparent Vm clearly decreases with increasing inhibitor. By inspection it appears that the Km might also be decreasing as well, so it could be uncompetitive. Double-reciprocal plots would make it easier to see. However, for a definitive answer, the data should be subjected to a nonlinear fit to the mathematical equations for COMP, UNCOMP, and MIXED inhibition.
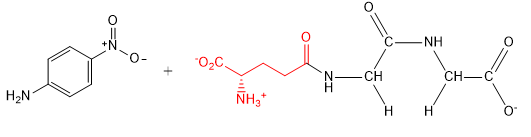
b. For the reaction of L-glutamic acid γ-4-nitroanilide HCl (GpNA) and 40 mm glycyl-glycine (glygly) (acceptor), draw the products of the reaction.

c. What wavelength would you choose to monitor the course of the reaction? ___405 nm____ nm
This is the optimal wavelength for absorbance of p-nitroanilide which produces a yellow color in solution.
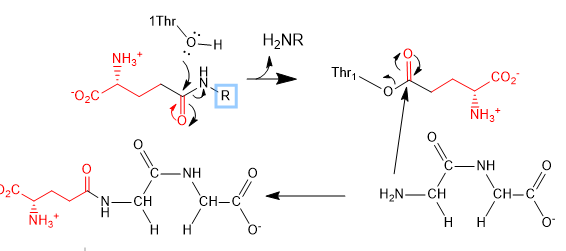
d. The mechanism seems to involve a covalent intermediate of the substrate with the enzyme through an N terminal Thr. This threonine is conserved in human, rat, mouse, and pig. Draw a mechanism showing the initial reaction of glutathione (abbreviated structure shown below) with the N terminal Thr and then in the subsequent reaction with the acceptor Gly-Gly (represented in your diagram as R’NH2)

e. Site-directed mutagenesis of human GGT has identified four additional amino acids that are essential to GGT activity Arg 107, Asp-423, Ser-451, and Ser-452 . All four of these amino acids are identical in human, rat, mouse, and pig.

- Which amino acid might likely be found at (a)? __Asp or Glu (- charged)___
- Which amino acid might likely be found at (b)? __Arg or Lys (+ charged)___
- What type of catalytic mechanism best describes the role of Thr 1 ? __nucleophilic catalysis, covalent catalysis or group transfer___
- What type of catalytic mechanism best describes the role of Ser 451 and 452? stabilize - charge in oxyanion of transition state formed from carbonyl
f. Complete the Cleland diagram below to show the likely kinetic mechanism based on your answers from above. A = GPNA, B = Gly-Gly. What are P and Q?
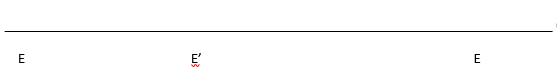
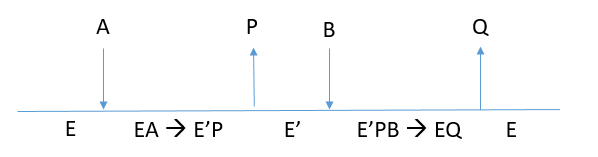
P is the amine leaving group (p-nitroanline) and Q is Gly-Gly that has been connected to glu through the gamma carboxyl group
g. Kinetic analyses of GGT inhibition by OU749 are shown in the graphs to the right. Shown are double reciprocal plots of the initial velocities of human kidney GGT at varying GpNA (A) and Gly-Gly (B) in the presence of 40 mm glygly (left) or 3 mm GpNA (right) with the following different fixed amounts of inhibitor OU749: 0 μm (▪), 15.2 μm (▴), 31.3 μm (▵), 62.5 μm (⋄), and 125 μm (○). What types of inhibition are show in Graph A? Graph B?
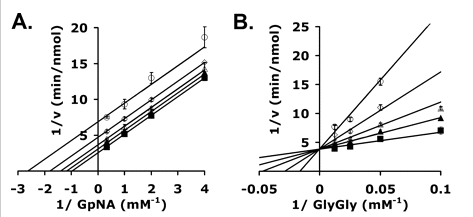
Kinetic Analysis of GGT inhibition by OU749. A–D, double reciprocal plots of the initial velocities of human kidney GGT (A and B) in the presence of 40 mm glygly (A)) or 3 mm GpNA (B) with 0 μm (▪), 15.2 μm (▴), 31.3 μm (▵), 62.5 μm (⋄), and 125 μm (○) OU749 (curves move upward with increasing inhibitor). Data shown are average of triplicate values ± S.D. (For many points, the error bars are smaller than the symbol.)
h. In multi--substrate reaction, the following effects of dead-end inhibitors (I) are observed
-
Slope changes when I and S bind to the same form of the enzyme OR I binds to a form of E (on the horizontal line) which is connected to the form that S binds, and I binds first (for example, I binds to E and then S binds).
-
Y Intercept changes when: I and S bind to different forms of the enzyme unless I binds first and the binding of I and S are in rapid equilibrium
Complete the following with the word same or different based on your previous answers and knowledge. OU749 binds to a (the) different form of enzyme as GpNA.
OU749 binds to a (the) ___same_________ form of enzyme as GlyGly
i. Under what conditions do uncompetitive inhibitors lead to more inhibition in vivo than competitive inhibition? (one phrase).
In vivo under the steady state when a huge increase in substrate is required to maintain the steady state in presence of an uncompetitive inhibitor.
j. The plain old amino acid serine was added to the enzyme. What type of inhibitor might it be? _competitive_______ Show this by positioning Ser into the active site cartoon cartoon diagram below.
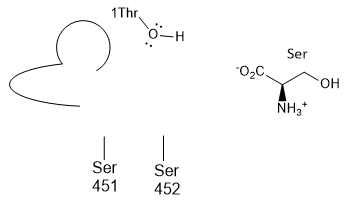
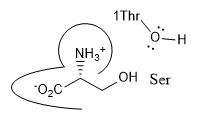
k. A Ser-borate complex (right) is a much more effective inhibitor. From your knowledge of how enzymes catalyze chemical reactions, explain why in a single phrase.
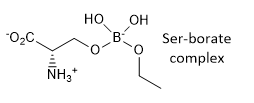
The Ser-borate complex is similar in structure to the transition state so it binds more tightly than the substrate. It is a transition state analog.
k. Site specific mutagenesis was performed to see how changing specific serines (S) would affect activity. (WT = wild-type, unmutated control). The following mutants, denoted as S###X where X is the new amino acid (blacked out in the table below) were made. Which amino acid would the investigators choose to replace Ser in the mutants? __Ala_to maintain side chain size_____
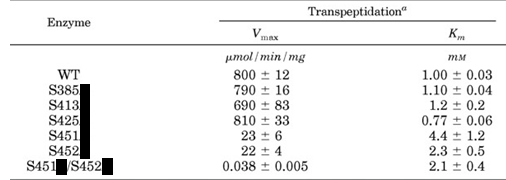
m. Only one kinetic parameter is significantly changed (think of the % changes on the parameters). What step in catalysis is affected in the mutants?
Vm which dramatically decreases suggesting that kcat, the rate at which bound substrate is converted to product, is affected.


