16.8: The Immune System
- Page ID
- 19398
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Immunology is a full semester course at most universities, so this section will only touch on a few basic concepts that should be easily accessible to the student who has nearly completed the cell biology course. At its core, immunology is about adaptation. That is, since an animal has no preconception of the various potential infections it may be subject to, it must have a system in place that is flexible enough to deal with almost anything that comes along. Obviously, the systems are not perfect, but considering the wide range of pathogens, immune systems are remarkably efficient. In humans, there are two types of immune response to infection: the innate response, which is relatively nonspecific, and the adaptive, or acquired, response, which has more specificity.
The innate immune responses are common to all animals, and act on large classes of pathogens. For example, Toll-like receptors on phagocytes recognize a variety of bacterial surface molecules such as the flagellin specific to bacterial flagella, or the peptidoglycan components of bacterial cell walls. When these receptors are activated, the phagocyte goes into action, enveloping the offending bacteria or virus, and breaking it down. This depends on recognizing the external surface, so bacteria or viruses that do not have a recognizable molecule on their surface are able to escape this particular line of defense.
Defensins, which are found on a variety of surfaces (skin, cornea, gut) as well as in circulation, are small (18-45 amino acids) cysteine-rich cationic proteins that bind to a variety of pathogenic viruses, bacteria, and fungi. It is unclear how they may work against viruses, other than perhaps attacking infected host cells, but against bacteria and fungi the mode of operation is generally to bind to the cell membrane and form a pore that allows ions and other small molecules to ow out killing the pathogen. Complement, a group of proteins (~20) circulating in the blood, can act similarly against pathogenic cells.
Finally, natural killer (NK) cells, lymphocytes that target any cell that does not carry cell surface proteins that are normally found on cells from the animal, can kill not only attacking cells, but virally infected cells that have stopped producing their normal proteins (including the recognition protein) because they are busy producing viral proteins. NK cells can even be effective against some cancer cells if they have downregulated cell surface protein expression as part of their de-differentiation and de-adhesion.
The adaptive immune system, which is only found in vertebrates, is what most people think of when the human immune system is mentioned. We and other vertebrates also have an innate immune system, but all the molecules and cells that normally come to mind — antibodies, T-cells, B-cells — are part of the adaptive immune response. There are two components to the adaptive response, a humoral response, in which proteins (antibodies) floating in the blood bind to the infectious agent and prevent if from binding to cells or targeting it for the cellular response, which is mediated by T cells that can specifically recognize and kill the targeted pathogen.
Antibodies
Front and center in the adaptive immune response are antibodies. These proteins may be either secreted by or attached to the surface of B cells, the lymphocytes that differentiate either in bone marrow (adult) or liver (fetus), as opposed to those called T cells, which differentiate in the thymus gland. Incidentally, if you see sweetbreads on a menu, that would be thymus. Yum. You can take that seriously or sarcastically depending on how you think my tastes run.
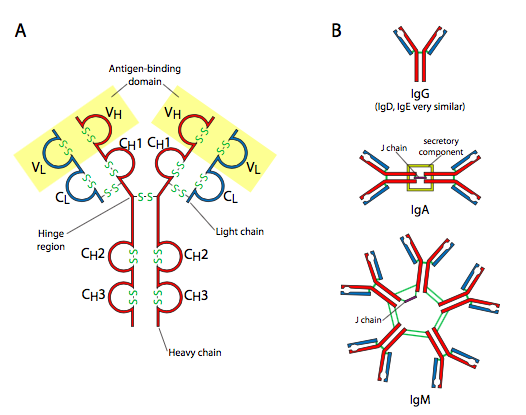
Back to the antibodies. The different types of antibodies, IgA, IgD, IgE, IgG, and IgM, are all based on the IgG structure (Figure \(\PageIndex{18}\)), which is roughly Y-shaped, and composed of two heavy chains and two light chains. These chains have disulfide bond-stabilized loops (recall the Ig-like loops in the cell adhesion molecules a few chapters back?), and the combination of the distal light chain loop and distal heavy chain loop make the antigen binding site. The antigen is defined as the molecule, or more specifically the part of a molecule that is recognized by the particular antibody. Since the antibody is meant to mediate highly specific recognition of a wide variety of invading pathogens, there must be a way to create at least as many different antibodies. This is made possible by the process of DNA rearrangement. This mechanism is also used to generate diversity in T-cell receptors, which are quite different structurally, but also need to be available with an extremely wide variety of specific binding sites.