29.01A: Overview of Metabolism
- Page ID
- 53873
An Overview of Metabolic Pathways - Catabolism
Biological cells have a daunting task. They must carry out 1000s of different chemical reactions required to carry out cell function. These reactions can include opposing goals such as energy production and energy storage, macromolecule degradation and synthesis, and breakdown and synthesis of small molecules. All of these reactions are catalyzed by proteins and RNAs enzymes whose activities must be regulated, again through chemical reactions, to avoid a futile and energy wasting scenario of having opposing pathways functioning simultaneously in a cell.
Metabolism can be divided into two main parts, catabolism, the degradation of molecules, usually to produce energy or small molecules useful for cell function, and anabolism, the synthesis of larger biomolecules from small precursors.
CATBOLISM: Catabolic reactions involve the breakdown of carbohydrates, lipids, proteins, and nucleic acids to produce smaller molecules and biological energy in the form of heat or small thermodynamically reactive molecules like ATP whose further degradation can drive endergonic process such as biosynthesis. Our whole world is reliant on the oxidation of organic hydrocarbons to water and carbon dioxide to produce energy (at the expense of releasing a potent greenhouse gas, CO2). In the biological world, reduced molecules like fatty acids and partially oxidized molecules such as glucose polymers (glycogen, starch), as well as simple sugars, can be partially or fully oxidized to ultimately produce CO2 as well. Energy released from oxidative reactions is used to produce molecules like ATP as well as heat. Oxidative pathways include glycolysis, the tricarboxylic acid cycle (aka Kreb's cycle) and mitochondrial oxidative phosphorylation/electron transport. To fully oxidize carbon in glucose and fatty acids to carbon dioxide requires splitting C-C bonds and the availability of series of oxidizing agents that can perform controlled, step-wise oxidation reactions, analogous to the sequential oxidation of methane, CH4 to methanol (CH3OH), formaldehyde (CH2O) and carbon dixoxide.
- Glycolysis: This most primitive of metabolic pathways is found in perhaps all organisms. In glycolysis, glucose (C6H12O6), a 6C molecule, is split (or lysed) into two, 3C carbon molecules, glyceraldehyde-3-phosphate, which are then partially oxidized under anaerobic conditions (without O2) to form two molecules of pyruvate (CH3COCO2-). Instead of the very strong oxidizing agent, O2, a weaker one, NAD+ is used, which is reduced in the process to form NADH. Since none of the carbon atoms is oxidized to the state of CO2, little energy is released compared to the complete oxidation to CO2. This pathway comes to a screeching halt if all cellular NAD+ is converted to NADH as NAD+ is not replenished by the simple act of breathing as is the case with O2 in aerobic oxidation. To prevent the depletion of NAD+ from inhibiting the cycle and to allow the cycle to continue under anaerobic conditions, excess NADH is reconverted to NAD+ when the other product of glycolysis, pyruvate is converted to lactate by the enzyme lactate dehydrogenase. Glycolysis occurs in the cytoplasm of the cell.

- Tricarboxylic Acid (Kreb's) Cycle: The TCA cycle is an aerobic pathway which takes place in an intracellular organelle called the mitochondria. It takes pyruvate, the incompletely oxidized product from glycolysis, and finishes the job of oxidizing the 3C atoms all the way to CO2. First the pyruvate moves into the mitochondria where is is oxidized to the 2C molecule acetylCoA with the release of one CO2 by the enzyme pyruvate dehydrogenase. The acetyl-CoA then enters the TCA cycle where two more CO2 are released. As in glycolysis, C-C bonds are cleaved and C is oxidized by NAD+ and another related oxidizing agent, FAD. What is very different about this pathway is that instead of being a series of linear, sequential reactions with one reactant (glucose) and one product (two pryuvates), it is a cyclic pathway. This has significant consequences since if any of the reactants within the pathways becomes depleted, the whole cyclic pathway can slow down and stop. To see how this happens consider the molecule oxaloacetate (OAA) which condenses with acetyl-CoA to form citrate (see diagram below). In this reaction, one OAA is consumed. However, when the cycle returns, one malate is converted to OAA so there is no net loss of OAA, unless OAA is pulled out of the TCA cycle for other reactions, which happens.
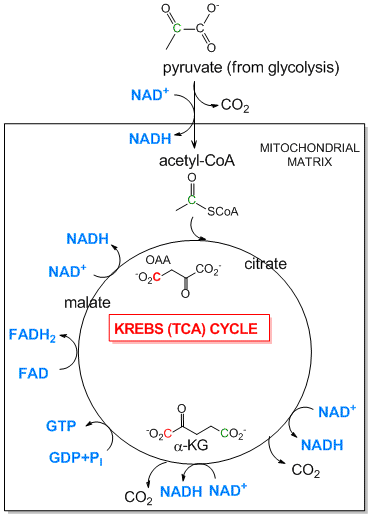
- Mitochondrial Oxidative Phosphorylation/Electron Transport: The TCA cycle accomplishes what glycolysis didn't, that is the cleavage of all C-C bonds in glucose (in the form of pyruvate and acetyl-CoA, and the complete oxidation of all C atoms to CO2. Yet two problem remains. The pool of oxidizing molecules, NAD+ and FAD get converted to their reduced forms, NADH and FADH2. Unless NAD+ and FAD are regenerated, as was the case in anaerobic conditions when pyruvate gets converted to lacate, the pathway would again come to a grinding halt. In addition, not much ATP is made in the cycle (in the form of a related molecule GTP). Both these problems are resolved as the resulting NADH and FADH2 formed are reoxidized by mitochondrial membrane enzyme complexes which pass electrons from the oxidized NADH and FADH2 to increasingly potent oxidizing agents until they are accepted by the powerful oxidant O2,which is converted reduced to water. The net oxidation of NADH and FADH2 by dioxygen is greatly exergonic, and the energy released by the process drives the synthesis of ATP from ADP and Pi by an mitochondrial enzyme complex, the F0F1ATPase.

Feeder Pathways: Other catabolic pathways produce products that can enter glycolysis or the TCA cycle. Two examples are given below.
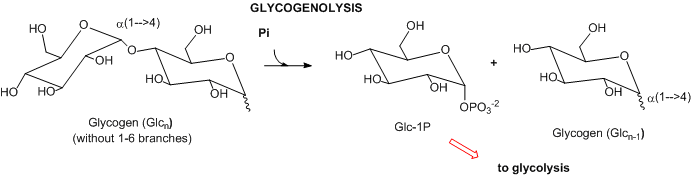
- Complex carbohydrates: In mammals, the major carbohydrate storage molecule is glycogen, a polymer of glucose linked a1-4 with a1-6 branches. The terminal acetal linkages in this highly branched polymer is cleaved sequentially at the ends not through hydrolysis but through phosphorolysis to produce lots of glucose-1-phosphate which can enter glycolysis.
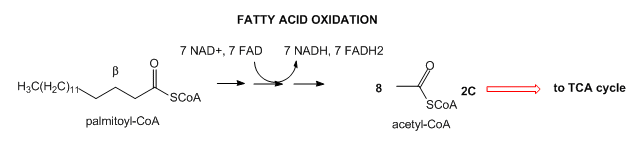
- Lipids: Lipids are stored mostly as triacylglycerides in fat cells (adipocytes). When needed for energy, fatty acids are hydrolyzed from the glycerol backbone of the triacylglyceride, and send into cells where they broken down in an oxidative process to form acetyl-CoA with the concomitant production of lots of NADH and FADH2. These can then enter the mitochondrial oxidative phosphorylation/electrons transport system, which produces, under aerobic conditions, lots of ATP.
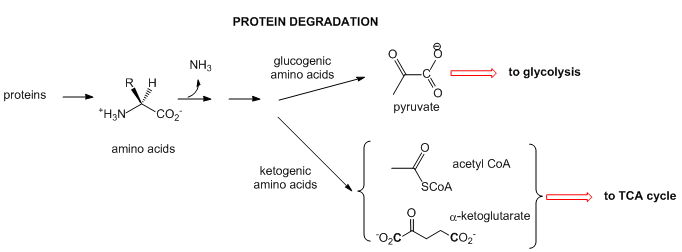
- Proteins: When intracellular proteins get degraded, they from individual amino acids. The amine N is lost as it enters the urea cycle. The rest of some amino acid structures can be ultimately converted to acetyl-CoA or keto acids (like alpha-ketoglutarate- a-KG) that are TCA intermediate. These amino acids are called ketogenic. Alternatively, some amino acids, after deamination, are coveted to pyruvate which can either enter the TCA cycle or in the liver be used to synthesize glucose in an anabolic process. These amino acids are called glucogenic. Chemical reactions such as these can be used to replenish intermediates in the TCA cycle which can become depleted as they are withdraw for other reactions.
Anabolic Reactions
Anabolic reactions are those that lead to the synthesis of biomolecules. In contrast to the catabolic reactions just discussed (glycolysis, TCA cycle and electron transport/oxidative phosphorylation) which lead to the oxidative degradation of carbohydrates and fatty acids and energy release, anabolic reactions lead to the synthesis of more complex biomolecules including biopolymers (glycogen, proteins, nucleic acids) and complex lipids. Many biosynthetic reactions, including those for fatty acid synthesis, are reductive and hence require reducing agents. Reductive biosynthesis and complex polymer formation require energy input, usually in the form of ATP whose exergonic cleavage is coupled to endergonic biosynthesis.
Cells have evolved interesting mechanism so as not to have oxidative degradation reactions (which release energy) proceed at the same time and in the same cell as reductive biosynthesis (which requires energy input). Consider this scenario. You dive into a liver cell and find palmitic acid, a 16C fatty acid. From where did it come? Was it just synthesized by the liver cell or did it just enter the cell from a distant location such as adipocytes (fat cells). Should it be oxidized, which should happen if there is a demand for energy production by the cell, or should the liver cell export it, perhaps to adipocytes, which might happen if there is an excess of energy storage molecules? Cells have devised many ways to distinguish these opposing needs. One is by using a slightly different pool of redox reagents for anabolic and catabolic reactions. Oxidative degradation reactions typically use the redox pair NAD+/NADH (or FAD/FADH2) while reductive biosynthesis often uses phosphorylated variants of NAD+, NADP+/NADPH. In addition, cells often carry out competing reactions in different cellular compartments. Fatty acid oxidation of our example molecule (palmitic acid) occurs in the mitochondrial matrix, while reductive fatty acid synthesis occurs in the cytoplasm of the cell. Fatty acids entering the cell destined for oxidative degradation are transported into the mitochondria by the carnitine transport system. This transport system is inhibited under conditions when fatty acid synthesis is favored. We will discuss the regulation of metabolic pathways in a subsequent section. One of the main methods, as we will see, is to activate or inhibit key enzymes in the pathways under a given set of cellular conditions. The key enzyme in fatty acid synthesis, acetyl-CoA carboxylase, is inhibited when cellular conditions require fatty acid oxidation.
The following examples give short descriptions of anabolic pathways. Compare them to the catabolic pathways from the previous section.
- Glucose synthesis, better known as Gluconeogenesis: In glycolysis, glucose (C6H12O6), a 6C molecule, is converted to two, 3C molecules (pyruvate) in an oxidative process that requires NAD+ and makes two net ATP molecules. In a few organs, most predominately in the liver, the reverse pathway can take place. The liver does this to provide glucose to the brain when the body is deficient in circulating glucose, for example, under fasting and starving conditions. (The liver under these conditions can get its energy from oxidation of fatty acids). The reactions in gluconeogenesis are the same reactions in glycolysis but run in reverse, with the exception of three glycolytic steps which are essentially irreversible. These three steps have bypass enzymes in the gluconeogenesis pathway. Although the synthesis of glucose is a reductive pathway, it uses NADH instead of NADPH as the redundant as the same enzyme used in glycolysis is simply run in reverse. Gluconeogenesis, which also occurs in the cortex of the kidney, is more than just a simple reversal of glycolysis, however. It can be thought of as the net synthesis of glucose from non-carbohydrate precursors. Pyruvate, as seen in the section on catabolism, can be formed from protein degradation to glucogenic amino acids which can be converted to pyruvate. It can also be formed from triacylglycerides from the 3C molecule glycerol formed and released from adipocytes after hydrolysis of three fatty acids from triacylglycerides. However, in humans, glucose can not be made in net fashion from fatty acids. Fatty acids can be converted to acetyl-CoA by fatty acid oxidation. The resulting acetyl-CoA can not form pyruvate since the enzyme that catalyzes the formation for acetyl-CoA from pyruvate, pyruvate dehydrogenase, is irreversible and there is no bypass reaction known. The acetyl-CoA can enter the TCA cycle but since the pathway is cyclic and proceeds in one direction, it can not form in net fashion oxaloacetate. Although oxaloacetate can be remove from the TCA cycle and be use to form phosphoenolpyuvate, a glycolytic intermediate, one acetyl-CoA condenses with one oxaloacetate to form citrate which leads back to one oxaloacetate. Hence fatty acids can not be converted to glucose and other sugars in a net fashion.
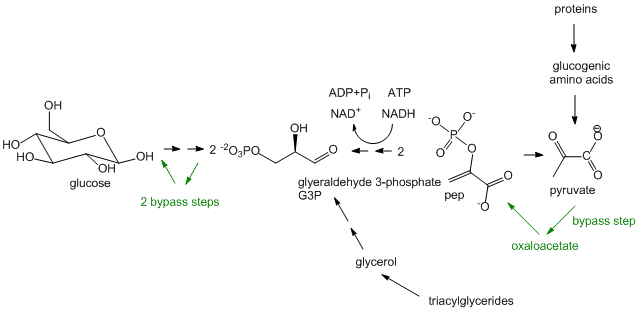
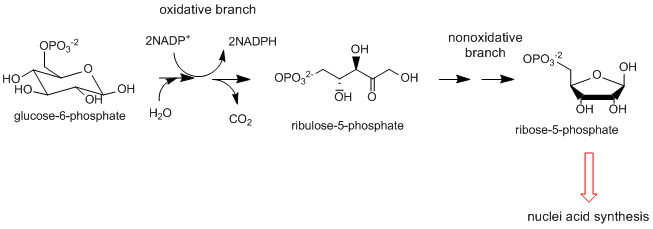
- Pentose Phosphate Shunt: This two-part pathway doesn't appear to start as a reductive biosynthetic pathway as the first part is the oxidative conversion of a glycolytic intermediate, glucose-6-phosphate, to ribulose-5-phosphate. The next, nonoxidative branch leads to the formation of ribose-5-phosphate, a key biosynthetic intermediate in nucleic acid synthesis as well as erthyrose-4-phosphate used for biosynthesis of aromatic amino acids . The oxidative branch is important in reductive biosynthesis as it is a major source of the reductant NADPH used in biosynthetic reactions.
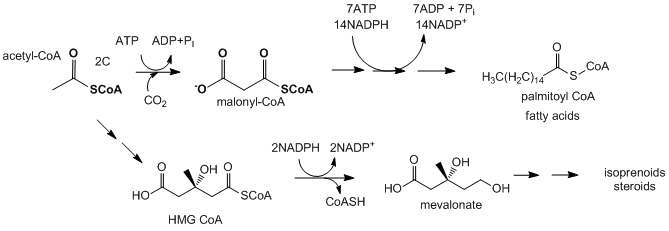
- Fatty acid and isoprenoid/sterol biosynthesis: Acetyl-CoA is the source of carbon atoms for the synthesis of more complex lipids such as fatty acids, isoprenoids, and sterols. When energy needs in a cell are not high, citrate, the condensation product of oxaloacetate and acetyl-CoA in the TCA cycle, builds up in the mitochondrial matrix. It is then transported by the citrate transporter (an inner mitochondrial membrane protein) to the cytoplasm, where it is cleaved back to oxaloacetate and acetyl-CoA by the cytoplasmic enzyme citrate lyase. The oxaloacetate is returned to the mitochondria by conversion first to malate (reduction reaction using NADH), which can move back into the mitochondria through the malate transporter, or further conversion to pyruate, using the cytosolic malic enzyme, which uses NADP+ to oxidize malate to pyruvate which then enters the mitochondria. The acetyl-CoA formed in the cytoplasm can then be used in reductive biosynthesis using NADPH as the reductant to form fatty acids, isoprenoids, and sterols. The NADPH for the reduction comes from the oxidative branch of the pentose phosphate pathway and from the reaction catalyzed by malic enzyme. The liver cells can still run the glycolytic pathway as the NADH/NAD+ ratio is low in the cytoplasm while NADPH/NADP+ ratio is high.
Regulation and Integration
None of these pathways exist in isolation. They are connected in a complicated web of interactions. Instead of cursory summaries of how these pathways are all interconnected, we have chosen to present a series of published articles that are freely available for reuse through Creative Common's licenses. They describe organ- and systems-specific metabolisms and emerging understanding of metabolism in disease states. These include:
- Regulation of glucose metabolism from a liver-centric perspective
- Emerging role of the brain in the homeostatic regulation of energy and glucose metabolism
- Skeletal Muscle Regulates Metabolism
- Intestinal Fructose and Glucose Metabolism in Health and Disease
- Metabolic consequences of obesity and type 2 diabetes: Balancing genes and environment for personalized care
- Lipid-Induced Mechanisms of Metabolic Syndrome
- Fundamentals of cancer metabolism
- Immunometabolism: Cellular Metabolism Turns Immune Regulator
- Ethanol metabolism: The good, the bad, and the ugly
- Gut microbiota-derived metabolites as central regulators in metabolic disorders



