5.6: The Ability to Evade Adaptive Immune Defenses
- Page ID
- 3193
- State four ways the antibody molecules made during adaptive immunity protect us against bacteria.
- Briefly describe at least three ways a bacterium might evade our adaptive immune defenses and name a bacterium that does each.
Overview of Adaptive Immune Defenses
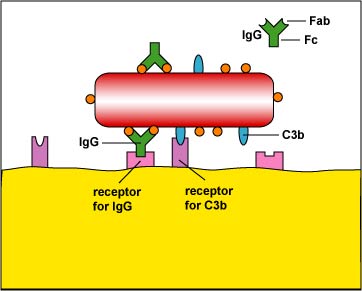
One of the major defenses against bacteria is the immune defenses' production of antibody molecules against the organism. The "tips" of the antibody, called the Fab portion (Figure \(\PageIndex{1}\)) have shapes that are complementary to portions of bacterial proteins and polysaccharides called epitopes. The "bottom" of the antibody, called the Fc portion (Figure \(\PageIndex{1}\)) binds to receptors on phagocytes and NK cells) and can activate the classical complement pathway.

There are various ways that the antibodies the body makes during adaptive immunity protect the body against bacteria:
a. As mentioned above under phagocytosis, some antibodies such as IgG and IgE function as opsonins and stick bacteria to phagocytes (Figure \(\PageIndex{2}\)).
b. Antibodies, such as IgG, IgA, and IgM, can bind to bacterial adhesins, pili, and capsules and in this way block their attachment to host cells.
c. IgG and IgM can also activate the classical complement pathway providing all of its associated benefits.
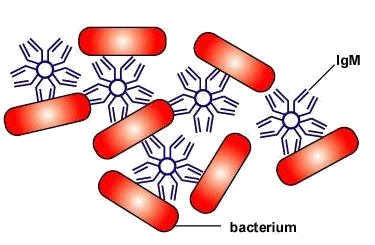
d. IgA and IgM can clump bacteria together enabling them to be more readily removed by phagocytes (Figure \(\PageIndex{3}\)).
These mechanisms will be discussed in greater detail in Unit 6.
- Staphylococcus aureus produces protein A, a protein that binds to the Fc portion of antibodies.
How might this enable S. aureus to resist adaptive immunity?
- Many bacteria that colonize the mucous membranes produce immunoglobulin protease, an enzyme that hydrolizes antibodies of the IgA class.
How might this enable these bacteria to resist adaptive immunity?
Resisting Adaptive Immune Defenses
Bacteria utilize a variety of mechanisms to resist antibodies made during adaptive immunity. These include the following:
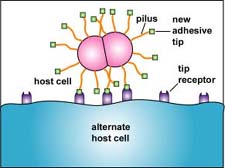
a. Certain bacteria can evade antibodies is by changing the adhesive tips of their pili as mentioned above with Escherichia coli and Neisseria gonorrhoeae (Figure \(\PageIndex{4}\)).
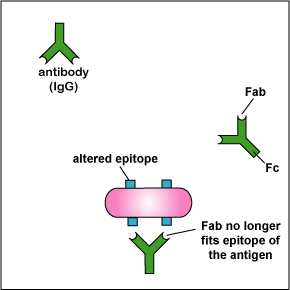
Bacteria can also vary other surface proteins so that antibodies previously made against those proteins will no longer "fit." (Figure \(\PageIndex{5}\)). For example, N. gonorrhoeae produces Rmp protein (protein III) that protects against antibody attack by antibodies made against other surface proteins (such as adhesins) and the lipooligosaccharide (LOS) of the bacterium.
b. Strains of Neisseria meningitidis have a capsule composed of sialic acid while strains of Streptococcus pyogenes (group A beta streptococci) have a capsule made of hyaluronic acid. Both of these polysaccharides closely resemble carbohydrates found in human tissue and because they are not recognized as foreign by the lymphocytes that carry out the adaptive immune responses, antibodies are not made against those capsules. Likewise, some bacteria are able to coat themselves with host proteins such as fibronectin, lactoferrin, or transferrin and in this way avoid having antibodies being made against them because they are unable to be recognized as foreign by lymphocytes.
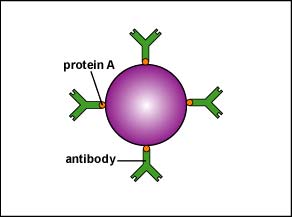
c. Staphylococcus aureus produces protein A while Streptococcus pyogenes produces protein G. Both of these proteins bind to the Fc portion of the antibody IgG, the portion that is supposed to bind the bacterium to phagocytes during enhanced attachment (Figure \(\PageIndex{1}\)). The bacteria become coated with antibodies in a way that does not result in opsonization (Figure \(\PageIndex{6}\)).
d. Salmonella species can undergo phase variation of their capsular (K) and flagellar (H) antigens, that is, they can change the molecular shape of their capsular and flagellar antigens so that antibodies made against the previous form no longer fit the new form (Figure \(\PageIndex{5}\)).
e. Bacteria such as Haemophilus influenzae, Streptococcus pneumoniae, Helicobacter pylori, Shigella flexneri, Neisseria meningitidis, Neisseria gonorrhoeae and enteropathogenic E. coli produce immunoglobulin proteases. Immunoglobulin proteases degrade the body's protective antibodies (immunoglobulins) that are found in body secretions, a class of antibodies known as IgA.
f. Many pathogenic bacteria, as well as normal flora, form complex bacterial communities as biofilms. Bacteria in biofilms are often able to communicate with one another by a process called quorum sensing (discussed later in this unit) and are able to interact with and adapt to their environment as a population of bacteria rather than as individual bacteria. By living as a community of bacteria as a biofilm, these bacteria are better able to resist attack by antibiotics and are better able to resist the host immune system.
Summary
- There are various ways that the antibodies the body makes during adaptive immunity protect the body against bacteria.
- Some antibodies such as IgG and IgE function as opsonins and stick bacteria to phagocytes (opsonization or enhanced attachment).
- Antibodies, such as IgG, IgA, and IgM, can bind to bacterial adhesins, pili, and capsules and in this way block their attachment to host cells.
- IgG and IgM can activate the classical complement pathway providing all of its associated benefits.
- IgA and IgM can clump bacteria together enabling them to be more readily removed by phagocytes.
- Antitoxin antibodies, mainly IgG, are made against bacterial exotoxins. They combine with the exotoxin molecules before they can interact with host target cells and thus neutralize the toxin.
- Bacteria utilize a variety of mechanisms to resist antibodies made during adaptive immunity.
- Some bacteria can vary their surface proteins or polysaccharides so that antibodies previously made against those proteins will no longer "fit."
- Some bacteria are able to coat themselves with host proteins and in this way avoid having antibodies being made against them because they are unable to be recognized as foreign
- Some bacteria produce immunoglobulin proteases that degrade the body's protective antibodies (immunoglobulins) that are found in body secretions.


