7.7C: Protein Folding, Modification, and Targeting
- Page ID
- 9288
In order to function, proteins must fold into the correct three-dimensional shape, and be targeted to the correct part of the cell.
LEARNING OBJECTIVES
Discuss how post-translational events affect the proper function of a protein
Key Takeaways
Key Points
- Protein folding is a process in which a linear chain of amino acids attains a defined three-dimensional structure, but there is a possibility of forming misfolded or denatured proteins, which are often inactive.
- Proteins must also be located in the correct part of the cell in order to function correctly; therefore, a signal sequence is often attached to direct the protein to its proper location, which is removed after it attains its location.
- Protein misfolding is the cause of numerous diseases, such as mad cow disease, Creutzfeldt-Jakob disease, and cystic fibrosis.
Key Terms
- prion: a self-propagating misfolded conformer of a protein that is responsible for a number of diseases that affect the brain and other neural tissue
- chaperone: a protein that assists the non-covalent folding/unfolding of other proteins
Protein Folding
After being translated from mRNA, all proteins start out on a ribosome as a linear sequence of amino acids. This linear sequence must “fold” during and after the synthesis so that the protein can acquire what is known as its native conformation. The native conformation of a protein is a stable three-dimensional structure that strongly determines a protein’s biological function. When a protein loses its biological function as a result of a loss of three-dimensional structure, we say that the protein has undergone denaturation. Proteins can be denatured not only by heat, but also by extremes of pH; these two conditions affect the weak interactions and the hydrogen bonds that are responsible for a protein’s three-dimensional structure. Even if a protein is properly specified by its corresponding mRNA, it could take on a completely dysfunctional shape if abnormal temperature or pH conditions prevent it from folding correctly. The denatured state of the protein does not equate with the unfolding of the protein and randomization of conformation. Actually, denatured proteins exist in a set of partially-folded states that are currently poorly understood. Many proteins fold spontaneously, but some proteins require helper molecules, called chaperones, to prevent them from aggregating during the complicated process of folding.
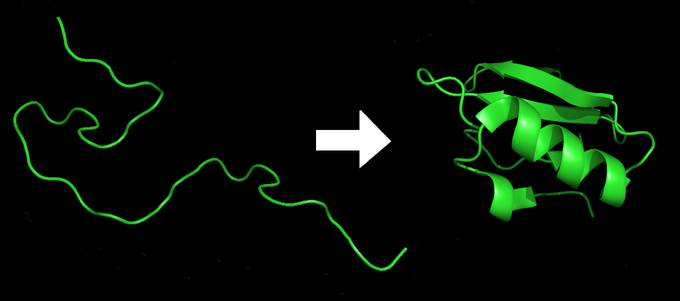
Protein Modification and Targeting
During and after translation, individual amino acids may be chemically modified and signal sequences may be appended to the protein. A signal sequence is a short tail of amino acids that directs a protein to a specific cellular compartment. These sequences at the amino end or the carboxyl end of the protein can be thought of as the protein’s “train ticket” to its ultimate destination. Other cellular factors recognize each signal sequence and help transport the protein from the cytoplasm to its correct compartment. For instance, a specific sequence at the amino terminus will direct a protein to the mitochondria or chloroplasts (in plants). Once the protein reaches its cellular destination, the signal sequence is usually clipped off.
Misfolding
It is very important for proteins to achieve their native conformation since failure to do so may lead to serious problems in the accomplishment of its biological function. Defects in protein folding may be the molecular cause of a range of human genetic disorders. For example, cystic fibrosis is caused by defects in a membrane-bound protein called cystic fibrosis transmembrane conductance regulator (CFTR). This protein serves as a channel for chloride ions. The most common cystic fibrosis-causing mutation is the deletion of a Phe residue at position 508 in CFTR, which causes improper folding of the protein. Many of the disease-related mutations in collagen also cause defective folding.
A misfolded protein, known as prion, appears to be the agent of a number of rare degenerative brain diseases in mammals, like the mad cow disease. Related diseases include kuru and Creutzfeldt-Jakob. The diseases are sometimes referred to as spongiform encephalopathies, so named because the brain becomes riddled with holes. Prion, the misfolded protein, is a normal constituent of brain tissue in all mammals, but its function is not yet known. Prions cannot reproduce independently and not considered living microoganisms. A complete understanding of prion diseases awaits new information about how prion protein affects brain function, as well as more detailed structural information about the protein. Therefore, improved understanding of protein folding may lead to new therapies for cystic fibrosis, Creutzfeldt-Jakob, and many other diseases.


