8.5: Evidence for Heteroduplexes from Recombination in Fungi
- Page ID
- 356
The mechanism by which recombination occurs has been studied primarily in fungi, such as the budding yeast Saccharomyces cerevisiae and the filamentous fungus Ascomycetes, and in bacteria. The fungi undergo meiosis, and hence some aspects of their recombination systems may be more similar to that of plants and animals than is that of bacteria. However, the enzymatic functions discovered by genetic and biochemical studies of recombination in bacteria are also proving to have counterparts in eukaryotic organisms as well. We will refer to studies mainly in fungi for the models of recombination, and to studies mainly in bacteria for the enzymatic pathways.
Many important insights into the mechanism of recombination have come from studies in fungi. One fundamental observation is that recombination proceeds by the formation of a region of heteroduplex, i.e. the recombination products have a region with one strand from one chromosome and the complementary strand from the other chromosome. Thus recombination is not a simple cut and paste operation, unlike the joining of two different molecules by recombinant DNA technology. The two recombining molecules are joined and form a hybrid, or heteroduplex, over part of their lengths.
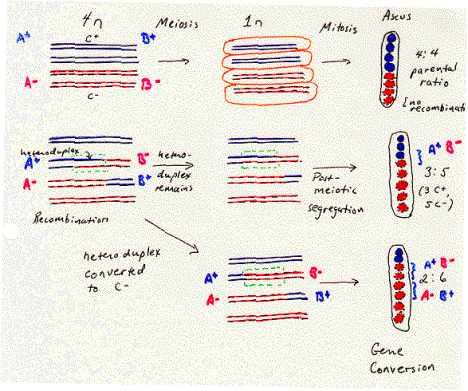
The anatomy and physiology of the filamentous fungus Ascomycetes allows one to observe this heteroduplex formed during recombination. A cell undergoing meiosis starts with a 4n complement of chromosomes (i.e. twice the diploid number) and undergoes two rounds of cell division to form four haploid cells. In fungi these haploid germ cells are spores, and they are found together in an ascus. They can be separated by dissection and plated individually to examine the phenotype of the four products of meiosis. This is called tetrad analysis.
The fungus Ascomycetes goes one step further. After meiosis is completed, the germ cells undergo one further round of replication and mitosis. This separates each individual polynucleotide chain (or “strand” in the sense used in nucleic acid biochemistry) of each DNA duplex in the meiotic products into a separate spore. The eight spores in the ascus reflect the genetic composition of each of the eight polynucleotide chains in the four homologous chromosomes. (The two sister chromatids in each homologous chromosome become two chromosomes after meiosis, and each chromosome is a duplex of two polynucleotide chains.)
The order of the eight spores in the ascus of Ascomycetes reflects the descent of the spores from the homologous chromosomes. As shown in Figure 8.5, a heterozygote with a “blue” allele on one homologous chromosome and a “red” allele on the other will normally produce four “blue” spores and four “red” spores. The four spores with the same phenotype were derived from one homologous chromosome and are adjacent to each other in the ascus. This is called a 4:4 parental ratio, i.e. with respect to the phenotypes of the parent of the heterozygote.
The evidence for heteroduplex formation comes from deviations from the normal 4:4 ratio. Sometimes a 3:5 parental ratio is seen for a particular genetic marker. This shows that one polynucleotide chain of one allele has been lost (giving 4-1=3 spores with the corresponding phenotype in the ascus) and replaced by the polynucleotide chain of the other allele (giving 4+1=5 spores with the corresponding phenotype). As illustrated in Figure 8.5, this is 3 blue spores and 5 red spores. The segment of the chromosome containing this gene was a heteroduplex with one chain from each of two alleles. The round of replication and mitosis that follows meiosis in this fungus allows the two chains to be separated into two alleles that generated a different phenotype in a plating assay. Thus this 3:5 ratio results from post-meiotic segregation of the two chains of the different alleles. In this fungus, a region of heteroduplex can be directly observed by a plating assay.
The region of heteroduplex is associated with a recombination between the chromosomes. Other genes flank the region of heteroduplex shown in Figure 8.5. In many cases, the arrangement of alleles of these flanking genes has changed from that on the parental chromosomes, reflecting a recombination. For instance, let the region of heteroduplex be in a gene B, flanked by gene A in the left and gene C on the right. Each gene has a blue allele and a red allele, making the parental chromosomes AbBbCb and ArBrCr. If one monitored the phenotypes of determined by genes A and C (in addition to B) in the third and fourth spores (derived from the chromosome with the heteroduplex), they would see the phenotypes for the nonparental chromosomes AbBbCr and AbBrCr. This change in the flanking markers (genes A and C) reflects a recombination. Thus the heteroduplex can be found between markers that have undergone recombination.
Other markers can show a 2:6 parental ratio. This means that one of the alleles (formerly blue in fig. 8.5) has been changed to the other allele (now red), in a process called gene conversion. This can occur between flanking markers that have been switched because of recombination. Thus like the heteroduplex, the region of gene conversion is associated with recombination. Models for recombination need to incorporate both phenomenon into their proposed mechanism.
Exercise 8.3.
Imagine that you are studying a fungus that generates an ascus with 8 spores like Ascomycetes, in which the products of meiosis complete an additional round of replication and mitosis. You generate a heterozyous strain by mating a parent that was homozyous for the markers leu+, SmR, ade8+ and another that was leu-, SmS, ade8-. Previous studies had shown that all three markers are linked in the order given. Each of these pairs of alleles can be distinguished in a plating assay. The allele leu+ confers leucine auxotrophy whereas leu- confers leucine prototrophy. The allele SmR confers resistance to spectinomycin whereas SmS is sensitive to this antibiotic. Colonies of fungi with the ade8+ allele give a red color in under appropriate conditions in a plate, but those with the ade8- are white. Analysis of the individual spores from an ascus gave the following phenotypes results. The spores are numbered in the order they were in the ascus. What are the corresponding genotypes of the chromosome in each spore? How do you interpret these results with respect to recombination?
| Spore | leucine | Spectinomycin | Color in ade test |
|---|---|---|---|
| 1 | prototroph | resistant | red |
| 2 | prototroph | resistant | red |
| 3 | prototroph | resistant | white |
| 4 | prototroph | sensitive | white |
| 5 | auxotroph | sensitive | red |
| 6 | auxotroph | sensitive | red |
| 7 | auxotroph | sensitive | white |
| 8 | auxotroph | sensitive | white |


