15.7A: Sexual Reproduction
- Page ID
- 5496
Sexual reproduction is the formation of a new individual following the union of two gametes. In humans and the majority of other eukaryotes plants as well as animals the two gametes differ in structure ("anisogamy") and are contributed by different parents. Gametes need motility to be able to meet and unite and food to nourish the developing embryo. In animals (and some plants), these two rather contrasting needs are met by anisogametes: sperm that are motile (and small) and eggs that contain food.
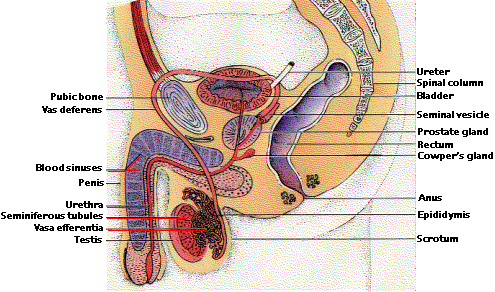
Sex Organs of the Human Male
The reproductive system of the male has two major functions: (1) production of sperm and (2) delivery of these to the reproductive tract of the female. Sperm production - spermatogenesis - takes place in the testes. Each testis is packed with seminiferous tubules (laid end to end, they would extend more than 20 meters) where spermatogenesis occurs.
Spermatogenesis
The walls of the seminiferous tubules consist of diploid spermatogonia, stem cells that are the precursors of sperm. Spermatogonia divide by mitosis to produce more spermatogonia or differentiate into spermatocytes. Meiosis of each spermatocyte produces 4 haploid spermatids. This process takes over three weeks to complete. Then the spermatids differentiate into sperm, losing most of their cytoplasm in the process.
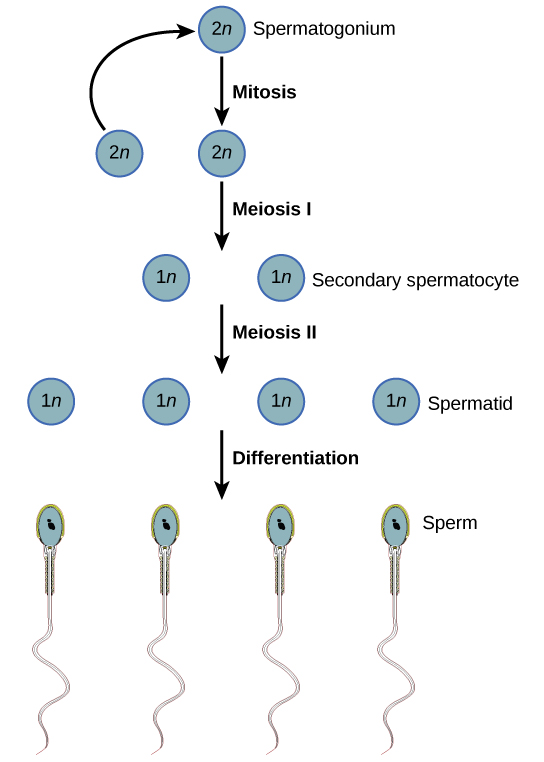
For simplicity, the figure shows the behavior of just a single pair of homologous chromosomes with a single crossover. With 22 pairs of autosomes and an average of two crossovers between each pair, the variety of gene combinations in sperm is very great.
Sperm
Sperm cells are little more than flagellated nuclei. Each consists of a head, which has an acrosome at its tip and contains a haploid set of chromosomes in a compact, inactive, state, a midpiece containing mitochondria and a single centriole, and a tail which is a flagellum. This electron micrograph (courtesy of Dr. Don W. Fawcett and Susumu Ito) shows the sperm cell of a bat. Note the orderly arrangement of the mitochondria. They supply the ATP to power the whiplike motion of the tail.
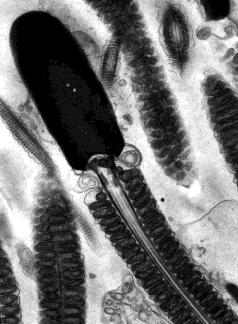
An adult male manufactures over 100 million sperm cells each day. These gradually move into the epididymis where they undergo further maturation. The acidic environment in the epididymis keeps the mature sperm inactive. In addition to making sperm, the testis is an endocrine gland. Its principal hormone, testosterone, is responsible for the development of the secondary sex characteristics of men such as the beard, deep voice, and masculine body shape. Testosterone is also essential for making sperm. Testosterone is made in the interstitial cells (also called Leydig cells) that lie between the seminiferous tubules.
LH
Interstitial cells are, in turn, the targets for a hormone often called interstitial cell stimulating hormone (ICSH). It is a product of the anterior lobe of the pituitary gland. However, ICSH is identical to the luteinizing hormone (LH) found in females, and I prefer to call it LH.
FSH
Follicle-stimulating hormone (also named for its role in females) acts directly on spermatogonia to stimulate sperm production (aided by the LH needed for testosterone synthesis).
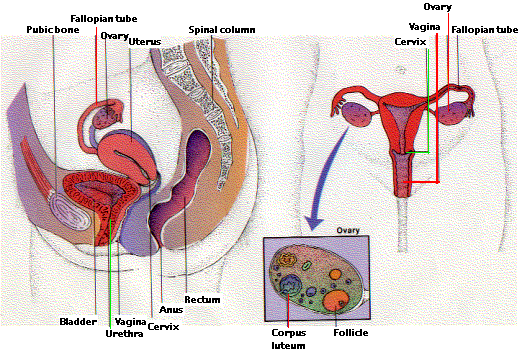
Sex Organs of the Human Female
The responsibility of the female mammal for successful reproduction is considerably greater than that of the male.
She must
- manufacture eggs
- be equipped to receive sperm from the male
- provide an environment conducive to fertilization and implantation
- nourish the developing baby not only before birth but after too
Oogenesis
Egg formation takes place in the ovaries. In contrast to males, the initial steps in egg production occur prior to birth. Diploid stem cells called oogonia divide by mitosis to produce more oogonia and primary oocytes. By the time the fetus is 20 weeks old, the process reaches its peak and all the oocytes that she will ever possess (~4 million of them) have been formed (*). By the time she is born, only about 1 million of these remain (the others eliminated by apoptosis). Each has begun the first steps of the first meiotic division stopping at the diplotene stage of meiosis I.
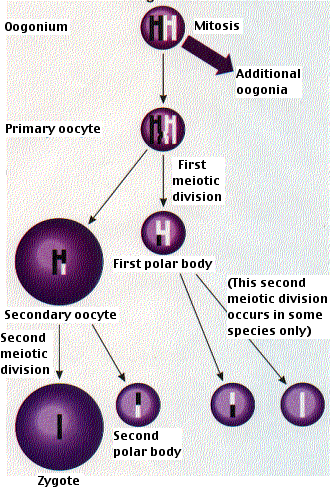
No further development occurs until years later when the girl becomes sexually mature. Then the primary oocytes recommence their development, usually one at a time and once a month. The primary oocyte grows much larger and completes meiosis I, forming a large secondary oocyte and a small polar body that receives little more than one set of chromosomes. Which chromosomes end up in the egg and which in the polar body is entirely a matter of chance.
In humans (and most vertebrates), the first polar body does not go on to meiosis II, but the secondary oocyte does proceed as far as metaphase of meiosis II and then stops.
Only if fertilization occurs will meiosis II ever be completed. Entry of the sperm restarts the cell cycle breaking down MPF (M-phase promoting factor) and turning on the anaphase-promoting complex (APC). Completion of meiosis II converts the secondary oocyte into a fertilized egg or zygote (and also a second polar body). As in the diagram for spermatogenesis, the behavior of the chromosomes is greatly simplified.
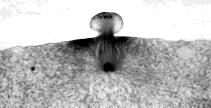
The photomicrograph (courtesy of Turtox) shows polar body formation during oogenesis in the whitefish. Even allowing for the fact that fish eggs are larger than mammalian eggs, you can readily see how the polar body gets little more than one set of chromosomes.
These events take place within a follicle, a fluid-filled envelope of cells surrounding the developing egg. The ripening follicle also serves as an endocrine gland. Its cells make a mixture of steroid hormones collectively known as estrogen. Estrogen is responsible for the development of the secondary sexual characteristics of a mature woman, e.g.,
- a broadening of the pelvis
- development of the breasts
- growth of hair around the genitals and in the armpits
- development of adipose tissue leading to the more rounded body contours of adult women.
Estrogen continues to be secreted throughout the reproductive years of women During this period, it plays an essential role in the monthly menstrual cycle.
Ovulation
Ovulation occurs about two weeks after the onset of menstruation. In response to a sudden surge of LH, the follicle ruptures and discharges a secondary oocyte. This is swept into the open end of the fallopian tube and begins to move slowly down it.
Copulation and Fertilization
For fertilization to occur, sperm must be deposited in the vagina within a few (5) days before or on the day of ovulation. Sperm transfer is accomplished by copulation. Sexual excitation dilates the arterioles supplying blood to the penis. Blood accumulates in three cylindrical spongy sinuses that run lengthwise through the penis. The resulting pressure causes the penis to enlarge and erect and thus able to penetrate the vagina. Movement of the penis back and forth within the vagina causes sexual tension to increase to the point of ejaculation. Contraction of the walls of each vas deferens propels the sperm along. Fluid is added to the sperm by the seminal vesicles, Cowper's glands, and the prostate gland. These fluids provide
- a source of energy (fructose)
- an alkaline environment to activate the sperm
- perhaps in other ways provide an optimum chemical environment for them
The mixture of sperm and accessory fluids is called semen. It passes through the urethra and is expelled into the vagina. Physiological changes occur in the female as well as the male in response to sexual excitement, although these are not as readily apparent. In contrast to the male, however, such responses are not a prerequisite for copulation and fertilization to occur. Once deposited within the vagina, the sperm proceed on their journey into and through the uterus and on up into the fallopian tubes. It is here that fertilization may occur if an "egg" is present (strictly speaking, it is still a secondary oocyte until after completion of meiosis II).
Although sperm can swim several millimeters each second, their trip to and through the fallopian tubes may be assisted by muscular contraction of the walls of the uterus and the tubes. The trip is fraught with heavy mortality. An average human ejaculate contains over one hundred million sperm, but only a few dozen complete the journey, arriving within 15 minutes of ejaculation. And of these, only one will succeed in fertilizing the egg.
Sperm swim towards the egg by chemotaxis following a gradient of progesterone secreted by cells surrounding the egg. Progesterone opens CatSper ("cation sperm") channels in the plasma membrane surrounding the anterior portion of the sperm tail. This allows an influx of Ca2+ ions which causes the flagellum to beat more rapidly and vigorously. Fertilization begins with the binding of a sperm head to the glycoprotein coating of the egg (called the zona pellucida). Exocytosis of the acrosome at the tip of the sperm head releases enzymes that digest a path through the zona and enable the sperm head to bind to the plasma membrane of the egg. The binding is mediated by the binding of two membrane proteins:
- Izumo1 on the surface of the sperm
- Juno, its receptor on the egg surface
Fusion of their respective membranes allows the entire contents of the sperm to be drawn into the cytosol of the egg. Even though the sperm's mitochondria enter the egg, they are almost always destroyed by autophagy and do not contribute their genes to the embryo. So human mitochondrial DNA is almost always inherited from mothers only. Within moments, enzymes released from the egg cytosol act on the zona making it impermeable to other sperm that arrive. Soon the nucleus of the successful sperm enlarges into the male pronucleus. At the same time, the egg (secondary oocyte) completes meiosis II forming a second polar body and the female pronucleus.
The male and female pronuclei move toward each other while duplicating their DNA in S phase. Their nuclear envelopes disintegrate. A spindle is formed (following replication of the sperm's centriole), and a full set of dyads assembles on it. The fertilized egg or zygote is now ready for its first mitosis. When this is done, 2 cells each with a diploid set of chromosomes are formed.
In sea urchins, the block to additional sperm entry and the fusion of the pronuclei are triggered by nitric oxide generated in the egg by the sperm acrosome.
Pregnancy
Development begins while the fertilized egg is still within the fallopian tube. Repeated mitotic divisions produces a solid ball of cells called a morula. Further mitosis and some migration of cells converts this into a hollow ball of cells called the blastocyst. Approximately one week after fertilization, the blastocyst embeds itself in the thickened wall of the uterus, a process called implantation, and pregnancy is established.
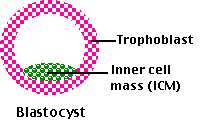
Fig.15.7.1.7 Blastocyst
The blastocyst produces two major collections of cells:
- Three or four blastocyst cells develop into the inner cell mass, which will form 3 extraembryonic membranes: amnion, yolk sac, and (a vestigial) allantois and in about 2 months, become the fetus and, ultimately, the baby.
- The remaining 100 or so cells form the trophoblast, which will develop into the chorion that will go on to make up most of the placenta. All the extraembryonic membranes play vital roles during development but will be discarded at the time of birth.
The placenta grows tightly fused to the wall of the uterus. Its blood vessels, supplied by the fetal heart, are literally bathed in the mother's blood. Although there is normally no mixing of the two blood supplies, the placenta does facilitate the transfer of a variety of materials between the fetus and the mother such as
- receiving food
- receiving oxygen and discharging carbon dioxide
- discharging urea and other wastes
- receiving antibodies (chiefly of the IgG class). These remain for weeks after birth, protecting the baby from the diseases to which the mother is immune.
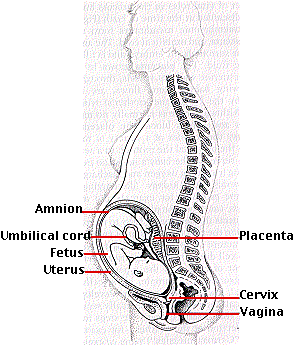
But the placenta is not simply a transfer device. Using raw materials from the mother's blood, it synthesizes large quantities of proteins and also some hormones. The metabolic activity of the placenta is almost as great as that of the fetus itself. The umbilical cord connects the fetus to the placenta. It receives deoxygenated blood from the iliac arteries of the fetus and returns oxygenated blood to the liver and on to the inferior vena cava.
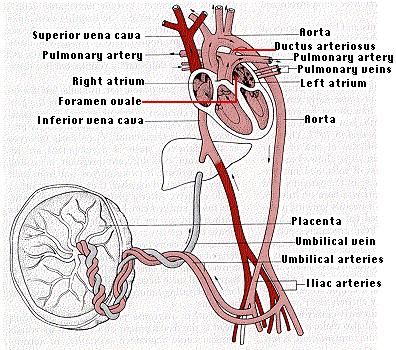
Because its lungs are not functioning, circulation in the fetus differs dramatically from that of the baby after birth. While within the uterus, blood pumped by the right ventricle bypasses the lungs by flowing through the foramen ovale and the ductus arteriosus.
Although the blood in the placenta is in close contact with the mother's blood in the uterus, intermingling of their blood does not normally occur. However, some of the blood cells of the fetus usually do escape into the mother's circulation — where they have been known to survive for decades. This raises the possibility of doing prenatal diagnosis of genetic disorders by sampling the mother's blood rather than having to rely on the more invasive procedures of amniocentesis and chorionic villus sampling (CVS).
Fragments of fetal DNA (~ 300 bp long) from apoptotic cells of the placenta are also found in the mother's plasma as early as 5 weeks after implantation. These can be tested for various forms of aneuploidy, e.g. the trisomy 21 of Down syndrome. Far rarer is the leakage of mother's blood cells into the fetus. However, it does occur. A few pregnant women with leukemia or lymphoma have transferred the malignancy to their fetus. Some babies have also acquired melanoma from the transplacental passage of these highly-malignant cells from their mother.
During the first 2 months of pregnancy, the basic structure of the baby is being formed. This involves cell division, cell migration, and the differentiation of cells into the many types found in the baby. During this period, the developing baby - called an embryo - is very sensitive to anything that interferes with the steps involved. Virus infection of the mother, e.g., by rubella ("German measles") virus or exposure to certain chemicals may cause malformations in the developing embryo. Such agents are called teratogens ("monster-forming"). The tranquilizer, thalidomide, taken by many pregnant European women between 1954 and 1962, turned out to be a potent teratogen and was responsible for the birth of several thousand deformed babies.
After about two months, all the systems of the baby have been formed, at least in a rudimentary way. From then on, development of the fetus, as it is now called, is primarily a matter of growth and minor structural modifications. The fetus is less susceptible to teratogens than is the embryo. Pregnancy involves a complex interplay of hormones.
The placenta is an allograft
One of the greatest unsolved mysteries in immunology is how the placenta survives for 9 months without being rejected by the mother's immune system. Every cell of the placenta carries the father's genome (a haploid set of his chromosomes); including one of his #6 chromosomes where the genes for the major histocompatibility antigens (HLA) are located.
One partial exception: none of the genes on the father's X chromosome are expressed. While X-chromosome inactivation is random in the cells of the fetus, it is NOT random in the cells of the trophoblast. In every cell of the trophoblast — and its descendants — it is the paternal X chromosome that is inactivated. But this does not solve our problem because the genes for all the major histocompatibility antigens are located on chromosome 6, which is not inactivated.
Thus the placenta is immunologically as foreign to the mother as a kidney transplant would be. Yet it thrives. Despite a half-century of research, the mechanism for this immunologically privileged status remains uncertain. But one thing is clear - The mother is not intrinsically tolerant of the father's antigens. She will promptly reject a skin transplant from the father. She develops antibodies against his histocompatibility antigens expressed by the fetus. In fact, women who have borne several children by the same father are often excellent sources of anti-HLA serum for use in tissue typing. So what accounts for the phenomenon? Some possibilities:
- The placenta does not express class II histocompatibility antigens.
- Nor does it express the strongly-immunogenic class I histocompatibility antigens (HLA-A, HLA-B). It does express HLA-C, but this is only weakly immunogenic.
- The cells of the placenta secrete progesterone, which is immunosuppressive.
- In lab rats the embryos (and the mother's endometrium) secrete corticotropin-releasing hormone (CRH). This hormone induces the expression of Fas ligand (FasL) on the cells of the placenta. Activated T cells express Fas so any threatening T cells would commit suicide by apoptosis when they encounter FasL on their target.
- In laboratory mice the cells of the placenta degrade the amino acid tryptophan. Tryptophan is essential for T-cell function. When mice are treated with an inhibitor of the Trp-degrading enzyme, their fetuses are promptly aborted by the action of the mother's lymphocytes. (D. H. Munn, et. al., Science, 281: 1191, 21 Aug 1998.)
- In mice, expression of genes encoding cytokines needed to attract effector T cells (e.g., CTLs) into a tissue is suppressed in the cells of the placenta.
- Perhaps most important of all is the increased production in the mother of immunosuppressive regulatory T cells (Treg).
- Depletion of Treg cells in pregnant mice leads to spontaneous abortion while
- injection of Treg cells into mice that are otherwise prone to abortion enables them to carry their fetuses to term.
- In humans, the number of Treg cells rises during pregnancy (in the fetus as well as the mother).
On July 25, 2013 Louise Brown celebrated her 35th birthday. She was the first of what today number some four million (worldwide) "test tube babies"; that is, she developed from an egg that was fertilized outside her mother's body - the process called in vitro fertilization (IVF). IVF involves
- Harvesting mature eggs from the mother. This is not an easy process. The mother must undergo hormonal treatments to produce multiple eggs, which then must be removed (under anesthesia) from her ovaries.
- Harvesting sperm from the father. Harvesting is usually no problem, but often the sperm are defective in their ability to fertilize (so setting the stage for ICSI).
- Mixing sperm and eggs in a culture vessel ("in vitro").
- Culturing the fertilized eggs for several days until they have developed to at least the 8-cell stage.
- Placing two or more of these into the mother's uterus (which her hormone treatments have prepared for implantation).
- Keeping one's fingers crossed as only about one-third of the attempts result in a successful pregnancy.
Intra cytoplasmic Sperm Injection (ICSI)
Successful IVF assumes the availability of healthy sperm. But many cases of infertility arise from defects in the father's sperm. Often these can be overcome by directly injecting a single sperm into the egg. In the U.S. today, some two-thirds of ART procedures employ ICSI (even though as many as half of these do not involve male infertility).
Ooplasmic Transfer
Infertility in some cases may stem from defects in the cytoplasm of the mother's egg. To circumvent these, cytoplasm can be removed from the egg of a young, healthy woman ("Donor egg") and injected along with a single sperm into the prospective mother's egg. Although a few healthy children have been born following ooplasmic transfer, the jury is still out on its safety, and it is not approved for use in the U.S. One reason for concern is that ooplasmic transfer results in an egg carrying both the mother's mitochondria and mitochondria from the donor (in normal fertilization, all the mitochondria in the father's sperm are destroyed in the egg). This condition called heteroplasmy creates a child having two different mitochondrial DNA genomes in all of its cells.
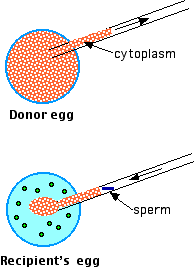
Fig.15.7.1.9 ooplasmic transfer
In rare, but important, cases, the defect in the prospective mother's cytoplasm is the result of her having mitochondria with a mutant gene. Ooplasmic transfer is of no help in these cases because the fertilized egg will still contain a preponderance of the mother's defective mitochondria. But three techniques worked out on laboratory animals show promise of being adapted to aid such women to produce healthy young.
Three Possible Ways to Prevent Transmission of Mitochondrial Diseases
Maternal Spindle Transfer
Researchers in Oregon reported in the 17 September 2009 issue of Nature that they had been able to produce 4 healthy rhesus monkeys with no mitochondria from their biological mother.
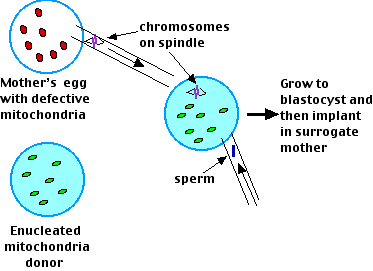
Their procedure:
- Remove the spindle with all its attached chromosomes from the mother's oocyte at metaphase II of meiosis. They managed to do this without any of her mitochondria being withdrawn as well.
- Enucleate the oocyte of the mitochondria donor and then insert the mother's chromosomes — still attached to the spindle — into it. Then inject a sperm from the father.
- Allow the fertilized egg to develop into a blastocyst.
- Implant this in the uterus of a surrogate mother.
- The result: 4 healthy babies each with the nuclear genes of their mother and father but none of the mitochondria of their mother.
Pronuclear Transfer
In this procedure,
- An egg containing mutant mitochondria is fertilized by IVF.
- The two pronuclei are removed and injected into a fertilized egg with healthy mitochondria and whose own pronuclei have been removed.
- Allow the egg to develop into a blastocyst.
- Implant this in the uterus.
Both procedures (1) and (2) are under investigation for use in humans.
Polar Body Transfer
Both the first polar body (formed before fertilization) and the second polar body (formed after fertilization) contain a genome equivalent to that of the secondary oocyte and zygote respectively. However, they contain few, if any, mitochondria. In mice, transfer of either polar body to an enucleated recipient egg (with healthy mitochondria) yield young mice with few, if any, of the donor mother's mitochondria with their defective mtDNA. So in mice, at least, this technique produces offspring with less heteroplasmy than the other two techniques.
If any of these techniques can be applied to humans (there are safety and regulatory hurdles still to be overcome), it would allow women carrying defective mitochondria to bear babies free of the ailment.
The Upside of ART
- It has allowed some four million previously-infertile couples to have children.
- It permits screening (on one cell removed from the 8-celled morula) for the presence of genetic disorders - thus avoiding starting a pregnancy if a disorder is found.
- One can use frozen sperm allowing fatherhood for a man who is no longer able to provide fresh sperm.
- Because a number of morulas are created, the extras can be frozen, stored, and used later
- if the initial attempt fails (the prospective mother must still receive hormones to prepare her uterus for implantation and the success rate is lower with thawed morulas).
- Where regulations permit, the extras can be used as a source of embryonic stem (ES) cells.
The Downside of ART
- Although improving, the success rate is still sufficiently low (~35%) that the process often has to be repeated (which is physically demanding as well as very expensive).
- Because several morulas are usually transferred, multiple births are common (about 50%), and as is often the case with multiple births, the babies are born early and weigh less (~one-third of all ART babies in the U.S. are born early). To reduce the number of twins, triplets, etc., more ART centers are turning to "single-embryo transfer" (SET). Some ART centers find that they can increase the success rate and thus rely more on SET by culturing the morulas for 5–6 days, instead of the usual 2–3 days, before transferring them (by now they have become blastocysts) to the mother.
- The risk of birth defects may be increased slightly (from ~6% in "normal" pregnancies to ~8% in ART pregnancies).
- ART procedures in experimental animals often result in a failure of correct gene imprinting. Whether this will pose a problem for humans remains to be seen.
Birth and Lactation
Exactly what brings about the onset of labor is still not completely understood. Probably a variety of integrated hormonal controls are at work. A growing body of evidence implicates a rise in the level of fetal DNA in the mother's blood as a trigger for the onset of labor. The first result of labor is the opening of the cervix. With continued powerful contractions, the amnion ruptures and the amniotic fluid (the "waters") flows out through the vagina. The baby follows, and its umbilical cord can be cut. The infant's lungs expand, and it begins breathing. This requires a major switchover in the circulatory system. Blood flow through the umbilical cord, ductus arteriosus, and foramen ovale ceases, and the adult pattern of blood flow through the heart, aorta, and pulmonary arteries begins. In some infants, the switchover is incomplete, and blood flow through the pulmonary arteries is inadequate. Failure to synthesize enough nitric oxide (NO) is one cause. Shortly after the baby, the placenta and the remains of the umbilical cord (the "afterbirth") are expelled.
At the time of birth, and for a few days after, the mother's breasts contain a fluid called colostrum. It is rich in calories and proteins, including antibodies that provide passive immunity for the newborn infant. Three or four days after delivery, the breasts begin to secrete milk.
- Its synthesis is stimulated by the pituitary hormone prolactin (PRL).
- Its release is stimulated by a rise in the level of oxytocin when the baby begins nursing.
- Milk also contains an inhibitory peptide. If the breasts are not fully emptied, the peptide accumulates and inhibits milk production. This autocrine action thus matches supply with demand.
Birth Control
The following table summarizes the various birth control method available and in use today.
| Method | Popularity | Pregnancy Rate |
|---|---|---|
| Natural family planning (rhythm) | 1% | 25 |
| Male condom | 16% | 17 |
| Oral contraceptives ("the pill") | 28% | 0.3–8.7* |
| Intrauterine devices (IUD) | 8.5% | 0.1–1.0* |
| Implants, e.g., Implanon® | ~1% | 0.05–1.0* |
| DMPA injections | ~3.5% | 6.7 |
| Male + Female Sterilization | 37% | <<1% |
| None | 85 | |
| * The lower value is found under ideal conditions; i.e., among highly-motivated women receiving good medical care. | ||
Contributors and Attributions
John W. Kimball. This content is distributed under a Creative Commons Attribution 3.0 Unported (CC BY 3.0) license and made possible by funding from The Saylor Foundation.


