E5. Oxidation States of Nitrogenase Fe centers
- Page ID
- 5719
First Half: It would be difficult to assign specific oxidation states to each Fe ion in the M complex. Instead we can assign relative changes in the oxidation states as the reaction proceeds from E0 to E4. In each step of the LT model, 1 electron is added. We will first assign this to a average Fe ion, M0, with an arbitrarily assigned oxidation state of 0. On addition of 1 electron, the oxidation state would go from M0 to M-1 as the metal is reduced. The M-1 state is then oxidized as an electron is transferred to H+, and when 2 electrons are transferred, and a single H- is made. The diagram below shows the change in oxidation state in going from E0 to E4.
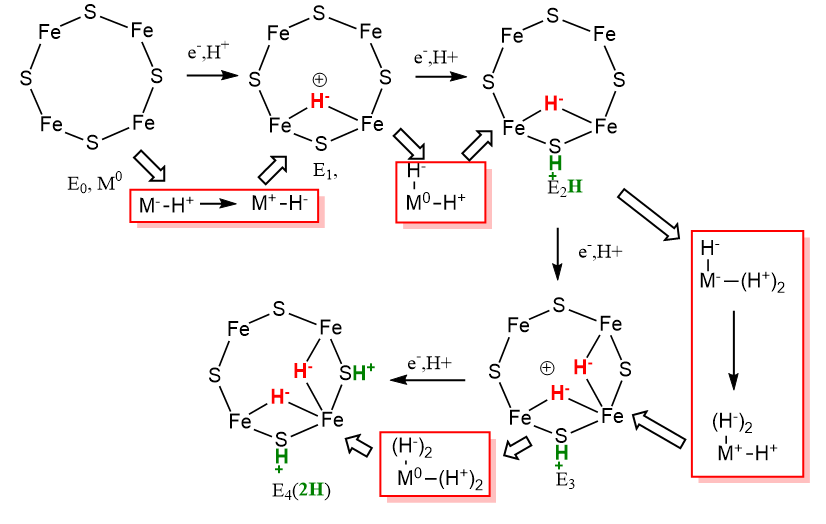
The red boxes highlight thermodynamic cycle-like steps which show how the changes in redox state of the Fe ions (M) could be visualized. Note that in going from E0 to E4, the actual oxidation state of M changes from 0 to +1 to 0 to +1 and back to 0. That is quite amazing given that 4 electrons have been added. Note also that in the red box going from step E0 to E1, M goes from -1 to +1 which corresponds to our description of an oxidative addition when the metal center looses two electrons. This mechanism shows that nitrogenase could be considered a "hydride storage device".
Second Half (facing forward to production NH3):
How does N2 initially interact with E4? It must depend on how the hydrides are released as H2, which evidence shows occurs by reductive elimination (re) and not hydride protonation (hp). On addition, the N2 very quickly is converted to diazene, HN=NH, with the departing H2 taking with it 2 H+s and 2 electrons (or reducing equivalents). These events could occur as shown in the figure below.
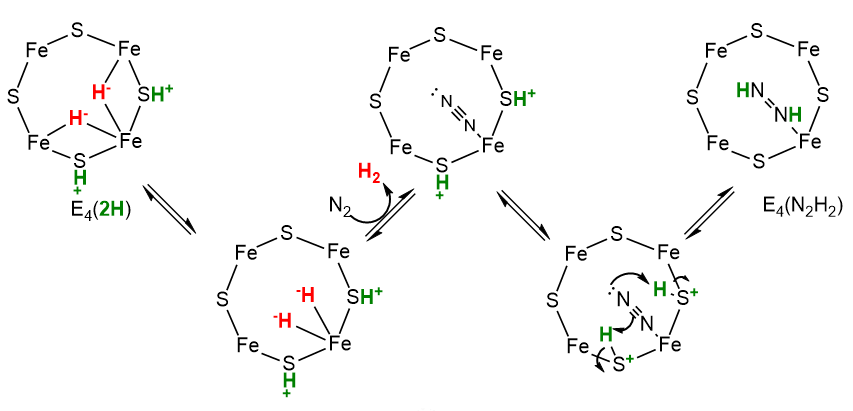
Now, with N2 bound as diazene (N2H2) and H2 released, the rest of the reaction could occur as shown below. One new step, a migratory insertion, is shown.
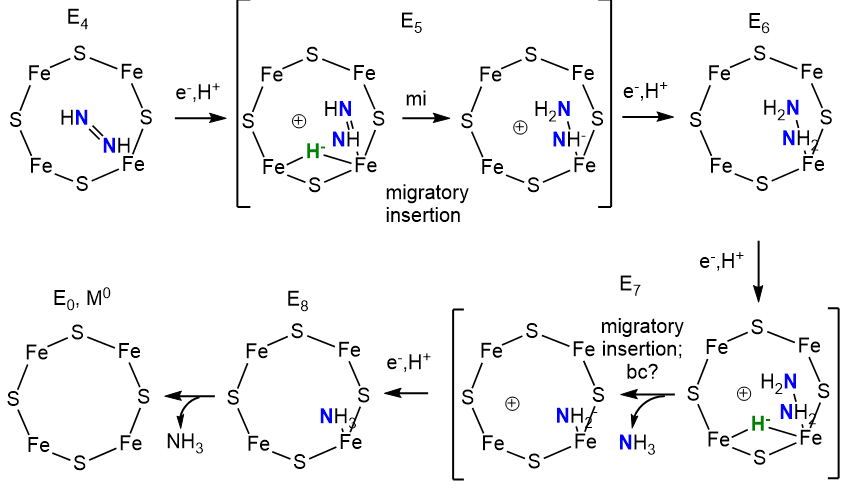
The two halves of the reaction are similar with bridging hydrides utilized. The E4 Janus intermediate links the two halves together.
Contributors


