11.8: Gel Blotting
- Page ID
- 4930
Gel blotting is a technique for visualizing a particular subset of macromolecules — proteins, or fragments of DNA or RNA — initially present in a complex mixture. The steps:
- Separate the molecules by electrophoresis. This is done in a gel which allows the molecules to migrate under the influence of the electric field.
- "Blot" them with a nitrocellulose filter. For unknown reasons, the molecules stick tightly to the filter and will retain their relative positions when flooded with fluid at the next step.
- Bathe the filter with a solution containing a "probe": a molecule that will combine specifically with the target molecules; that is, the one(s) you are looking for and carries a mean of visualization, e.g. a radioactive or fluorescent marker.
Southern Blot
The diagram illustrates the procedure for detecting DNA fragments containing a particular sequence. DNA is extracted from the cell and is partially digested by a restriction endonuclease. The resulting DNA fragments are separated by electrophoresis and then denatured to form single-stranded molecules (ssDNA). Without altering their positions, the separated bands of ssDNA are transferred to a nitrocellulose filter and exposed to radiolabeled cDNA or RNA. If the probe detects complementary DNA sequences, it will bind to them. The presence of the probe in a particular band is revealed by autoradiography.
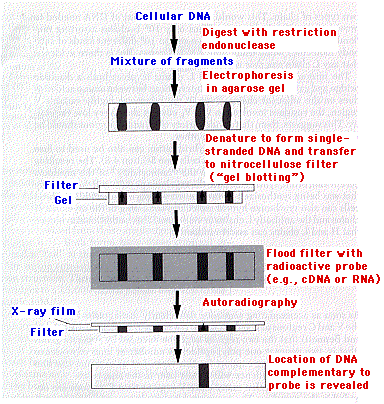
This procedure was developed by E. M. Southern and the finished product is called a "Southern blot". The same basic procedure can also be used to separate and visualize RNA molecules and protein molecules. As a humorous extension of the term "Southern blot", these have been dubbed "Northern" and "Western" blots, respectively (Table \(\PageIndex{1}\)).
| Type of Blot | Molecules separated by electrophoresis | Probe |
|---|---|---|
| Southern | ssDNA | cDNA or RNA |
| Northern | denatured RNA | RNA or cDNA |
| Western | Protein | Antibodies |


