7.3: Ionizing Radiation
- Page ID
- 346
High energy radiation, such as X-rays, \(\gamma\)-rays, and \(\beta\) particles (or electrons) are powerful mutagens. Since they can change the number of electrons on an atom, converting a compound to an ionized form, they are referred to as ionizing radiation. They can cause a number of chemical changes in DNA, including directly break phosphodiester backbone of DNA, leading to deletions. Ionizing radiation can also break open the imidazole ring of purines. Subsequent removal of the damaged purine from DNA by a glycosylase generates an apurinic site.
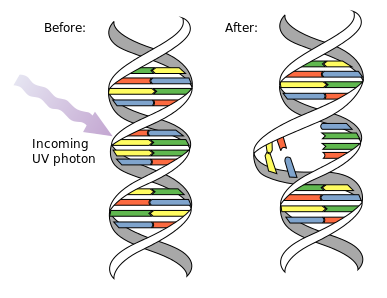
Ultraviolet Radiation
Ultraviolet radiation with a wavelength of 260 nm will form pyrimidine dimers between adjacent pyrimidines in the DNA. The dimers can be one of two types (Figure 7.11). The major product is a cytobutane-containing thymine dimer (between C5 and C6 of adjacent T's). The other product has a covalent bond between position 6 on one pyrimidine and position 4 on the adjacent pyrimidine, hence it is called the "6-4" photoproduct.
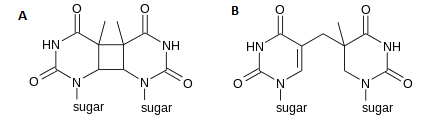
The pyrimidine dimers cause a distortion in the DNA double helix. This distortion blocks replication and transcription.
Exercise \(\PageIndex{1}\)
What is the physical basis for this distortion in the DNA double helix?


