4.9: Perfect Enzymes
- Page ID
- 3036
Now, if we think about what an ideal enzyme might be, it would be one that has a very high velocity and a very high affinity for its substrate. That is, it wouldn’t take much substrate to get to \(V_{max}/2\) and the \(K_{cat}\) would be very high. Such enzymes would have values of \(K_{cat} / K_M\) that are maximum. Interestingly, there are several enzymes that have this property and their maximal values are all approximately the same. Such enzymes are referred to as being “perfect" because they have reached the maximum possible value. Why should there be a maximum possible value of \(K_{cat} / K_M\). The answer is that movement of substrate to the enzyme becomes the limiting factor for perfect enzymes. Movement of substrate by diffusion in water has a fixed rate and that limitation ultimately determines how fast the enzyme can work. In a macroscopic world analogy, factories can’t make products faster than suppliers can deliver materials. It is safe to say for a perfect enzyme that the only limit it has is the rate of substrate diffusion in water.
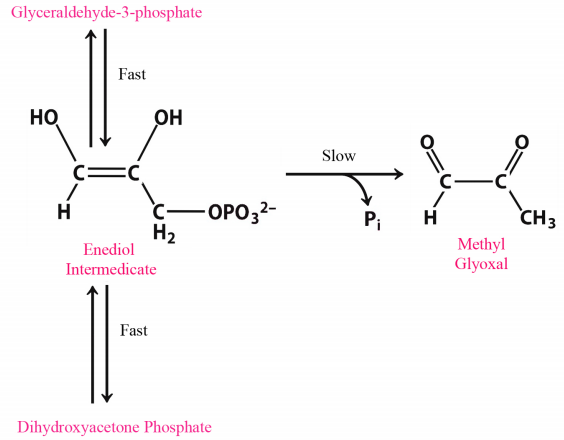
Given the “magic" of enzymes alluded to earlier, it might seem that all enzymes should have evolved to be “perfect." There are very good reasons why most of them have not. Speed can be a dangerous thing. The faster a reaction proceeds in catalysis by an enzyme, the harder it is to control. As we all know from learning to drive, speeding causes accident. Just as drivers need to have speed limits for operating automobiles, so too must cells exert some control on the ‘throttle’ of their enzymes. In view of this, one might wonder then why any cells have evolved any enzymes to perfection. There is no single answer to the question, but a common one is illustrated by the perfect enzyme known as triose phosphate isomerase (TPI), which catalyzes a reaction in glycolysis (figure on previous page). The enzyme appears to have been selected for this ability because at lower velocities, there is breakdown of an unstable enediol intermediate that then readily forms methyl glyoxal, a cytotoxic compound. Speeding up the reaction provides less opportunity for the unstable intermediate to accumulate and fewer undesirable byproducts are made.
Dr. Kevin Ahern and Dr. Indira Rajagopal (Oregon State University)


