W2017_Lecture_12_reading
- Page ID
- 8136
ATP
Another chemical compound we need to become familiar with is adenosine triphospate (ATP). The main cellular role of ATP is as a "short term" energy transfer device for the cell. The hydrolysis reactions that liberate one or more of ATP's phosphates are exergonic and many, many cellular proteins have evolved to interact with ATP in ways that help to facilitate the transfer of energy from hydrolysis to myriad other cellular functions. In this way, ATP is often called the “energy currency” of the cell - it has reasonably fixed values of energy to transfer to or from itself and can exchange that energy between many potential donors and acceptors. We will see many examples of ATP "at work" in the cell - be on the lookout for them and as you see them try to think of them as functional examples of Nature's uses for ATP that you could be expected to see in another reaction or context.
ATP Structure and Function
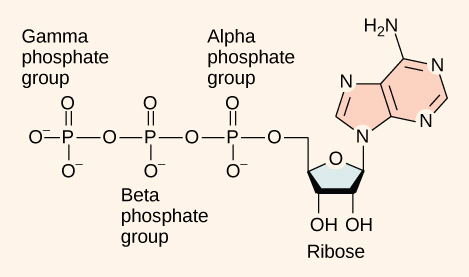
At the heart of ATP is the nucleotide adenosine monophosphate (AMP). Like the other nucleotides AMP is composed of a nitrogenous base (an adenine molecule) bonded to a ribose molecule and a single phosphate group. The addition of a second phosphate group to this core molecule results in the formation of adenosine diphosphate (ADP); the addition of a third phosphate group forms adenosine triphosphate (ATP).
ATP (adenosine triphosphate) has three phosphate groups that can be removed by hydrolysis to form ADP (adenosine diphosphate) or AMP (adenosine monophosphate).
The phosphorylation or condensation of phosphate groups onto AMP is an endergonic process. By contrast, the hydrolysis of one or two phosphate groups from ATP, a process called dephosphorylation, is exergonic. Why? Let's recall that the terms endergonic and exergonic refer to the sign on the difference in free energy of a reaction between the products and reactants, ΔG. In this case we are explicitly assigning direction to the reaction, either in the direction of phosphorylation or dephosphorylation of the nucleotide. In the phosphorylation reaction the reactants are the nucleotide and an inorganic phosphate while the products are a phosphorylated nucleotide and WATER. In the dephosphorylation/hydrolysis reaction, the reactants are the phosphorylated nucleotide and WATER while the products are inorganic phosphate and the nucleotide minus one phosphate.
Since Gibbs free energy is a state function, it doesn't matter how the reaction happens, you just consider the beginning and ending states. So, let's for example examine the hydrolysis of ATP. The reactants ATP and water are characterized by their atomic makeup and the kinds of bonds between the constituent atoms and some free energy can be associated with each of the bonds and their possible configurations - likewise for the products. If we examine the reaction from the standpoint of the products and reactants and ask "how can we recombine atoms and bonds in the reactants to get the products?", we find that a phosphoanhydride bond between an oxygen and a phosphorus must be broken in the ATP, a bond between an oxygen and hydrogen broken in the water, a bond made between the OH (that came from the splitting of water) and the phosphorus (from the freed PO3-2), and a bond must be formed between the H (derived from the splitting of water), and the terminal oxygen on the phosphorylated nucleotide. It is the sum of energies associated with all of those bond rearrangements (including those directly associated with water) that make this reaction exergonic. A similar analysis could be made with the reverse reaction.
Possible Exercise
Use the figure of ATP above and your knowledge of what a water molecule looks like to draw a figure of the reaction steps described above: breaking of phosphoanhydride bond, breaking of water, and formation of new bonds to form ADP and inorganic phosphate. Track the atoms in different colors if that helps.
Is there something special about the specific bonds involved in these molecules? Much is made in various texts about the types of bonds between the phosphates of ATP. Certainly, the properties of the bonds in ATP help define the molecule's free energy and reactivity. However, while it is appropriate to apply concepts like charge density and availability of resonance structures to this discussion, trotting these terms out as an "explanation" without a thorough understanding of how these factors influence the free energy of the reactants is a special kind of hand-waving that we prefer not to engage in. Most BIS2A students have not had any college chemistry and those that have are not likely to have discussed those terms in any meaningful way. So, trying to explain the process using the ideas above would do nothing but give a false sense of understanding, tend to assign some mystical quality to ATP and is "special" bonds that doesn't exist, and distract from the real point, that the hydrolysis reaction is exergonic because of the properties of ATP but ALSO because of the chemical properties of water and those of the reaction products. For this class, it is sufficient to know that dedicated physical chemists are still studying the process of ATP hydrolysis in solution and in the context of proteins and that they are still trying to account for the key enthalpic and entropic components of the component free energies. We'll just need to accept a certain degree of mechanistic chemical ignorance and be content with a description of gross thermodynamic properties. The latter is perfectly sufficient to have deep discussions about the relevant biology.
"High Energy" Bonds
What about the term "high energy bonds" that we so often hear associated with ATP? If there is nothing "special" about the bonds in ATP why do we always hear the term "high energy bonds" associated with the molecule? The answer is deceptively simple. In biology the term "high energy bond" is used to describe an exergonic reaction involving the hydrolysis of the bond in question that results in a "large" negative change in free energy. Remember that this change in free energy does not only have to do with the bond in question but rather the sum of all bond rearrangements in the reaction. What constitutes a large change? This, it seems, is a rather arbitrary assignment that is usually associated with an amount of energy that is associated with the types of anabolic reactions we typically observe in biology. If there is something special about the bonds in ATP it is not uniquely tied to the free energy of hydrolysis as there are plenty of other bonds whose hydrolysis result in greater negative differences in free energy.
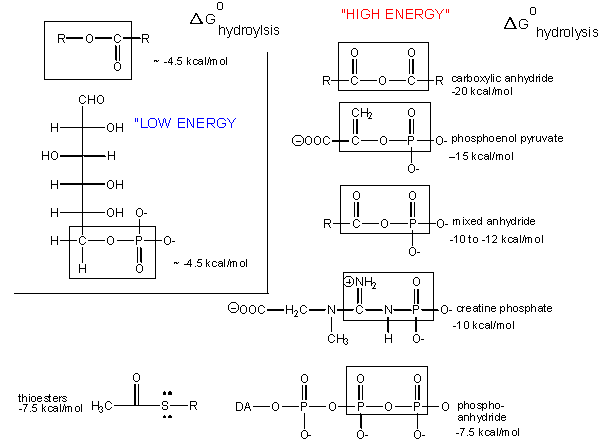
The free energy of hydrolysis of different types of bonds can be compared to that of the hydrolysis of ATP. Source: http://biowiki.ucdavis.edu/Biochemistry/Oxidation_and_Phosphorylation/ATP_and_Oxidative_Phosphorylation/Properties_of_ATP
Video on electron and electron/proton carriers
For a 7 minute YouTube video on the role of carriers in respiration click here.
External link discussing the energetics of coupling ATP hydrolysis to other reactions
The cycling of ATP pools
Estimates for the number of ATP molecules in a typical human cell range from ~3x107 (~5x10-17 moles ATP/cell) in a white blood cell to 5x109 (~9x10-15 moles ATP/cell) in an active cancer cell. While these numbers might seem large, and already amazing, consider that it is estimated that this pool of ATP turns over (becomes ADP and then back to ATP) 1.5 x per minute. Extending this analysis yields the estimate that this daily turnover amounts to roughly the equivalent of one body weight of ATP getting turned over per day. That is, if no turnover/recycling of ATP happened, it would take 1 body weights worth of ATP for the human body to function - hence our previous characterization of ATP as a "short term" energy transfer device for the cell.
While the pool of ATP/ADP may be recycled, some of the energy that is transferred in the many conversions between ATP, ADP and other biomolecules is also transferred to the environment. In order to maintain cellular energy pools energy must transfer in from the environment as well. Where does this energy come from? The answer depends a lot on where energy is available and what mechanisms Nature has evolved to transfer energy from the environment to molecular carriers like ATP. In nearly all cases, however, the mechanism of transfer has evolved to include some form of redox chemistry. In this and the sections that follow we are concerned with learning some critical examples of energy transfer from the environment, key types of chemistry and biological reactions involved in this process, and some key biological reactions and cellular components associated with energy flow between different parts of the living system. We focus first on reactions involved in the (re)generation of ATP in the cell (not those involved in the creation of the nucleotide per se but rather those associated with the transfer of phosphates onto AMP and ADP).
Video Link
For a more detailed explanation of ATP and how this molecule stores energy take a look at this video (10 minutes) by clicking here.
How do cells generate ATP?
A variety of mechanisms have emerged over the 3.25 billion years of evolution to create ATP from ADP and AMP. The majority of these mechanism are modifications on two themes: direct synthesis of ATP or indirect synthesis of ATP with two basic mechanisms known respectively as Substrate Level Phosphorylation (SLP) and oxidative phosphorylation. These topics are substantive enough that they will be discussed in detail in the next few modules. Suffice it to say both mechanisms rely on biochemical reactions that transfer energy from some energy source to ADP or AMP, to synthesize ATP.
Glycolysis: an overview
Organisms, whether unicellular or multicellular, need to find ways of getting at least two key things from their environment: (1) matter or raw materials for maintaining a cell and building new cells and (2) energy to help with the work of staying alive and reproducing. Energy and the raw materials may come from different places. For instance, organisms that primarily harvest energy from sunlight will get raw materials for building biomolecules from sources like CO2. By contract, some organisms rely on redox reactions with small molecules and/or reduced metals for energy and get their raw materials for building biomolecules from compounds unconnected to the energy source. Meanwhile, some organisms (including ourselves), have evolved to get energy AND the raw materials for building and cellular maintenance from sometimes associated sources.
Glycolysis is the first metabolic pathway discussed in BIS2A - a metabolic pathway is a series of linked biochemical reactions. Because of its ubiquity in biology, it is hypothesized that glycolysis was probably one of the earliest metabolic pathways to evolve (more on this later). Glycolysis is a 10 step metabolic pathway that is centered on the processing of glucose for both energy extraction from chemical fuel and for the processing of the carbons in glucose into various other biomolecules (some of which are key precursors of many much more complicated biomolecules). Our study of glycolysis should and therefore will be examined using the precepts outlined in the energy challenge rubric that ask us to formally consider what happens to BOTH matter and energy in this multistep process.
The Energy Story and Design Challenge
Our investigation of glycolysis is a good opportunity to examine a biological process using both the energy story and the design challenge rubrics and perspectives.
The design challenge rubric will try to get you to think actively about both broadly and specifically about why we are studying this pathway - what is so important about it? What "problems" does the evolution of a glycolytic pathway allow life to solve or overcome? We will also want to think about alternate ways to solve the same problems and why they may or may not have evolved. Later we will examine a hypothesis for how this pathway - and other linked pathways - may have actually evolved and thinking about alternative strategies for satisfying various constraints will come in handy then.
In the context of the energy story, we will ask you to think about glycolysis as a process from which something can be learned by analyzing what happens to both matter and energy. That is, even though it is a 10 step biochemical pathway, we propose that some insight can be learned by carefully examining the process as a set of matter and energy inputs and outputs, a process with a beginning and an end.
So what is glycolysis? Let's start to find out.

| Enzyme | Step | ΔG/(kJ/mol) | ΔG°'/(kJ/mol) |
|---|---|---|---|
| Hexokinase | 1 | -34 | -16.7 |
| Phosphoglucose isomerase | 2 | -2.9 | 1.67 |
| Phosphofructokinase | 3 | -19 | -14.2 |
| Fructose-bisphosphate aldolase | 4 | -0.23 | 23.9 |
| Triose phosphate isomerase | 5 | 2.4 | 7.56 |
| Glyceraldehyde 3-phosphate dehydrogenase | 6 | -1.29 | 6.30 |
| Phosphoglycerate kinase | 7 | 0.09 | -18.9 |
| Phosphoglycerate mutase | 8 | 0.83 | 4.4 |
| Enolase | 9 | 1.1 | 1.8 |
| Pyruvate kinase | 10 | -23.0 | -31.7 |
| The measurements of the energy at standard state (ΔG°'/(kJ/mol)) compared with measurements taken from a living cell (ΔG/(kJ/mol)). Under conditions of constant temperature and pressure (ΔG°'/(kJ/mol)), reactions will occur in the direction that leads to a decrease in the value of the Gibbs free energy. Cellular measurements of ΔG can be dramatically different than ΔG°' measurements due to cellular conditions, such as concentrations of relevant metabolites etc. There are three large negative ΔG drops in the cell in the process of glycolysis. These reactions are considered irreversible and are often subject to regulation. | |||
Overall, the glycolytic pathway consists of 10 enzyme catalyzed steps. The primary input into this pathway is a single molecule of glucose, though we will discover that molecules may feed in and out of this pathway at various steps. We will focus our attention on (1) consequences of the overall process (2) several key reactions that highlight important types of biochemistry and biochemical principles we will want to carry forward to other contexts and (3) alternative fates of the intermediates and products of this pathway.
Note for reference that glycolysis is an anaerobic process, there is no requirement for molecular oxygen in glycolysis (oxygen gas is not a reactant in any of the chemical reactions in glycolysis). Glycolysis occurs in the cytosol or cytoplasm of cells. For a short (3 minute) overview YouTube video of glycolysis click here.
First half of glycolysis: Energy Investment Phase
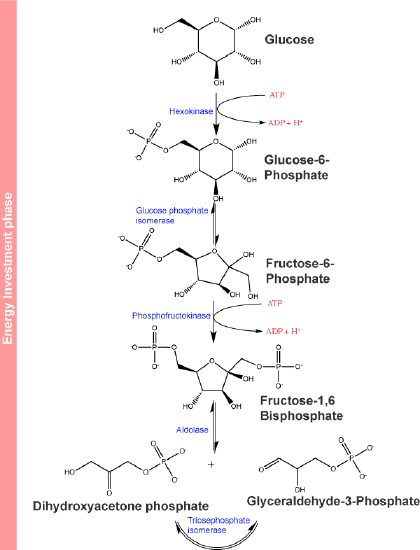
The first few steps of glycolysis are typically referred to as an "energy investment phase" of the pathway. This, however, doesn't make much intuitive sense (in the framework of a design challenge, it's not clear what problem this energy investment solves) if one only looks at glycolysis as an "energy producing" pathway and until these steps of glycolysis are put into a broader metabolic context. We'll try to build that story as we go, so for now just recall that we mentioned that some of the first steps are often associated with energy investment and ideas like "trapping" and "commitment" that are noted in the figure below.
Step 1 of glycolysis:
The first step in glycolysis shown below, is catalyzed by hexokinase (enzyme 1 in the figure below), an enzyme with broad specificity that catalyzes the phosphorylation of six-carbon sugars. Hexokinase catalyzes the phosphorylation of glucose, where glucose and ATP are substrates for the reaction, producing a molecule glucose-6-phosphate and ADP as products.
The first half of glycolysis is called the energy investment phase. In this phase, the cell expends two ATP into the reactions. Attribution: Marc T. Facciotti (original work)
Suggested discussion
The paragraph above states that the enzyme hexokinase has "broad specificity". This means that it can catalyze reactions with different sugars - not just glucose. From a molecular perspective, can you explain why this might be the case? Does this challenge your conception of enzyme specificity? If you Google the term "enzyme promiscuity" (don't worry it's safe for work) does this give you a broader appreciation for enzyme selectivity and activity?
The conversion of glucose to the negatively charged glucose-6-phosphate significantly reduces the likelihood that the phosphorylated glucose leaves the cell by diffusion across the hydrophobic interior of the plasma membrane. It also "marks" the glucose in a way that effectively tags it for several different possible fates (see figure below).

Note that this figure indicates that glucose-6-phosphate can, depending on cellular conditions, be directed to multiple fates. While it is a component of the glycolytic pathway it is not only involved in glycolysis but also in the storage of energy as glycogen (CYAN) and in the building of various other molecules like nucleotides (RED).
Source: Marc T. Facciotti (original work)
As the figure above indicates, glycolysis is but one possible fate for glucose-6-phosphate (G6P). Depending on cellular conditions, G6P may be diverted to the biosynthesis of glycogen (an form of energy storage) or it may be diverted into the pentose phosphate pathway for the biosynthesis of various biomolecules, including nucleotides. This means that G6P, while involved in the glycolytic pathway is not solely tagged for oxidation at this phase. Perhaps, showing the broader context that this molecule is involved in (in addition to the rationale that tagging glucose with a phosphate decreases the likelihood that it will leave the cell) helps to explain the seemingly contradictory (if you only consider glycolysis as an "energy producing" process) reason for transferring energy from ATP onto glucose if it is only to be oxidized later - that is, glucose is not only used by the cell for harvesting energy and several other metabolic pathways depend on the transfer of the phosphate group.
Step 2 of glycolysis:
In the second step of glycolysis, an isomerase catalyzes the conversion of glucose-6-phosphate into one of its isomers, fructose-6-phosphate. An isomerase is an enzyme that catalyzes the conversion of a molecule into one of its isomers.
Step 3 of glycolysis:
The third step of glycolysis is the phosphorylation of fructose-6-phosphate, catalyzed by the enzyme phosphofructokinase. A second ATP molecule donates a phosphate to fructose-6-phosphate, producing fructose-1,6-bisphosphate and ADP as products. In this pathway, phosphofructokinase is a rate-limiting enzyme and its activity is tightly regulated. It is allosterically activated by AMP when concentration of AMP are high and moderately allosterically inhibited by ATP at the same site. Citrate - a compound we'll discuss soon - also acts as a negative allosteric regulator of this enzyme. In this way, phosphofructokinase monitors or senses molecular indicators of the energy status of the cells and can in response act as a switch that turns on or off the flow of substrate through the rest of the metabolic pathway depending on whether there is “sufficient” ATP relative in the system. The conversion of fructose-6-phosphate into fructose 1,6-bisphosphate is sometimes referred to as a commitment step by the cell to the oxidation of the molecule in the rest of the glycolytic pathway by creating a substrate for and helping to energetically drive the next highly endergonic (under standard conditions) step of the pathway.
Suggested discussion
We discussed allosteric regulation of an enzyme in earlier modules but did so in a context where the enzyme was "alone". Now let's consider the enzyme in the context of an extended metabolic pathway(s). Can you now express why allosteric regulation is functionally important and how it can be used to regulate the flow of compounds through a pathway? Try to express yourself.
Step 4 of glycolysis:
In the fourth step in glycolysis an enzyme, Fructose-bisphosphate aldolase, cleaves 1,6-bisphosphate into two three-carbon isomers: dihydroxyacetone-phosphate and glyceraldehyde-3-phosphate.
Second Half: Energy Payoff Phase
If viewed in the absence of other metabolic pathways, glycolysis has thus far cost the cell two ATP molecules and produced two small, three-carbon sugar molecules: dihydroxyacetone-phosphate (DAP) and glyceraldehyde-3-phosphate (G3P). When viewed in a broader context this investment of energy to produce a variety of molecules that can be used in a variety of other pathways doesn't seem like such a bad investment.
Both DAP and G3P can proceed through the second half of glycolysis. We now examine these reactions.
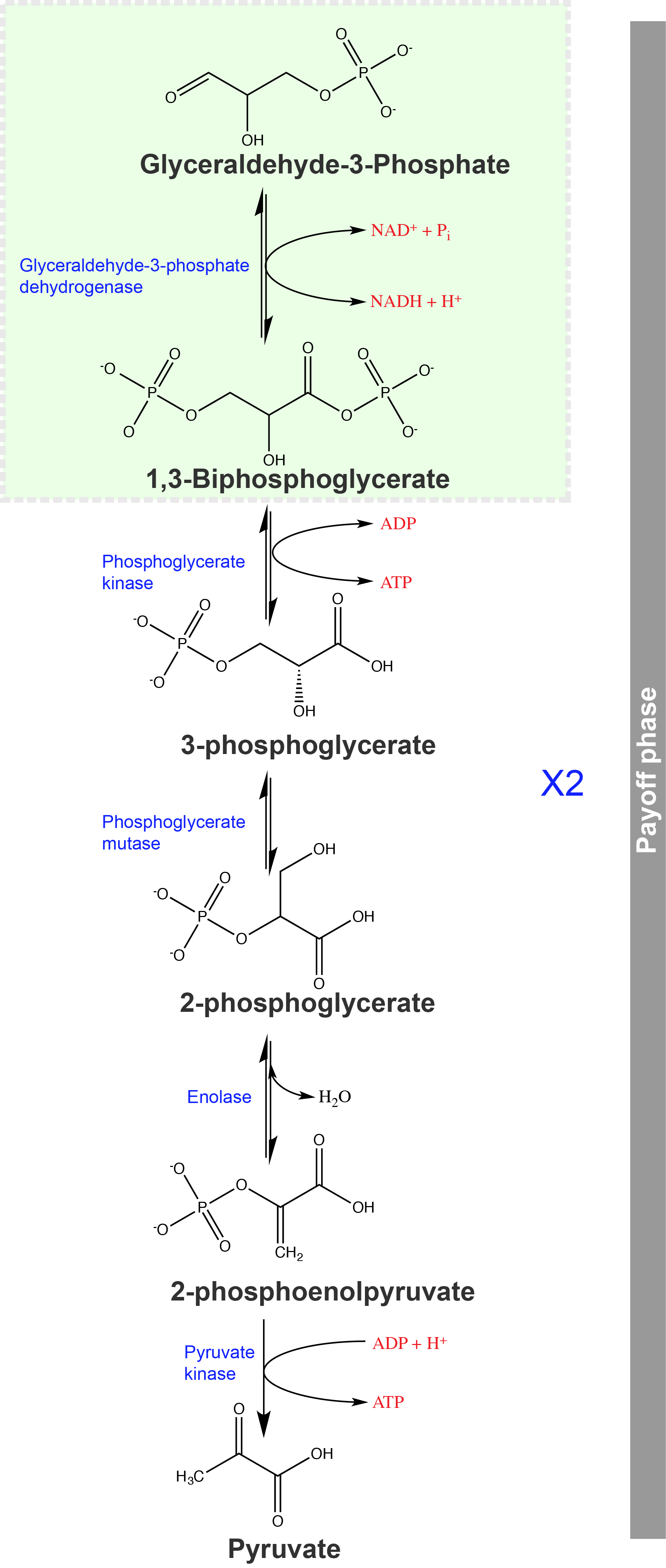
The second half of glycolysis is called the energy payoff phase. In this phase, the cell gains two ATP and 2 NADH compounds. At the end of this phase glucose has become partially oxidized to form pyruvate. Attribution: Marc T. Facciotti (original work).
Step 5 of glycolysis:
In the fifth step of glycolysis, an isomerase transforms the dihydroxyacetone-phosphate into its isomer, glyceraldehyde-3-phosphate. The 6 carbon glucose has therefore now been converted into two phosphorylated 3-carbon molecules of G3P.
Step 6 of glycolysis:
The sixth step is key and one from which can now leverage our understanding of the several types of chemical reactions that we've studied so far. If you're energy focused, this is finally a step of glycolysis where some of the reduced sugar is oxidized. The reaction is catalyzed by the enzyme glyceraldehyde-3-phosphate dehydrogenase. This enzyme catalyzes a multistep reaction between three substrates, glyceraldehyde-3-phosphate, the cofactor NAD+, and inorganic phosphate (Pi) and produces three products 1,3-bisphosphoglycerate, NADH and H+. One can think of this reaction as two reactions: (1) an oxidation/reduction and (2) a condensation reaction in which an inorganic phosphate is transferred onto a molecule. In this particular case, the redox reaction (a transfer of electrons off of G3P and onto NAD+ is exergonic and the phosphate transfer happens to be endergonic. The net standard free energy change hovers around zero - more on this later. The enzyme, here acts as a molecular coupling agent to couple the energetics of the exergonic reaction to that of the endergonic reaction thus driving both forward. This processes happens through a multi-step mechanism in the enzyme's active site and involving the chemical activity of a variety of functional groups.
It is important to note that this reaction depends upon the availability of the oxidized form of the electron carrier, NAD+. If we consider that there is a limiting pool of NAD+ we can then conclude that the reduced form of the carrier (NADH) must be continuously oxidized back into NAD+ in order to keep this step going. If NAD+ is not available, the second half of glycolysis slows down or stops.
Step 7 of glycolysis:
The seventh step of glycolysis, catalyzed by phosphoglycerate kinase (an enzyme named for the reverse reaction), 1,3-bisphosphoglycerate transfers a phosphate to ADP, forming one molecule of ATP and a molecule of 3-phosphoglycerate. This reaction is exergonic and is also an example of substrate-level phosphorylation.
Note: Possible discussion:
If a transfer of a phosphate from 1,3-BPG to ADP is exergonic, what does that say about the free energy of hydrolysis of the phosphate from 1,3-BPG as compared to the free energy of hydrolysis of the terminal phosphate on ATP?
Step 8 of glycolysis:
In the eighth step, the remaining phosphate group in 3-phosphoglycerate moves from the third carbon to the second carbon, producing 2-phosphoglycerate (an isomer of 3-phosphoglycerate). The enzyme catalyzing this step is a mutase (isomerase).
Step 9 of glycolysis:
Enolase catalyzes the ninth step. This enzyme causes 2-phosphoglycerate to lose water from its structure; this is a dehydration reaction, resulting in the formation of a double bond that increases the potential energy in the remaining phosphate bond and produces phosphoenolpyruvate (PEP).
Step 10 of glycolysis:
The last step in glycolysis is catalyzed by the enzyme pyruvate kinase (the enzyme in this case is named for the reverse reaction of pyruvate’s conversion into PEP) and results in the production of a second ATP molecule by substrate-level phosphorylation and the compound pyruvic acid (or its salt form, pyruvate). Many enzymes in enzymatic pathways are named for the reverse reactions, since the enzyme can catalyze both forward and reverse reactions (these may have been described initially by the reverse reaction that takes place in vitro, under non-physiological conditions).
Outcomes of Glycolysis
A couple of things to consider:
One of the clear outcomes of glycolysis is the biosynthesis of compounds that can enter into a variety of metabolic pathways. Likewise compounds coming from other metabolic pathways can feed into glycolysis at various points. So, this pathway can be part of a central exchange for carbon flux within the cell.
If glycolysis is run long enough, the constant oxidation of glucose with NAD+ can leave the cell with a problem; how to regenerate NAD+ from the 2 molecules of NADH produced. If the NAD+ is not regenerated all of the cell's NAD will be nearly completely transformed into NADH. So how do cells regenerate NAD+
Pyruvate is not completely oxidized, there is still some energy to be extracted - how might this happen? Also, what should the cell do with all of that NADH? Is there any energy there to extract?
Note: Strongly suggested discussion/exercise:
Can you write an energy story for the overall process of glycolysis. For energy terms just worry about describing things in terms of whether they are exergonic or endergonic. When I say overall process I mean overall process: glucose should be listed in the reactants and pyruvate listed on the product side of the arrow.
Substrate Level Phosphorylation (SLP)
The simplest rout to synthesize ATP is substrate level phosphorylation. ATP molecules are generated (that is, regenerated from ADP) as a direct result of a chemical reaction that occurs in catabolic pathways. A phosphate group is removed from an intermediate reactant in the pathway, and the free energy of the reaction is used to add the third phosphate to an available ADP molecule, producing ATP. This very direct method of phosphorylation is called substrate-level phosphorylation. It can be found in a variety of catabolic reactions, most notably in two specific reactions in glycolysis (which we will discuss specifically later). Suffice it to say what is required is a high energy intermediate whose oxidation is sufficient to drive the synthesis of ATP.
In this reaction the reactants are a phosphorylated carbon compound called G3P (from reaction 6 of glycolysis), an ADP molecule and the products are 1,3-BPG and ATP. The transfer of the phosphate from G3P to ADP to form ATP in the active site of the enzyme is substrate level phosphorylation. This occurs twice in glycolysis and once in the TCA cycle (for a subsequent reading).