3.6: APPENDIX D- Abl inhibitors
- Page ID
- 26476
A great recent review2
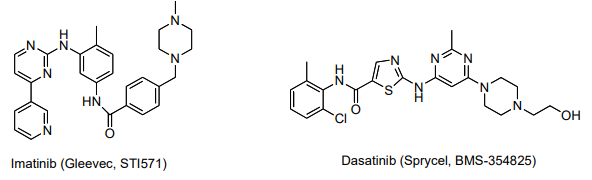
Imatinib (Gleevec) is an inhibitor of Bcr-Abl that was FDA approved in 2001 as a firstline treatment for CML.3 Gleevec resistance in CML patients has been linked to a number of point mutations resulting in amino acid substitutions in the Bcr-Abl protein.
Dasatinib was granted accelerated approval by the FDA in 2006 for treating patients with Gleevec-resistant forms of CML. Dasatinib has been shown to inhibit 14 or 15 Gleevecresistant mutants of Bcr-Abl. It does not inhibit the T315I mutant.4 ,5
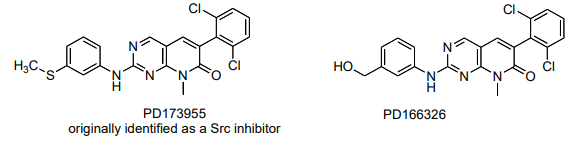
These compounds were studied as potential CML drugs thought to target Src kinase. Instead they appear to inhibit Bcr-Abl activity. PD173955 and five other related compounds were studied for inhibition of Bcr-Abl-dependent cell growth.6 A similar compound (PD180970) was found to inhibit several relevant Bcr-Abl isoforms, but NOT the T315I mutation. 7
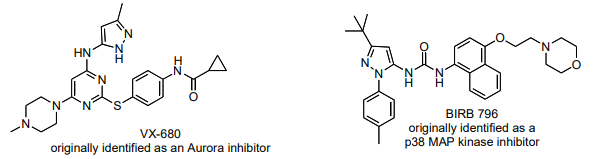
These compounds have been identified as inhibitors of Bcr-Abl mutants (namely the T315I mutant) that are resistant to Gleevec, dasatinib, and PD180970.8 VX-680 is currently in clinical trials.9 In session 15, you will evaluate a crystal structure of VX-680 bound to the H396P mutant of the Bcr-Abl kinase domain.10 Abl autophosphorylation reference11 Abl kinase activity assay (coupled to NADH oxidation) references12, 13


