3.2: The Greenhouse Effect
- Page ID
- 36543
The process by which the atmosphere absorbs the sun’s energy and prevents it from being radiated back out to space has often been compared to that of a greenhouse, leading to the nickname the greenhouse effect. It is the same process that occurs when you leave your car sitting in the sun with the windows rolled up. The sun’s rays are powerful enough to pass through the glass of the windows (or through the Earth’s atmosphere) and be absorbed by the dashboard and seats (or the Earth’s surface); however, when these surfaces emit energy, it is not powerful enough to pass back out through the window glass (or atmosphere) and in so doing becomes trapped within the car (or atmosphere) causing it to warm. There are certain atmospheric gases, termed greenhouses gases (or GHGs) that behave like the car windows, increasing the amount of energy retained in the atmosphere, and increasing the amount of warming that occurs. In Figure 3.1.1 the greenhouse effect is represented by the curved arrow showing 95% of the energy emitted by the Earth’s surface that is reabsorbed, and the amount of GHGs in the atmosphere drive the size of this arrow.
The primary GHGs considered in this section are carbon dioxide (CO2), methane (CH3), and nitrous oxide (N2O). Scientists at the National Oceanic and Atmospheric Administration (NOAA) have been tracking concentrations of these and other GHGs in the atmosphere for decades (Fig 3.2.1) and have found that all three continue to steadily increase.

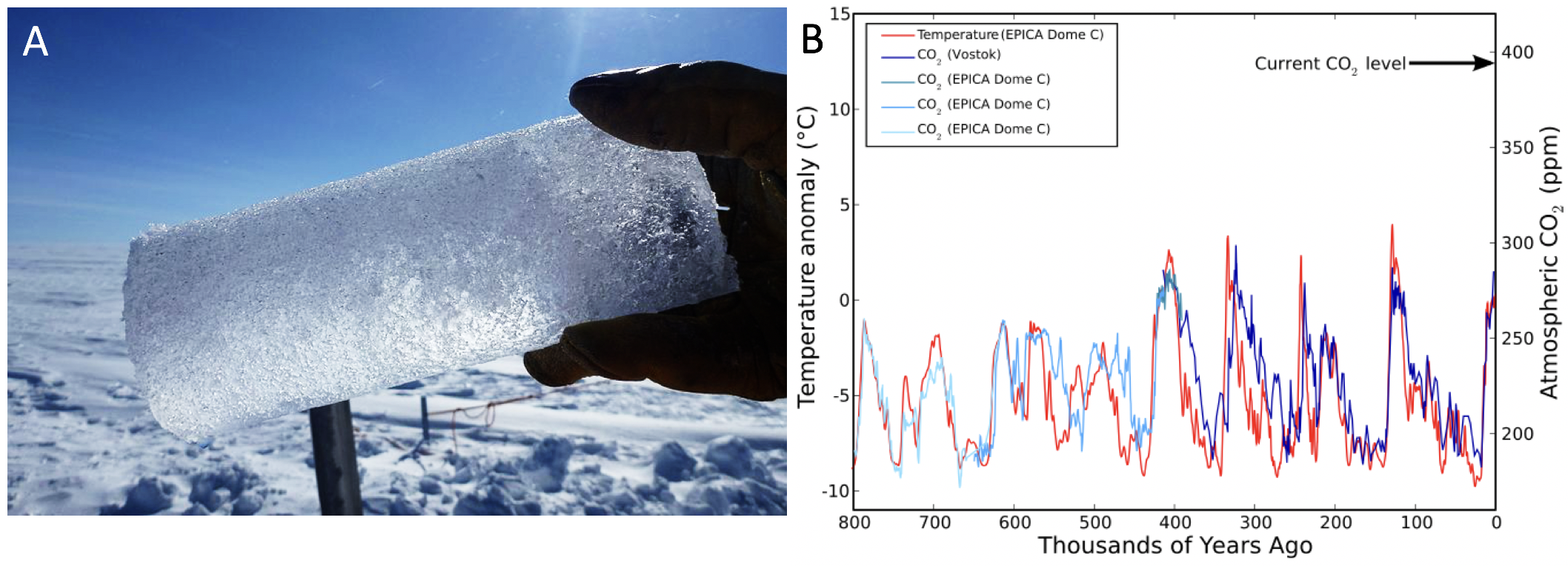
These increases are particularly pronounced when compared to past GHG concentrations. Scientists can measure past atmospheric composition in ice sheets in Greenland and Antarctica. As the ice sheets formed, small bubbles of air were trapped in the ice, and coring deep into the ice sheet (Fig 3.2.2a) allows scientists to reconstruct atmospheric composition from direct measurements as far back as 800,000 years (Fig 3.2.2b). From these measurements, we can see that atmospheric CO2 levels (and temperature, as estimated from oxygen isotope composition of the ice itself) have fluctuated significantly throughout the past. The ice sheets document a clear pattern of periodic increases and decreases in CO2, which are coupled with increases and decreases in temperature. Periods of low CO2 and low temperatures are glacial periods (also referred to as ‘ice ages’), and periods of high CO2 and high temperature are inter-glacial periods. For the past 800,000 years, Earth has oscillated between glacial and inter-glacial periods roughly every 100,000 years. The maximum level that CO2 concentrations reached in the last 800,000 years was approximately 300 parts per million (ppm). Global CO2 levels are now over 400 ppm, a level that scientists estimate has not occurred on Earth since the Pliocene Epoch, approximately 3 million years ago.
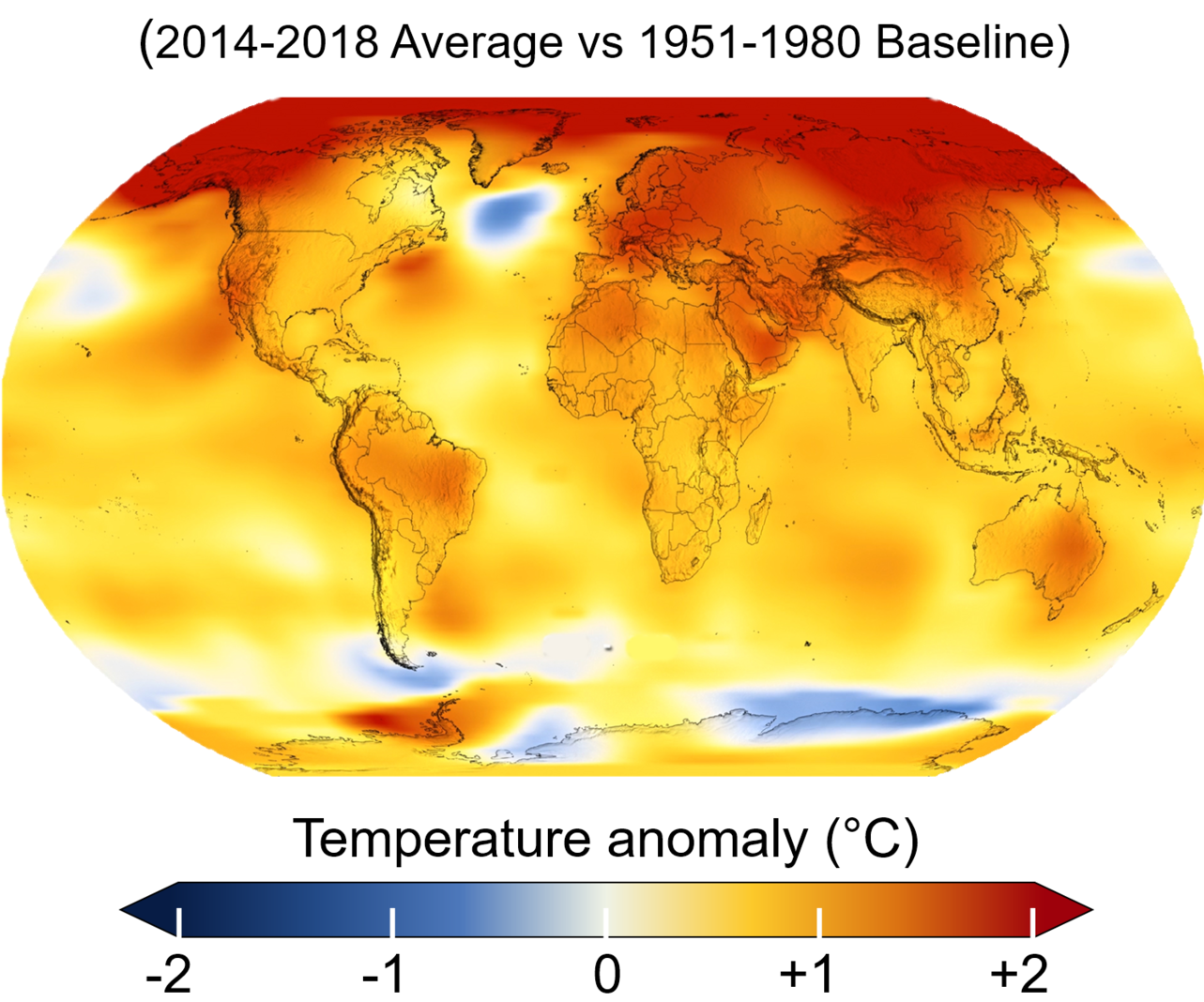
This increase in CO2 (as well as other GHGs) increases the amount of solar energy that is retained in the atmosphere as opposed to radiated back out to space, which increases the temperature of the Earth’s surface. Figure 3.2.3 shows the temperature anomaly for the decade 2014-2018 as compared to the average from 1951-1980. The National Aeronautics and Space Administration (NASA) reports that the global average temperature has increased approximately 1.4° Fahrenheit (0.8° C) since 1880, though that warming has not been evenly distributed across the Earth. As can be seen in Figure 3.2.3, the polar regions, particularly the Arctic, have warmed much more than other areas. This pattern is particularly concerning given the feedbacks that warming in polar regions may have on the melting of ice sheets and sea ice.
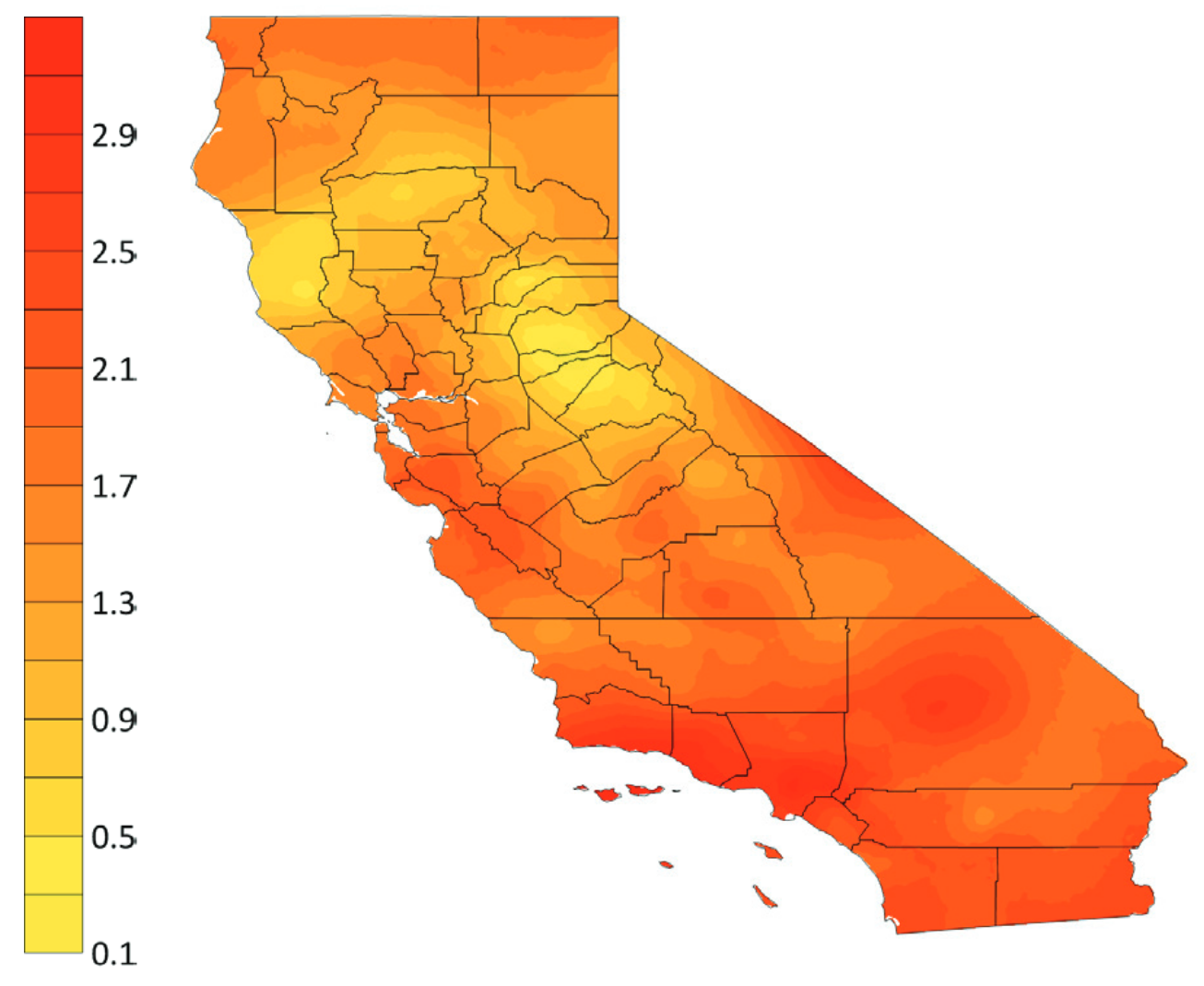
In 2018, the state of California released its Fourth Climate Change Assessment report, which outlines the impact that global climatic changes are having and will have on the state (Fig 3.2.4). Some regions, most notably southern California, have already experienced nearly 3°F increases in annual average temperatures since the beginning of the 20th century.