A Hypothesis for How ETCs May Have Evolved
A proposed link between SLP/fermentation and the evolution of ETCs:
In a previous discussion of energy metabolism, we explored substrate level phosphorylation (SLP) and fermentation reactions. While SLP and fermentation together are perfectly good ways to harvest energy, one byproduct of these reactions is the acidification of the cell. Early cells that used these modes of energy harvested therefore needed to co-evolve mechanisms that helped remove protons accumulated from SLP and fermentation from the cytosol (interior of the cell). One solution to the "proton problem" may have been the evolution of the F0F1-ATPase, a multi-subunit enzyme that translocated protons from the inside of the cell to the outside of the cell by hydrolyzing ATP (see the figure below). This arrangement works as long as small reduced organic molecules are abundant and freely available to generate ATP can through SLP that can "fuel" the business of the cell. However, as these biological processes continue, the small reduced organic molecules will become depleted. The resulting scarcity of fuel, therefore, puts a demand on cells to find alternative mechanisms to harness energy and/or to become more efficient.
In the scheme proposed above, one potential source of "wasted ATP" is its use in the removal of protons from the cell's cytosol; organisms that could find other mechanisms to expel accumulating protons while still preserving ATP could have a selective advantage. We hypothesize that this selective evolutionary pressure potentially led to the evolution of the first membrane-bound proteins that used red/ox reactions as their energy source (depicted in a second picture) to pump out the accumulating protons. Enzymes and enzyme complexes with these properties exist today in the as electron transport complexes like Complex I, the NADH dehydrogenase.

Figure 1. Proposed evolution of an ATP dependent proton translocator

Figure 2. As small reduced organic molecules become limited, organisms that can find alternative mechanisms to remove protons from the cytosol may have an a selective dvantage. The evolution of a proton translocator that uses red/ox reactions rather than ATP hydrolysis could substitute for the ATPase.
Continuing with this line of logic, if organisms evolved that could now use red/ox reactions to translocate protons across the membrane they would create an electrochemical gradient, separating both charge (positive on the outside and negative on the inside, creating an electrical potential) and pH (low pH outside, higher pH inside). With excess protons on the outside of the cell membrane, and the F0F1-ATPase no longer consuming ATP to translocate protons, we hypothesize that the electrochemical gradient could then power the F0F1-ATPase "backwards" — that is, to form or produce ATP by using the energy in the charge/pH gradients set up by the red/ox pumps (as depicted below). We call this arrangement an electron transport chain (ETC).

Figure 3. The evolution of the ETC; the combination of the red/ox driven proton translocators coupled to the production of ATP by the F0F1-ATPase.
NoTE: Extended reading on the evolution of electron transport chains
If you're interested in the the evolution of electron transport chains, check out this more in-depth discussion of the topic at NCBI.
Possible NB Discussion  Point
Point
Dinitrophenol (DNP) is a small chemical that serves to uncouple the flow of protons across the inner mitochondrial membrane to the ATP synthase, making the membrane leaky to protons. People used it until 1938 as a weight-loss drug. What effect would you expect DNP to have on the difference in pH across both sides of the inner mitochondrial membrane? Why do you think this might be an effective weight-loss drug? Why might it be dangerous? Can you think of any scenarios where it is non-harmful, or even beneficial, to uncouple proton flow with ATP synthase?
Light Energy and Pigments
Light Energy
The sun emits an enormous amount of electromagnetic radiation (solar energy) that spans a broad swath of the electromagnetic spectrum, the range of all radiation frequencies. When solar radiation reaches Earth, a fraction of this energy interacts with and may transfer to the matter on the planet. This energy transfer results in a wide variety of different phenomena, from influencing weather patterns to driving myriad biological processes. In BIS2A, we are largely concerned with the latter, and below, we discuss some very basic concepts related to light and its interaction with biology.
First, however we need to refresh a few key properties of light:
- Light in a vacuum travels at a constant speed of 299,792,458 m/s. We often abbreviate the speed of light with the variable "c".
- Light has properties of waves. A specific "color" of light has a characteristic wavelength.
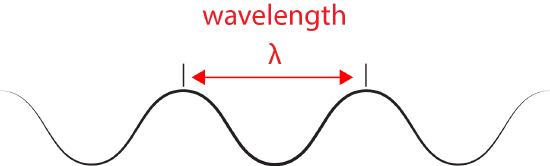
We refer to the distance between peaks in a wave as the wavelength and abbreviate it with the Greek letter lambda (Ⲗ). Attribution: Marc T. Facciotti (original work)
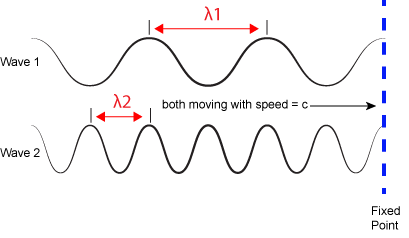
The inverse proportionality of frequency and wavelength. Wave 1 has a wavelength that is 2x that of wave 2 (Ⲗ1 > Ⲗ2). If the two waves are traveling at the same speed (c)—imagine that both of the whole lines that are dragged past the fixed vertical line at the same speed—then the number of times a wave peak passes a fixed point is greater for wave 2 than wave 1 (f2 > f1). Attribution: Marc T. Facciotti (original work)
3. Finally, each frequency (or wavelength) of light is associated with a specific energy. We'll call energy "E". The relationship between frequency and energy is:
\[E = h \times f\]
where h is a constant called the Planck constant (~6.626x10-34 Joule•second when frequency is expressed in cycles per second). Given the relationship between frequency and wavelength, you can also write E = h*c/Ⲗ. Therefore, the larger the frequency (or shorter the wavelength), the more energy is associated with a specific "color". Wave 2 in the figure above is associated with greater energy than wave 1.
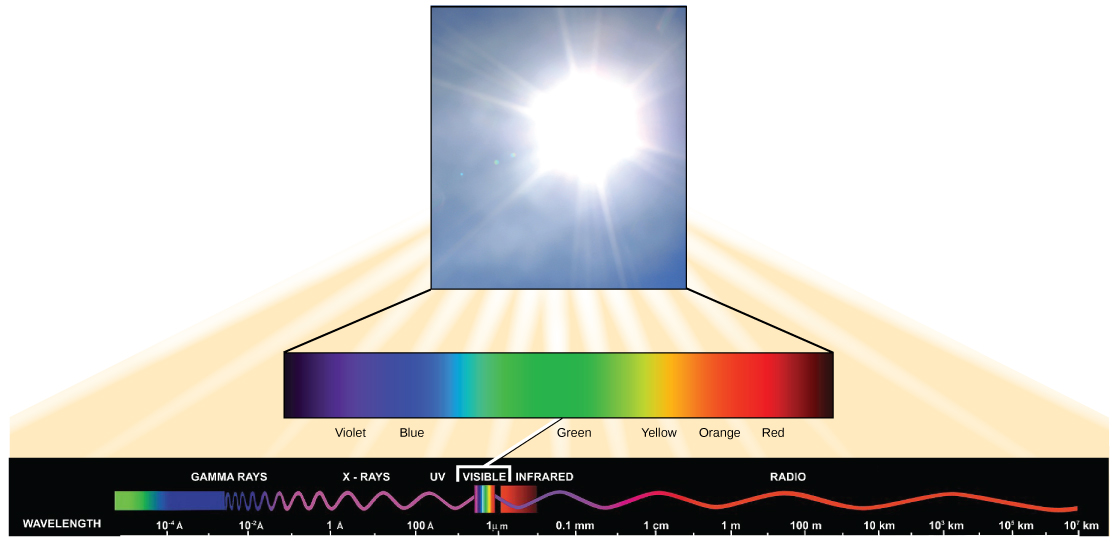
The sun emits energy in the form of electromagnetic radiation. All electromagnetic radiation, including visible light, is characterized by its wavelength. The longer the wavelength, the less energy it carries. The shorter the wavelength, the more energy is associated with that band of the electromagnetic spectrum.
The Light We See
The visible light seen by humans as white light is composed of a rainbow of colors, each with a characteristic wavelength. Certain objects, such as a prism or a drop of water, disperse white light to reveal the colors to the human eye. In the visible spectrum, violet and blue light have shorter (higher energy) wavelengths while the orange and red light have a longer (lower energy) wavelengths.
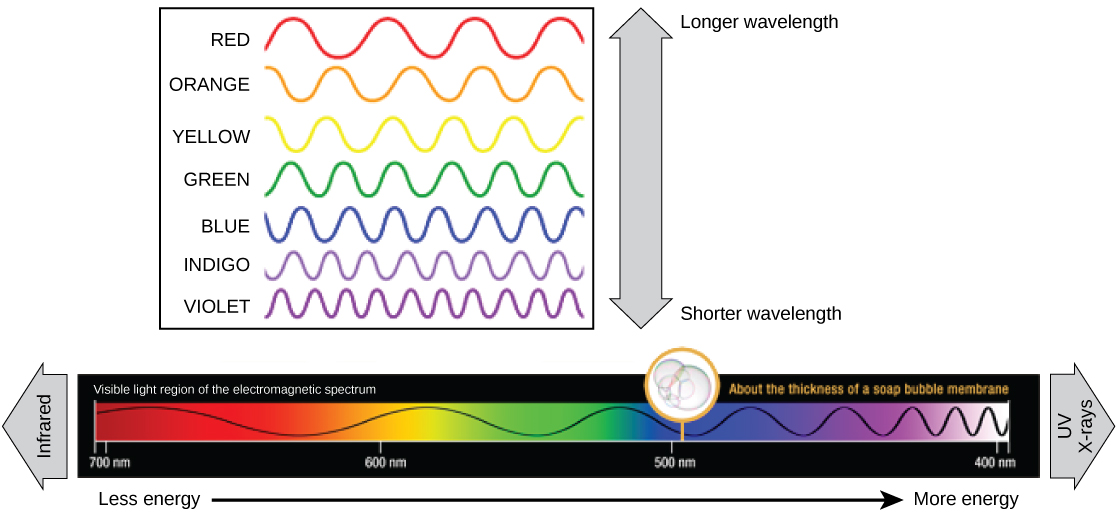
The colors of visible light do not carry the same amount of energy. Violet has the shortest wavelength and therefore carries the most energy, whereas red has the longest wavelength and carries the least amount of energy. Credit: modification of work by NASA
Absorption by Pigments
The interaction between light and biological systems occurs through several mechanisms, some of which you may learn about in upper division courses in cellular physiology or biophysical chemistry. In BIS2A, we concern ourselves mostly with the interaction between light and biological pigments. These interactions can start a variety of light-dependent biological processes that can be grossly grouped into two functional categories: cellular signaling and energy harvesting. Signaling molecules perceive changes in the environment (in this case, changes in light). An example of a signaling interaction might be the interaction between light and the pigments expressed in an eye. Light/pigment interactions that are involved in energy harvesting are used for—not surprisingly—capturing the energy in the light and transferring it to the cell to fuel biological processes. Photophosphorylation, which we will learn more about soon, is one example of an energy harvesting interaction.
Possible NB Discussion  Point
Point
Photophosphorylation is a process involving an electron transport chain that allows organisms to harvest energy from light. Some of you may already be familiar with this process. Many of you are learning about this for the first time. Given your current knowledge base, offer your best explanation or hypothesis as to how light interacts with the ETC. You will have a chance to revisit this topic very soon.
At the center of the biological interactions with light are groups of molecules we call organic pigments. Whether in the human retina, chloroplast thylakoid, or microbial membrane, organic pigments often have specific ranges of energy or wavelengths that they can absorb. The sensitivity of these molecules for different wavelengths of light is due to their unique chemical makeups and structures. A range of the electromagnetic spectrum is given a couple of special names because of the sensitivity of some key biological pigments: The retinal pigment in our eyes, when coupled with an opsin sensor protein, “sees” (absorbs) light predominantly between the wavelengths between of 700 nm and 400 nm. Because this range defines the physical limits of the electromagnetic spectrum that we can actually see with our eyes, we refer to this wavelength range as the “visible range”. For similar reasons, as plants pigment molecules tend to absorb wavelengths of light mostly between 700 nm and 400 nm, plant physiologists refer to this range of wavelengths as "photosynthetically active radiation".
Three Key Types of Pigments We Discuss in BIS2A
Chlorophylls
Chlorophylls (including bacteriochlorophylls) are part of a large family of pigment molecules. There are five major chlorophyll pigments named: a, b, c, d, and f. Chlorophyll a is related to a class of more ancient molecules found in bacteria called bacteriochlorophylls. Chlorophylls are structurally characterized by ring-like porphyrin group that coordinates a metal ion. This ring structure is chemically related to the structure of heme compounds that also coordinate a metal and are involved in oxygen binding and/or transport in many organisms. We distinguish different chlorophylls from one another by different "decorations"/chemical groups on the porphyrin ring.
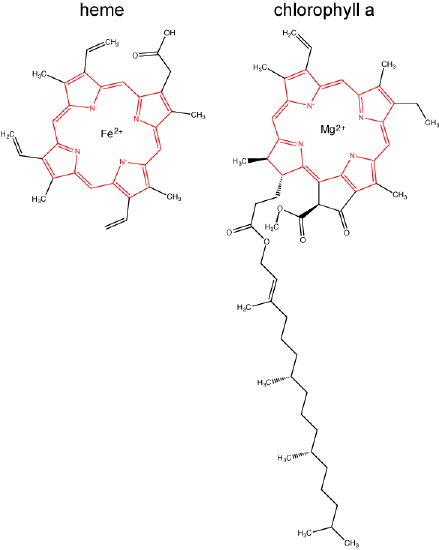
The structure of heme and chlorophyll a molecules. The common porphyrin ring is colored in red. Attribution: Marc T. Facciotti (original work)
Carotenoids
Carotenoids are the red/orange/yellow pigments found in nature. They are found in fruit—the red of tomato (lycopene), the yellow of corn seeds (zeaxanthin), or the orange of an orange peel (β-carotene)—which serve as biological "advertisements" to attract seed dispersers (animals or insects that may carry seeds elsewhere). In photosynthesis, carotenoids function as photosynthetic pigments. In addition, when a leaf is exposed to full sun, that surface is required to process an enormous amount of energy; If that energy is not managed properly, it can do significant damage. Therefore, many carotenoids help absorb excess energy in light and safely dissipate that energy as heat.
Flavonoids
Flavonoids are a very broad class of compounds that are found in great diversity in plants. These molecules come in many forms but all share a common core structure shown below. The diversity of flavonoids comes from the many combinations of functional groups that can "decorate" the core flavone.
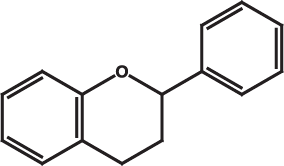
The core ring structure of flavans.
Each type of pigment can be identified by the specific pattern of wavelengths it absorbs from visible light. We define this characteristic as the pigment's absorption spectrum. The graph in the figure below shows the absorption spectra for chlorophyll a, chlorophyll b, and a type of carotenoid pigment called β-carotene (which absorbs blue and green light). Notice how each pigment has a distinct set of peaks and troughs, revealing a highly specific pattern of absorption. These differences in absorbance are because of differences in chemical structure (some of these are highlighted in the figure). Chlorophyll a absorbs wavelengths from either end of the visible spectrum (blue and red), but not green. Because chlorophyll reflects green light and absorbs other wavelengths of light, things containing this pigment appear green. Carotenoids absorb in the short-wavelength blue region and reflect the longer yellow, red, and orange wavelengths.
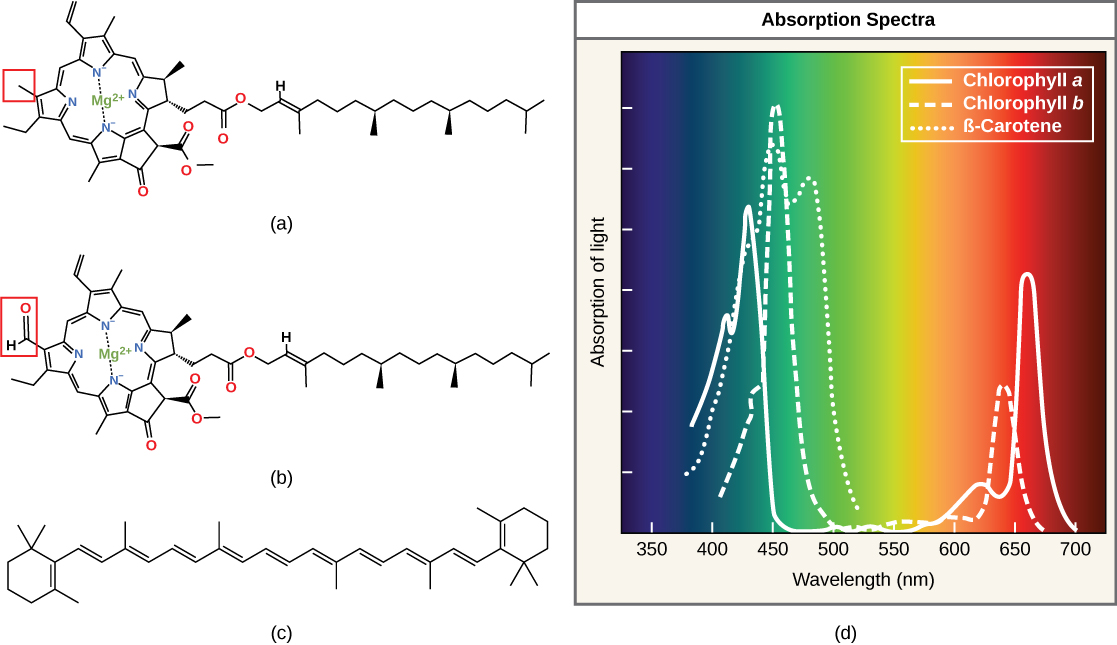
(a) Chlorophyll a, (b) chlorophyll b, and (c) β-carotene are hydrophobic organic pigments found in the thylakoid membrane. Chlorophyll a and b, which are identical except for the part indicated in the red box, are responsible for the green color of leaves. Note how the small amount of difference in chemical composition between different chlorophylls leads to different absorption spectra. β-carotene is responsible for the orange color in carrots. Each pigment has a unique absorbance spectrum (d).
Importance of having multiple different pigments
Not all photosynthetic organisms have full access to sunlight. Some organisms grow underwater where light intensity and the number of wavelengths decrease and change, respectively, with depth. Other organisms grow in competition for light. For instance, plants on the rainforest floor must be able to absorb any bit of light that comes through because the taller trees absorb most of the sunlight and scatter the remaining solar radiation. To account for these variable light conditions, many photosynthetic organisms have a mixture of pigments whose expression they can tune to improve the organism's ability to absorb energy from a wider range of wavelengths than would be possible with one pigment alone.
 Point
Point
 Point
Point









