S2019_Lecture_10_Reading
- Page ID
- 21551
The Endomembrane System
The endomembrane system (endo = “within”) is a group of membranes and organelles in eukaryotic cells that works together to modify, package, and transport lipids and proteins. It includes the nuclear envelope, lysosomes, and vesicles, which we’ve already mentioned, and the endoplasmic reticulum and Golgi apparatus, which we will cover shortly. Although not technically within the cell, the plasma membrane is included in the endomembrane system because, as you will see, it interacts with the other endomembranous organelles. The endomembrane system does not include the membranes of either mitochondria or chloroplasts.
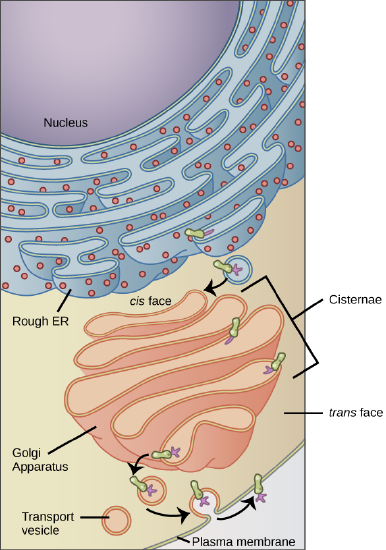
Membrane and secretory proteins are synthesized in the rough endoplasmic reticulum (RER). The RER also sometimes modifies proteins. In this illustration, a (green) integral membrane protein in the ER is modified by attachment of a (purple) carbohydrate. Vesicles with the integral protein bud from the ER and fuse with the cis face of the Golgi apparatus. As the protein passes along the Golgi’s cisternae, it is further modified by the addition of more carbohydrates. After its synthesis is complete, it exits as integral membrane protein of the vesicle that bud from the Golgi’s trans face and when the vesicle fuses with the cell membrane the protein becomes integral portion of that cell membrane. (credit: modification of work by Magnus Manske)
Possible discussion
If a peripheral membrane protein were synthesized in the lumen (inside) of the ER, would it end up on the inside or outside of the plasma membrane?
The Endoplasmic Reticulum
The endoplasmic reticulum (ER) (see figure above) is a series of interconnected membranous sacs and tubules that collectively modifies proteins and synthesizes lipids. However, these two functions are performed in separate areas of the ER: the rough ER and the smooth ER, respectively.
The hollow portion of the ER tubules is called the lumen or cisternal space. The membrane of the ER, which is a phospholipid bilayer embedded with proteins, is continuous with the nuclear envelope.
Rough ER
The rough endoplasmic reticulum (RER) is so named because the ribosomes attached to its cytoplasmic surface give it a studded appearance when viewed through an electron microscope (see figure below).
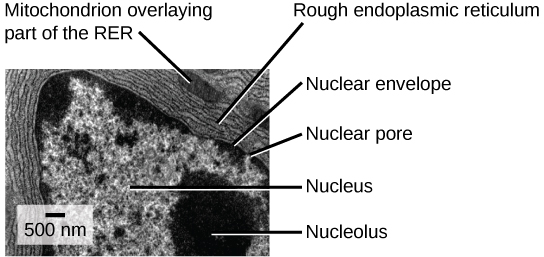
This transmission electron micrograph shows the rough endoplasmic reticulum and other organelles in a pancreatic cell. (credit: modification of work by Louisa Howard)
Ribosomes transfer their newly synthesized proteins into the lumen of the RER where they undergo structural modifications, such as folding or the acquisition of side chains. These modified proteins will be incorporated into cellular membranes—the membrane of the ER or those of other organelles—or secreted from the cell (such as protein hormones, enzymes). The RER also makes phospholipids for cellular membranes.
If the phospholipids or modified proteins are not destined to stay in the RER, they will reach their destinations via transport vesicles that bud from the RER’s membrane.
Since the RER is engaged in modifying proteins (such as enzymes, for example) that will be secreted from the cell, you would be correct in assuming that the RER is abundant in cells that secrete proteins. This is the case with cells of the liver, for example.
Smooth ER
The smooth endoplasmic reticulum (SER) is continuous with the RER but has few or no ribosomes on its cytoplasmic surface. Functions of the SER include synthesis of carbohydrates, lipids, and steroid hormones; detoxification of medications and poisons; and storage of calcium ions.
In muscle cells, a specialized SER called the sarcoplasmic reticulum is responsible for storage of the calcium ions that are needed to trigger the coordinated contractions of the muscle cells.
The Golgi Apparatus
We have already mentioned that vesicles can bud from the ER and transport their contents elsewhere, but where do the vesicles go? Before reaching their final destination, the lipids or proteins within the transport vesicles still need to be sorted, packaged, and tagged so that they wind up in the right place. Sorting, tagging, packaging, and distribution of lipids and proteins takes place in the Golgi apparatus (also called the Golgi body), a series of flattened membranes (see figure below).
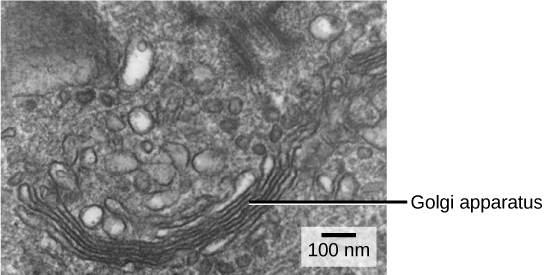
The Golgi apparatus in this white blood cell is visible as a stack of semicircular, flattened rings in the lower portion of the image. Several vesicles can be seen near the Golgi apparatus. (credit: modification of work by Louisa Howard)
The receiving side of the Golgi apparatus is called the cis face. The opposite side is called the trans face. The transport vesicles that formed from the ER travel to the cis face, fuse with it, and empty their contents into the lumen of the Golgi apparatus. As the proteins and lipids travel through the Golgi, they undergo further modifications that allow them to be sorted. The most frequent modification is the addition of short chains of sugar molecules. These newly modified proteins and lipids are then tagged with phosphate groups or other small molecules so that they can be routed to their proper destinations.
Finally, the modified and tagged proteins are packaged into secretory vesicles that bud from the trans face of the Golgi. While some of these vesicles deposit their contents into other parts of the cell where they will be used, other secretory vesicles fuse with the plasma membrane and release their contents outside the cell.
In another example of form following function, cells that engage in a great deal of secretory activity (such as cells of the salivary glands that secrete digestive enzymes or cells of the immune system that secrete antibodies) have an abundance of Golgi.
In plant cells, the Golgi apparatus has the additional role of synthesizing polysaccharides, some of which are incorporated into the cell wall and some of which are used in other parts of the cell.
Lysosomes
In addition to their role as the digestive component and organelle-recycling facility of animal cells, lysosomes are considered to be parts of the endomembrane system. Lysosomes also use their hydrolytic enzymes to destroy pathogens (disease-causing organisms) that might enter the cell. A good example of this occurs in a group of white blood cells called macrophages, which are part of your body’s immune system. In a process known as phagocytosis or endocytosis, a section of the plasma membrane of the macrophage invaginates (folds in) and engulfs a pathogen. The invaginated section, with the pathogen inside, then pinches itself off from the plasma membrane and becomes a vesicle. The vesicle fuses with a lysosome. The lysosome’s hydrolytic enzymes then destroy the pathogen (figure below).
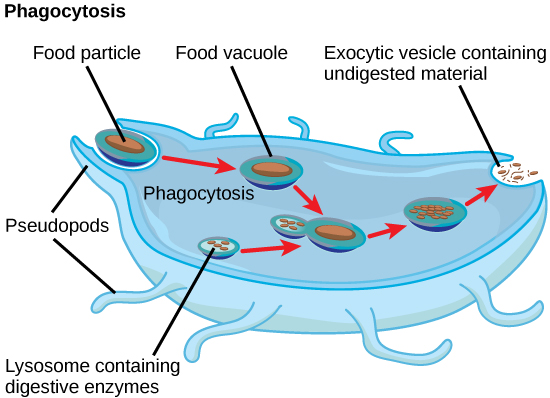
A macrophage has engulfed (phagocytized) a potentially pathogenic bacterium and then fuses with a lysosomes within the cell to destroy the pathogen. Other organelles are present in the cell but for simplicity are not shown.
Summary of Endomembranes
The endomembrane system includes the nuclear envelope, lysosomes, vesicles, the ER, and Golgi apparatus, as well as the plasma membrane. These cellular components work together to modify, package, tag, and transport proteins and lipids that form the membranes.
The RER modifies proteins and synthesizes phospholipids used in cell membranes. The SER synthesizes carbohydrates, lipids, and steroid hormones; engages in the detoxification of medications and poisons; and stores calcium ions. Sorting, tagging, packaging, and distribution of lipids and proteins take place in the Golgi apparatus. Lysosomes are created by the budding of the membranes of the RER and Golgi. Lysosomes digest macromolecules, recycle worn-out organelles, and destroy pathogens.
Free Response
Exercise 1
In the context of cell biology, what do we mean by form follows function? What are at least two examples of this concept?
“Form follows function” refers to the idea that the function of a body part dictates the form of that body part. As an example, compare your arm to a bat’s wing. While the bones of the two correspond, the parts serve different functions in each organism and their forms have adapted to follow that function.
Exercise 2
In your opinion, is the nuclear membrane part of the endomembrane system? Why or why not? Defend your answer.
Since the external surface of the nuclear membrane is continuous with the rough endoplasmic reticulum, which is part of the endomembrane system, then it is correct to say that it is part of the system.
The Cytoskeleton
The cytoskeleton is a network of different protein fibers that provides many functions: it maintains or changes the shape of the cell; it secures some organelles in specific positions; it enables movement of cytoplasm and vesicles within the cell; and it enables the cell to move in response to stimuli. There are three types of fibers within the cytoskeleton: microfilaments, intermediate filaments, and microtubules. Some of the cytoskeletal fibers work in conjunction with molecular motors which move along the fibers within the cell to carry out a diverse set of functions. There are two main families of cytoskeletally-associated molecular motors: dyneines and kinesins.
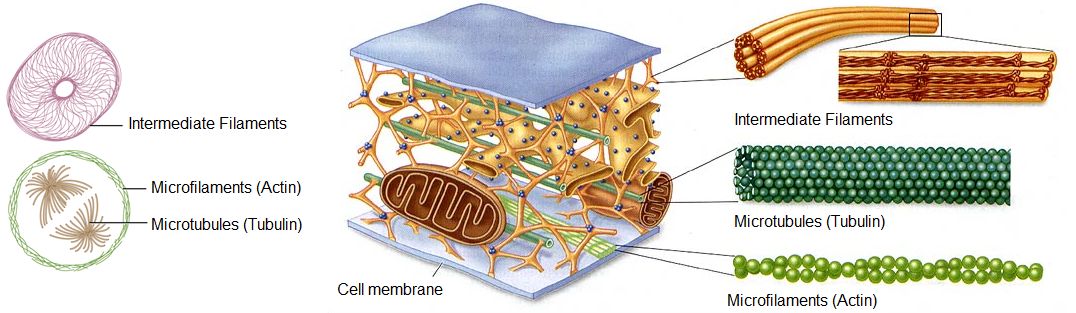
Figure 1. Microfilaments thicken the cortex around the inner edge of a cell; like rubber bands, they resist tension. Microtubules are found in the interior of the cell where they maintain cell shape by resisting compressive forces. Intermediate filaments are found throughout the cell and hold organelles in place.
Design challenge
Problem statement: Eukaryotic cells contain membrane-bound organelles that effectively separate materials, processes, and reactions from one another and from the cytoplasm. This in itself poses a problem for eukaryotes.
How can the cell purposely move and control the location of materials between these organelles? More specifically, how can a eukaryotic cell transport compounds from their place of origin (in most cases the cyotoplasm) to where they are needed (perhaps the nucleus, the mitochondria, or the cell surface)?
Note: possible discussion
Propose some reasons why cells—particularly large cells and/or cells with organelles—cannot rely on simple diffusion to move metabolites, building blocks, proteins, etc. to the locations in the cell where they are needed.
One possible solution is for the cell to create a network that can connect all the different parts of the cell together. This network could be used not only as a scaffold to hold components in place but also as a reference for direction. For example, we can use a map to determine the direction we need to travel and roads to connect and travel from home to campus. Likewise, an interconnecting network inside the cell can be used to direct and move compounds from one location to a final destination. Some of the required characteristics of this network are listed below. Can you add to this list?
Intracellular network
- The network needs to be extensive, and connect every area of the cell.
- The network needs to be flexible, able to change and adapt as the cell grows larger, divides into two cells, or physically moves from one environment to another.
- The network needs to be strong, able to hold up to mechanical pressure from inside the cell or from outside of the cell.
- The network needs to be composed of different fibers and each of these fibers needs to be for a specific connection in the cell. For example, certain fibers might be involved in holding organelles in place, and other fibers would be involved in connecting two different organelles.
- The fibers need to have directionality (or polarity), meaning they need to have a defined starting point and a defined end to help direct movement from one location to another.
- The fibers need to work with proteins that can convert chemical energy into kinetic energy, to actively transport compounds along the fibers.
Microfilaments
Actin
Microfilaments are cytoskeleton fibers composed of actin subunits. Actin is one of the most abundant proteins in eukaryotic cells and comprises 20% of total cellular protein by weight in muscle cells. The actin amino acid sequence is highly conserved in eukaryotic cells, meaning that the protein amino acid sequence, and therefore its final 3-D shape, has changed little over the course of evolution, maintaining more than 80% similarity between algae and humans.
Actin can be present as either a free monomer called G-actin (globular) or as part of a polymer microfilament called F-actin ("F" for filamentous). Actin must be bound to ATP in order to assemble into its filamentous form and maintain the structural integrity of the filament. The actin filament itself has structural polarity. This term "polarity", in reference to a cytoskeleton filament, does not mean what it did when we discussed polar functional groups earlier in this course. Polarity here refers to the fact that there are two distinct ends to the filament. These ends are called the "(-)" end and the "(+)" end. At the "(+)" end, actin subunits are adding onto the elongating filament and at the "(-)" end, actin subunits are disassembling or falling off of the filament. This process of assembly and disassembly is controlled by the ATP to ADP ratio in the cytoplasm.
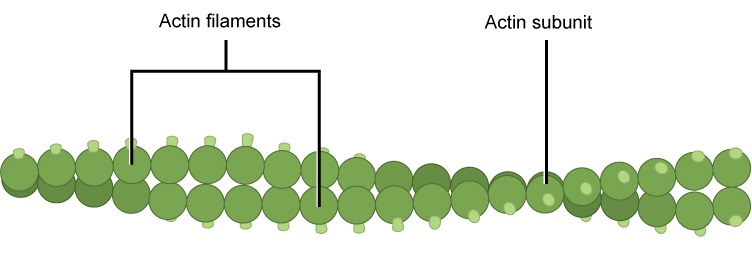
Figure 2. Microfilaments are the narrowest of the three cytoskeleton fibers, with a diameter of about seven nm. Microfilaments are composed of actin subunits which form into two intertwined strands.
Actin participates in many cellular processes, including muscle contraction, cell motility, cytokinesis during cell division, vesicle and organelle movement, and the maintenance of cell shape. Actin filaments serve as a track for the movement of a family of motor proteins called myosins discussed in more detail in a section below.
Link to learning:
To see an example of a white blood cell in action, click here and watch a short time-lapse video of the cell capturing two bacteria. It engulfs one and then moves on to the other.
Animations on actin filaments and how they work
Intermediate filaments
Intermediate filaments are made of several strands of fibrous proteins that are wound together. These elements of the cytoskeleton get their name from the fact that their diameter, eight to ten nm, is between those of the smaller microfilaments and the larger microtubules. The intermediate filaments are the most diverse group of cytoskeletal elements. Several types of fibrous proteins are found in the intermediate filaments. You are probably most familiar with keratin, the fibrous protein that strengthens your hair, nails, and the epidermis of the skin.
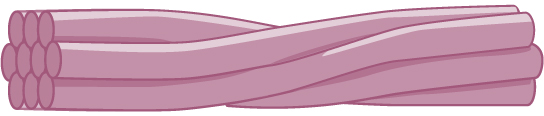
Figure 3. Intermediate filaments consist of several intertwined strands of fibrous proteins.
Intermediate filaments have no role in cell movement. Their function is purely structural. They bear tension, thus maintaining the shape of the cell, and anchor the nucleus and other organelles in place. The figure above shows how intermediate filaments create a cable-like supportive scaffolding inside the cell.
Microtubules
Microtubules are the largest component of the cytoskeleton and are found throughout the cytoplasm. These polymers are made up of globular protein subunits called α-tubulin and β-tubulin. Microtubules are found not only in eukaryotic cells but in some bacteria as well.
Both the α-tubulin and β-tubulin subunits bind to GTP. When bound to GTP, the formation of the microtubule can begin, this is called the nucleation event. As more GTP tubulin dimers assemble onto the filament, GTP is slowly hydrolyzed by β-tubulin to form GDP. Tubulin bound to GDP is less structurally robust and can lead to disassembly of the microtubule.
Much like the actin filaments discussed above, microtubules also have a distinct polarity that is critical for their biological function. Tubulin polymerizes end to end, with the β-subunits of one tubulin dimer contacting the α-subunits of the next dimer. These differences lead to different subunits being exposed on the two ends of the filament. The ends are designated the "(−)" and "(+)" ends. Unlike actin filaments, microtubules can elongate at both the "(+)" and "(-)" ends, but elongation is significantly more rapid at the "(+)" end.
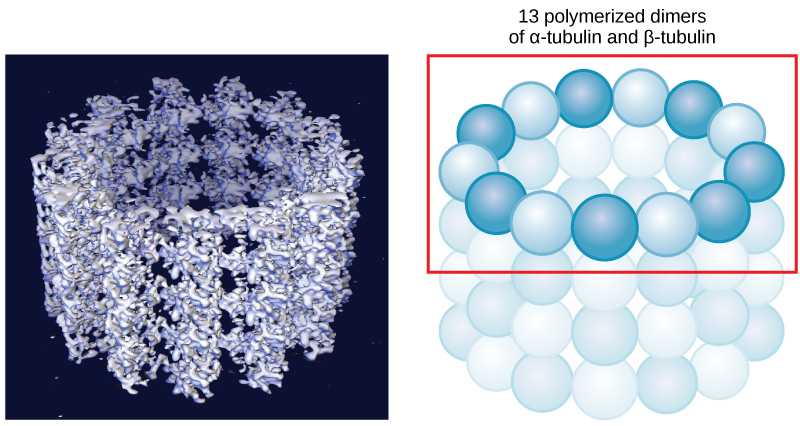
Figure 4. Microtubules are hollow. Their walls consist of 13 polymerized dimers of α-tubulin and β-tubulin (right image). The left image shows the molecular structure of the tube.
Microtubules help the cell resist compression, provide a track along which vesicles move through the cell, pull replicated chromosomes to opposite ends of a dividing cell, and are the structural elements of flagella, cilia, and centrioles (the latter are the two perpendicular bodies of the centrosome). In fact, in animal cells, the centrosome is the microtubule organizing center. In eukaryotic cells, flagella and cilia are quite different structurally from their counterparts in bacteria, discussed below.
Animations of the cytoskeleton
Where did these fibers come from?
The cytoskeleton probably has its origins in bacterial and/or archaeal ancestry. There are ancient relatives to both actin and tubulin in bacterial systems. In bacteria, the MreB protein and the ParM protein are believed to be early ancestors to Actin. MreB functions in maintaining cell shape and ParM functions in plasmid (DNA) partitioning. The FtsZ protein in bacteria functions in cytokinesis, it is a GTPase, spontaneously forms filaments and is hypothesized to be an ancient form of tubulin. These findings support the hypothesis that the eukaryotic cytoskeleton has its origins in the bacterial world.
Flagella and cilia
Flagella (singular=flagellum) are long, hair-like structures that extend from the plasma membrane and are used to move an entire cell (for example, sperm, Euglena). When present, the cell has just one flagellum or a few flagella. Cilia are short, hair-like structures that are used to move entire cells (such as paramecia) or substances along the outer surface of the cell (for example, the cilia of cells lining the fallopian tubes that move the ovum toward the uterus, or cilia lining the cells of the respiratory tract that trap particulate matter and move it toward your nostrils.) When cilia are present, there can be many of them, extending along the entire surface of the plasma membrane.
Despite their differences in length and number, flagella and cilia share a common structural arrangement of microtubules called a “9+2 array.” This is an appropriate name because a single flagellum or cilium is made of a ring of nine microtubule doublets, surrounding a single microtubule doublet in the center (Figure 5).
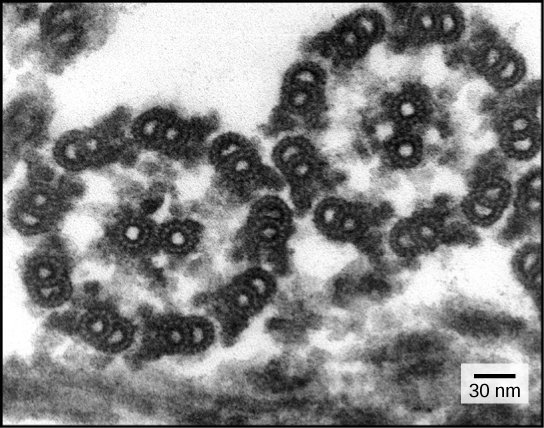
Figure 5. This transmission electron micrograph of two flagella shows the "9+2 array" of microtubules: nine microtubule doublets surround a single microtubule doublet. (credit: modification of work by Dartmouth Electron Microscope Facility, Dartmouth College; scale-bar data from Matt Russell)
For a video on flagellar and ciliar movement in eukaryotes, see the YouTube video: click here (you can skip the commericial).
Motor proteins
One function of the cytoskeleton is to move cellular components from one part of the cell to another. These cellular components are called "cargo" and are often stored within a vesicle for transport. You can think of the cytoskeleton as "railroad tracks" providing support and directionality inside of the cell.
Of course, if there are "railroad tracks" there needs to be an engine that can both move on the tracks and pull or push cargo along. In this case the engines are molecular motors that can move along the tracks in a specific direction. There are two families of molecular motors associated with the cytoskeleton; dyneines and kinesins. These motor proteins (train engines) and the cytoskeleton create a comprehensive network within the cell for moving vesicles (box cars) from one organelle to another or from one organelle to the cell surface.
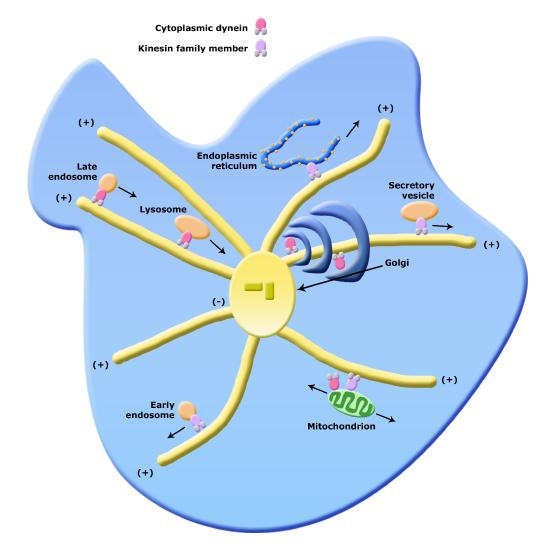
Figure 6. Organelle transport via microtubules and kinesins and dynes. Note that the figure is conceptual and only intended to show directionality of movement of various organelles; it does not necessarily represent all of their forms faithfully.
Cytoplasmic dyneins
Dynein is a protein complex that functions as a molecular motor. In cells, it converts the chemical energy from ATP hydrolysis into the mechanical energy of movement to 'walk' along the microtubule while carrying a vesicle. Dyneins bind to microtubules and move or "walk" from the plus "(+)" end of the cytoskeletal microtubule filament to the minus "(-)" end of the filament, which is usually oriented towards the cell center. Thus, they are often referred to as "minus end directed motors" and this vesicular transport is refereed to as retrograde transport. Cytoplasmic dynein moves processively along the microtubule, hydrolyzing ATP with each "step" it takes along the microtubule. During this process, one or the other of its "stalks" is always attached to the microtubule, allowing for the dynein motor (and its cargo) to "walk" a considerable distance along a microtubule without detaching.
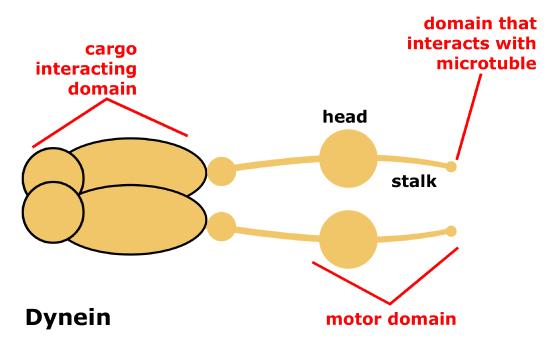
Figure 7. Schematic of cytoplasmic dynein motor protein. Dyneins are protein complexes composed of many smaller polypeptide subunits. The overall structure of the dynien motors are relatively simple, consisting of two identical complexes each having a motor domain that interacts with the microtubule, a stalk, or stem region that connects the motor head to the cargo interacting domain.
Cytoplamic dyneins are used in many different processes: they are involved in organelle movement such as the positioning of the Golgi complex and other organelles in the cell; they are used in the transport of cargo such as the movement of vesicles made by the endoplasmic reticulum, endosomes, and lysosomes; and they are responsible for the movement of chromosomes during cell division. Axonemal dyneins are motor proteins used in the sliding of microtubules in the axonemes of cilia and flagella in eukaryotic cells.
Kinesins
Kinesins, like cytoplasmic dyneins are motor-protein complexes that "walk" along the microtubules and are involved in vesicle transport. Unlike cytoplasmic dyneins, the polarity of kinesin movement is from the "(-)" end of the microtubule to the "(+)" end with the hydrolysis of ATP. In most cells, this entails transporting cargo from the center of the cell towards the periphery (the opposite direction to dyneins). This form of transport is known as anterograde or orthrograde transport. Like cytoplasmic dyneins, kinesins are involved in a variety of cellular processes including vesicle movement and chromosome movement during cell division.
The structure of kinesins are similar to cytoplasmic dyneins and is diagrammed in Figure 8. Members of the kinesin superfamily vary in shape, but the overall structure is that of a heterotetramer whose motor subunits (heavy chains) form a protein dimer (molecule pair) that binds two light chains.
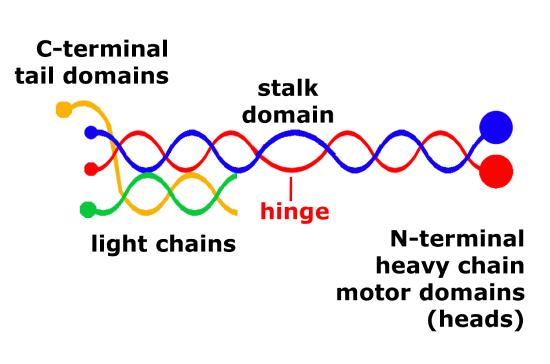
Figure 8. Schematic of kinesin motor proteins. The heavy chains comprise a globular head (the motor domain) at the amino terminal end connected via a short, flexible neck linker to the stalk—a long, central α-helical coiled-coil domain—that ends in a carboxy terminal tail domain which associates with the light-chains. The stalks of two light chains intertwine to form a coiled-coil that directs dimerization of the two heavy chains. In most cases transported cargo binds to the kinesin light chains, but in some cases cargo binds to the C-terminal domains of the heavy chains.
Animations of kinesin and dynein at work
How do the motors interact with cargo and the microtubules?
Cytoplasmic dyneins and kinesins interact with both cargo and microtubules in similar fashion. The light chains interact with receptors on the various cargo vesicles and the globular motor domains, specifically interact with the microtubules.
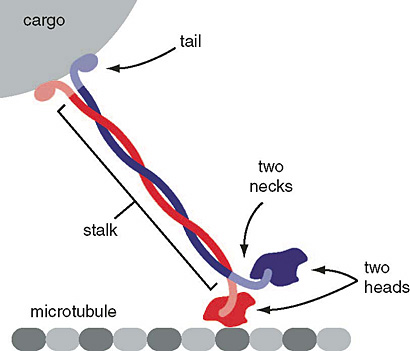
Figure 9. Schematic of kinesin motor protein carrying a cargo vesicle along a microtubule filament.
Note: possible discussion
What are the benefits for having multiple types of motor proteins? Multiple types of filaments? Filaments with polarity?

