The First Law of Thermodynamics
The first law of thermodynamics deals with the total amount of energy in the universe. It states that this total amount of energy is constant. In other words, there always has been, and always will be, exactly the same amount of energy in the universe.
According to the first law of thermodynamics, energy may be transferred from place to place, but it cannot be created or destroyed. Energy transfers take place around us all the time. Light bulbs transfer energy from electrical power stations into heat and photons of light. Gas stoves transfer energy stored in the bonds of chemical compounds into heat and light. (Heat, by the way, is the amount of energy transferred from one system to another because of a temperature difference.)
Plants perform one of the most biologically useful energy transfers on earth: they transfer energy in the photons of sunlight into the chemical bonds of organic molecules. In every one of these cases, energy is neither made nor destroyed, and we must try to account for all the energy when we examine some of these reactions.
The First Law and the Energy Story
The first law of thermodynamics is deceptively simple. Students often understand that energy cannot be created or destroyed. Yet, when describing an energy story of a process they often make the mistake of saying things such as "energy is produced from the transfer of electrons from atom A to atom B." While most of us will understand the point the student is trying to make, the wrong words are being used. Energy is not made or produced; it is simply transferred. To be consistent with the first law, when telling an energy story, make sure you try to track explicitly all the places that ALL the energy in the system at the start of a process goes by the end of a process.
The Second Law of Thermodynamics
An important concept in physical systems is entropy. Entropy relates to how energy can be distributed or dispersed within the particles of a system. The Second Law of Thermodynamics states that entropy is always increasing in a system and its surroundings (that is, everything inside and outside the system combined).
This idea helps explain the directionality of natural phenomena. The notion is that the directionality comes from the tendency for energy in a system to move towards a state of maximal dispersion. The Second Law, therefore, implies that in any transformation, we should look for an overall increase in entropy (or dispersion of energy), somewhere. As dispersion of energy in a system or its surroundings increases, the ability of the energy to be directed towards work decreases.
Keep in mind: you will find many examples in which the entropy of a system decreases locally. However, according to the Second Law, the entropy of the entire universe can never decrease. This must mean that there is an equal or greater increase in entropy somewhere else in the surroundings (most likely in a closely connected system) that compensates for the local decrease.
We associate the four scenarios below with increasing entropy of the system. Try to think of specific examples for when:
a. the system gains energy;
b. a change of state occurs from solid to liquid to gas;
c. a mixing of substances occurs;
d. the number of particles increases during a reaction.
Possible NB Discussion  Point
Point
Justify or refute the following statement: "Biological systems are an exception to the Second Law of Thermodynamics, since cells are known to arrange themselves into very highly organized structures (think: tissues, organs, etc.) rather than into a more disordered state." Make sure to check out what your peers are saying -- do you agree or disagree with their position and/or their rationale?
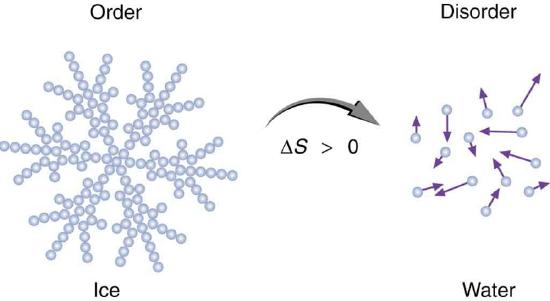
Figure 1. An increase in disorder can happen in different ways. An ice cube melting on a hot sidewalk is one example. Here, ice is displayed as a snowflake, with organized, structured water molecules forming the snowflake. Over time, the snowflake will melt into a pool of disorganized, freely moving water molecules. It is common to describe entropy as a measure of order as a way to simplify the more concrete description relating entropy to the number of states in which energy can be dispersed in a system. While the idea of measuring order to define entropy has some flaws, it is sometimes a useful, if imperfect, proxy. (Source)
If we consider the first and second laws together, we come to a useful conclusion. Whenever energy is transferred or redistributed within a system, entropy must increase. This increase in entropy is related to how "useful" the energy is to do work. Recall again that this energy becomes less and less available as entropy increases.
Keep in mind: you will find many examples in which the entropy of a system decreases locally. However, according to the Second Law, the entropy of the entire universe can never decrease. This must mean that there is an equal or greater increase in entropy somewhere else in the surroundings (most likely in a closely connected system) that compensates for the local decrease.
We conclude that while all the energy must be conserved, if the required change increases entropy, it means that some energy will become distributed in a way that makes it less useful for work. Most times, particularly in biology, some increase in entropy can be chalked up to a transfer of energy to heat in the environment.

 Point
Point


