Membranes*#
- Page ID
- 21312
Membranes
Plasma membranes enclose and define the borders between the inside and the outside of cells. They are typically composed of dynamic bilayers of phospholipids into which various other lipid-soluble molecules and proteins have also been embedded. These bilayers are asymmetric—the outer leaf differing from the inner leaf in lipid composition and in the proteins and carbohydrates that are displayed to either the inside or outside of the cell. One major function of the outer cell membrane is to communicate the cell’s unique identity to other cells. The proteins, lipids, and sugars displayed on the cell membrane allow for cells to be detected by and to interact with specific partners.
Various factors influence fluidity, permeability, and various other physical properties of the membrane. These include temperature, the configuration of the fatty acid tails (some
Amoebae Hunting Video
Cellular membranes
A subgoal in our "build-a-cell" design challenge is to create a boundary that separates the "inside" of the cell from the environment "outside". This boundary needs to serve multiple functions that include:
- Act as a barrier by blocking some compounds from moving in and out of the cell.
- Be selectively permeable
in order to transport specific compounds into and out of the cell. - Receive, sense, and transmit signals from the environment to inside of the cell.
- Project "self" to others by communicating identity to other nearby cells.

Figure 1. The diameter of a typical balloon is 25cm and the thickness of the plastic of the balloon of around 0.25mm. This is a 1000X difference. A typical eukaryotic cell will have a cell diameter of about
Fluid mosaic model
The fluid mosaic model describes the dynamic movement of the
For some insight into the history of our understanding of the plasma membrane structure, click here.
It is sometimes useful to start our discussion by recalling the size of the cell membrane relative to the size of the entire cell

Figure 2. The fluid mosaic model of the plasma membrane describes the plasma membrane as a fluid combination of phospholipids, cholesterol, and proteins. Carbohydrates attached to lipids (glycolipids) and
The principal components of a plasma membrane are lipids (phospholipids and cholesterol), proteins, and carbohydrates. Carbohydrates are present only on the exterior surface of the plasma membrane and
Phospholipids
Phospholipids are major constituents of the cell membrane.
Make sure to note in Figure 3 that the phosphate group has an R group linked to one of the oxygen atoms. R is a variable commonly used in these types of diagrams to indicate that some other atom or molecule is bound at that position. That part of the molecule can be different in different phospholipids—and will impart some different chemistry to the whole molecule. At the moment, however, you are responsible for being able to recognize this type of molecule (no matter what the R group is) because of the common core elements—the glycerol backbone, the phosphate group, and the two hydrocarbon tails.

Figure 3. A phospholipid is a molecule with two fatty acids and a
Attribution:
When many phospholipids
Possible NB Discussion  Point
Point
Earlier in the course, we discussed the Second Law of Thermodynamics, which states that the overall entropy of the universe is always increasing. Apply this law in the context of the formation of the lipid bilayer membrane. How is it possible that the lipids are able to spontaneously arrange themselves into such an organized structure instead of scatter into a more disordered state? Or in other words -- if the second law holds true, then how exactly does the spontaneous lipid organization lead to increased entropy?

Figure 4. In the presence of water, some phospholipids will spontaneously arrange themselves into a micelle.
Source: Created by
Membrane proteins
Proteins make up the second major component of plasma membranes. Integral membrane proteins, as their name suggests, integrate completely into the membrane structure, and their hydrophobic membrane-spanning regions interact with the hydrophobic region of the phospholipid bilayer.
Some membrane proteins associate with only one half of the bilayer, while others stretch from one side of the membrane to the other, and
Peripheral proteins are found on only one side of the membrane, but never embed into the membrane. They can be on the intracellular side or the extracellular side, and weakly or temporarily associated with the membranes.

Figure 5. Integral membranes proteins may have one or more α-helices (pink cylinders) that span the membrane (examples 1 and 2), or they may have β-sheets (blue rectangles) that span the membrane (example 3). (credit: “Foobar”/Wikimedia Commons)
Carbohydrates
Carbohydrates are a third major component of plasma membranes. They are always found on the exterior surface of cells and are bound either to proteins (forming glycoproteins) or to lipids (forming glycolipids). These carbohydrate chains may consist of 2–60 monosaccharide units and can be either straight or branched. Along with peripheral proteins, carbohydrates form specialized sites on the cell surface that allow cells to recognize each other (one of the core functional requirements noted above.
Membrane fluidity
The integral proteins and lipids exist in the membrane as separate molecules and they "float" in the membrane, moving with respect to one another. The membrane is not like a balloon, however; because of the elastic properties of its plastic a balloon can easily grow and shrink its surface area without popping and while also maintaining the same rough circular shape. By contrast the plasma membrane is not able to withstand isotropic stretching or compression and can be easily popped when an imbalance of solute between inside and out causes water to rush in suddenly. A sudden loss of water will cause it to shrivel and wrinkle, dramatically changing the shape of the cell. it is fairly rigid and can burst if penetrated or if a cell takes in too much water and the membrane is stretched too far. However, because of its mosaic nature, a very fine needle can easily penetrate a plasma membrane without causing it to burst (the lipids flow around the needle point), and the membrane will self-seal when the needle is extracted.
Different organisms and cell types in multicellular organisms can tune fluidity of their membrane to be more compatible with specialized functions and/or in response to environmental factors. This tuning can be accomplished by adjusting the type and concentration of various components of the membrane, including the lipids, their degree of saturation, the lipids, their degree of saturation, the proteins, and other molecules like cholesterol. There are two other factors that help maintain this fluid characteristic. One factor is the nature of the phospholipids themselves. In their saturated form, the fatty acids in phospholipid tails are saturated with hydrogen atoms. There are no double bonds between adjacent carbon atoms, resulting in tails that are relatively straight. By contrast, unsaturated fatty acids do not have a full complement of hydrogen atoms on their fatty acid tails and therefore contain some double bonds between adjacent carbon atoms; a double bond results in a bend in the string of carbons of approximately 30 degrees.

Figure 6. Any given cell membrane will be composed of a combination of saturated and unsaturated phospholipids. The ratio of the two will influence the permeability and fluidity of the membrane. A membrane composed of completely saturated lipids will be dense and less fluid, and a membrane composed of completely unsaturated lipids will be very loose and very fluid.
Saturated fatty acids, with straight tails, are compressed by decreasing temperatures, and they will press in on each other, making a dense and fairly rigid membrane. Conversely, when unsaturated fatty acids are compressed, the “kinked” tails elbow adjacent phospholipid molecules away, maintaining some space between the phospholipid molecules. This “elbow room” helps to maintain fluidity in the membrane at temperatures at which membranes with high concentrations of saturated fatty acid tails would “freeze” or solidify. The relative fluidity of the membrane is particularly important in a cold environment. Many organisms (fish are one example) are capable of adapting to cold environments by changing the proportion of unsaturated fatty acids in their membranes in response to the lowering of the temperature.
Cholesterol
Animal cells have cholesterol, an additional membrane constituent that assists in maintaining fluidity. Cholesterol, which lies right in between the phospholipids in the membrane, tends to dampen the effects of temperature on the membrane.Cholesterol both stiffens and increases membrane fluidity, depending on the temperature. Low temperatures cause phospholipids to pack together more tightly, creating a stiffer membrane. In this case, the cholesterol molecules serve to space the phospholipids apart and prevent the membrane from becoming totally rigid. Conversely, higher temperatures contribute to phospholipids moving farther apart from each other and therefore a more fluid membrane, but cholesterol molecules in the membrane take up space and prevent the complete dissociation of phospholipids.
Thus, cholesterol extends, in both directions, the range of temperature in which the membrane is appropriately fluid and consequently functional. Cholesterol also serves other functions, such as organizing clusters of transmembrane proteins into lipid rafts.

Figure 7. Cholesterol fits between the phospholipid groups within the membrane.
Review of the components of the membrane
| The components and functions of the plasma membrane | |
|---|---|
| Component | Location |
| Phospholipid | Main fabric of the membrane |
| Cholesterol | Between phospholipids and between the two phospholipid layers of animal cells |
| Integral proteins (e.g., integrins) | Embedded within the phospholipid layer(s); may or may not penetrate through both layers |
| Peripheral proteins | On the inner or outer surface of the phospholipid bilayer; not embedded within the phospholipids |
| Carbohydrates (components of glycoproteins and glycolipids) | Generally attached to proteins on the outside membrane layer |
Archaeal membranes
One major difference setting archaea apart from eukaryotes and bacteria is their membrane lipid composition. Though eukaryotes, bacteria, and archaea all use glycerol backbones in their membrane lipids, Archaea use long isoprenoid chains (20-40 carbons in length, derived from the five-carbon lipid isoprene) that are attached via ether linkages to glycerol, whereas eukaryotes and bacteria have fatty acids bonded to glycerol via ester linkages.
The polar head groups differ based on the genus or species of the Archaea and consist of mixtures of glyco groups (mainly disaccharides), and/or phospho groups primarily of phosphoglycerol, phosphoserine, phosphoethanolamine or phosphoinositol. The inherent stability and unique features of archaeal lipids have made them a useful biomarker for archaea within environmental samples, though approaches based on genetic markers are now more commonly used.
A second difference between bacterial and archaeal membranes that is associated with some archaea is the presence of monolayer membranes, as depicted below. Notice that the isoprenoid chain is attached to the glycerol backbones at both ends, forming a single molecule consisting of two polar head groups attached via two isoprenoid chains.
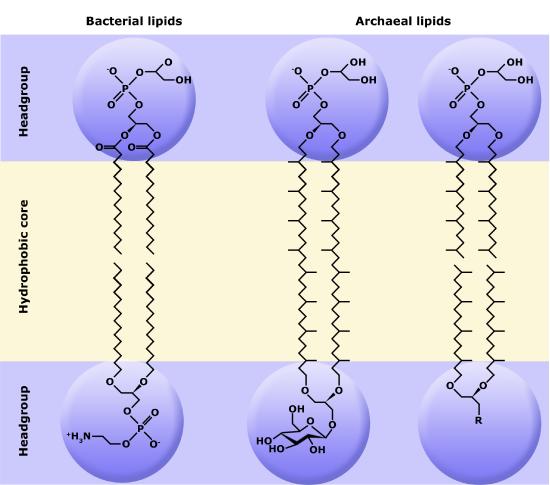
Figure 8. The exterior surface of the archaeal plasma membrane is not identical to the interior surface of the same membrane.
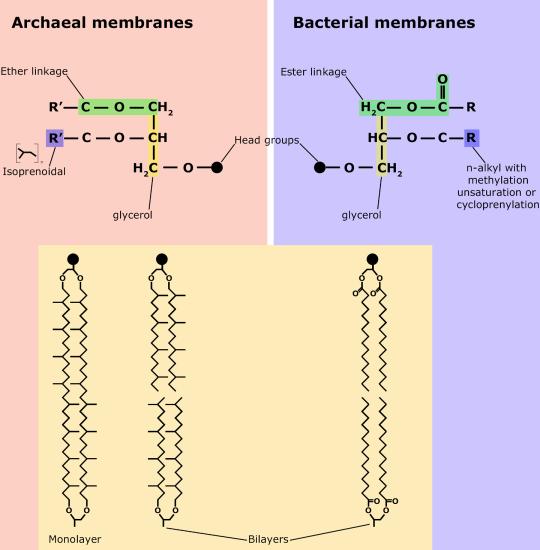
Figure 9. Comparisons of different types of archaeal lipids and bacterial/eukaryotic lipids


