1.20: Membranes and Transporters
- Page ID
- 8612
Membranes Overview
Plasma membranes enclose and define the borders between the inside and the outside of cells. They are typically composed of dynamic bilayers of phospholipids into which various other lipid soluble molecules and proteins have also been embedded. These bilayers are asymmetric - the outer leaf being different than the inner leaf in lipid composition and in the proteins and carbohydrates that are displayed to either the inside or outside of the cell. Various factors influence the fluidity, permeability and various other physical properties of the membrane. These include: temperature, the configuration of the fatty acid tails (some kinked by double bonds), the presence of sterols (i.e. cholesterol) embedded in the membrane, and the mosaic nature of the proteins embedded within it. Due to its hydrophobic nature, the cell membrane has selectivity; it is permeable to only some substances, while excluding others. In addition, the plasma membrane must in some cases be flexible enough to allow certain cells, such as amoebae, to change shape and direction as they move through the environment, hunting smaller, single-celled organisms.
This a video of an Amoeba . Movement of the Amoeba is shown. First the colorless ectoplasma moves in front of the pseodopodia, followed by the grained entoplasma. The video is done with the phase contrast technique. Amoebae Hunting Video
Cellular Membranes
A subgoal in our "Build-a-Cell" design challenge is to create a boundary that separates the "inside" of the cell from the environment "outside". This boundary needs to serve multiple functions that include:
- Act as a barrier: Block some compounds from moving in and out of the cell.
- Be selectively permeable: Transport specific compounds into and out of the cell.
- Receive, sense and transmit signals from the environment to inside of the cell.
- Project "self" to others: communicate identity to other nearby cells.
The diameter of a typical balloon is 25 cm, compared to the thickness of the plastic of the balloon, which is around 0.25 mm. This is a 1000X difference. A typical eukaryotic cell will have a cell diameter of about 50 μm, and a cell membrane thickness of 5 nm. This is a 10,000X difference.
Possible discussion
The ratio of the diameter of a eukaryotic cell compared to the thickness of membrane, is much greater than to that of a balloon stretched with air. To think that the boundary between life and nonlife is so small, and seemingly fragile, more so than a balloon, suggests that structurally the membrane must be very strong. Do you agree? If not, why not? What kinds of forces are on a membrane? What forces are on a balloon?
Fluid Mosaic Model
The plasma membrane was identified in the 1890s, and its chemical components were identified in 1915. The principal components identified at that time were lipids and proteins. The first widely accepted model of the plasma membrane’s structure was proposed in 1935 by Hugh Davson and James Danielli; it was based on the “railroad track” appearance of the plasma membrane in early electron micrographs. They theorized that the structure of the plasma membrane resembles a sandwich, with protein being analogous to the bread, and lipids being analogous to the filling. In the 1950s, advances in microscopy, notably transmission electron microscopy (TEM), allowed researchers to see that the core of the plasma membrane consisted of a double, rather than a single, layer. A model that better explains both the microscopic observations and the function of that plasma membrane was proposed by S.J. Singer and Garth L. Nicolson in 1972.
The explanation proposed by Singer and Nicolson is called the fluid mosaic model. The model has evolved somewhat over time, but it still best accounts for the structure and functions of the plasma membrane as we now understand them. The fluid mosaic model describes the structure of the plasma membrane as a mosaic of components—including phospholipids, cholesterol, proteins, and carbohydrates—that gives the membrane a fluid character. Plasma membranes range from 5 to 10 nm in thickness. For comparison, human red blood cells, visible via light microscopy, are approximately 8 µm wide, or approximately 1,000 times wider than a plasma membrane.

The fluid mosaic model of the plasma membrane describes the plasma membrane as a fluid combination of phospholipids, cholesterol, and proteins. Carbohydrates attached to lipids (glycolipids) and to proteins (glycoproteins) extend from the outward-facing surface of the membrane. This illustration is just a "slice" of membrane, and the fluidity is in only two dimensions- within the plane of the membrane, not across the membrane.
The proportions of proteins, lipids, and carbohydrates in the plasma membrane vary with organism and cell type, but for a typical human cell, protein accounts for about 50 percent of the composition by mass, lipids (of all types) account for about 40 percent of the composition by mass, with the remaining 10 percent of the composition by mass being carbohydrates. However, the concentration of proteins and lipids varies with different cell membranes. For example, myelin, an outgrowth of the membrane of specialized cells, insulates the axons of the peripheral nerves, contains only 18 percent protein and 76 percent lipid. The mitochondrial inner membrane contains 76 percent protein and only 24 percent lipid. The plasma membrane of human red blood cells is 30 percent lipid. Carbohydrates are present only on the exterior surface of the plasma membrane and are attached to proteins, forming glycoproteins, or attached to lipids, forming glycolipids.
Possible discussion
Consider the function of the axon of a very long nerve vs the function of the membranes found in mitochondria. Why might one have more protein embedded in its membranes than another?
Phospholipids
Phospholipids
Phospholipids are major constituents of the cell membrane, the outermost layer of cells. They are composed of fatty acid chains attached to a polar head group. Specifically, there are two fatty acid tails and a phosphate group as the polar head group (linked through a glycerol-like chain). The phospholipid is an amphipathic molecule, meaning it has a hydrophobic part and a hydrophilic part. The fatty acid chains are hydrophobic and cannot interact with water, whereas the phosphate-containing head group is hydrophilic and interacts with water.
Note
Make sure to note in the figure below that the phosphate group has an R-group linked to one of the oxygen atoms. R is a variable commonly used in these types of diagrams to indicate that some other atom or molecule is bound at that position. That part of the molecule can be different in different phospholipids - and will impart some different chemistry to the molecule. At the moment, however, you are responsible for being able to recognize this type of molecule (no matter what the R group is) because of the common core elements - the glycerol backbone, the phosphate group, and the two hydrocarbon tails.

A phospholipid is a molecule with two fatty acids and a modified phosphate group attached to a glycerol backbone. The phosphate may be modified by the addition of charged or polar chemical groups. Several chemical R groups may modify the phosphate. Choline, serine, and ethanolamine are shown here. These attach to the phosphate group at the position labeled R via their hydroxyl groups. Attribution: Marc T. Facciotti (own work)
A phospholipid bilayer forms as the basic structure of the cell membrane. The fatty acid tails of phospholipids face inside, away from water, whereas the phosphate group faces outside, hydrogen bonding with water. Phospholipids are responsible for the dynamic nature of the plasma membrane. If you were to mix some of these phosphoplipids with water in a test tube, they would spontaneously form a structure known as a micelle, where the hydrophilic phosphate heads face the outside and the fatty acids face the interior of this structure. A lipid bilayer (see the "fluid mosaic model" above) is another way of satisfying these same criteria- the hydrophobic tails are not exposed to water, while the polar heads are. Note that pure phospholipids would not form these structures- it's the presence of water, and the resulting hydrophobic/hydrophilic interactions, that results in the spontaneous formation of these organized structures.

In the presence of water, some phospholipids will spontaneously arrange themselves into a micelle. The lipids will be arranged such that their polar groups will be on the outside of the micelle, and the nonpolar tails will be on the inside. A lipid bilayer can also form, a two layered sheet only a few nanometers thick. The lipid bilayer consists of two layers of phospholipids organized in a way that all the hydrophobic tails align side by side in the center of the bilayer and are surrounded by the hydrophilic head groups.
Source: Created by Erin Easlon (own work)
Possible discussion
Above it says that if you were to take some pure phospholipids and drop them into water that some if it would spontaneously (on its own) form into micelles. This sounds a lot like something that could be described by an energy story. Go back to the energy story rubric and try to start creating an energy story for this process - I expect that the steps involving the description of energy might be difficult at this point (we'll come back to that later) but you should be able to do at least the first three steps. You can constructively critique (politely) each other's work to create an optimized story.
Possible discussion
Please note the figure above is highly "conceptual". What concepts are illustrated here? What details are left out?
Membrane Proteins
Proteins make up the second major component of plasma membranes. Integral proteins (some specialized types are called integrins) are, as their name suggests, integrated completely into the membrane structure, and their hydrophobic membrane-spanning regions interact with the hydrophobic region of the the phospholipid bilayer. Single-pass integral membrane proteins usually have a hydrophobic transmembrane segment that consists of 20–25 amino acids. Some span only part of the membrane—associating with a single layer—while others stretch from one side of the membrane to the other, and are exposed on either side. Some complex proteins are composed of up to 12 segments of a single protein, which are extensively folded and embedded in the membrane. This type of protein has a hydrophilic region or regions, and one or several mildly hydrophobic regions. This arrangement of regions of the protein tends to orient the protein alongside the phospholipids, with the hydrophobic region of the protein adjacent to the tails of the phospholipids and the hydrophilic region or regions of the protein protruding from the membrane and in contact with the cytosol or extracellular fluid. Peripheral proteins are found on either the exterior or interior surfaces of membranes; they are weakly or temporarily associated with the membranes. They can be attached (interact with) either to integral membrane proteins or interact weakly with the phospholipids within the membrane.

Integral membranes proteins may have one or more alpha-helices (pink cylinders) that span the membrane (examples 1 and 2), or they may have beta-sheets (blue rectangles) that span the membrane (example 3). (credit: “Foobar”/Wikimedia Commons)
Carbohydrates
Carbohydrates are the third major component of plasma membranes. They are always found on the exterior surface of cells and are bound either to proteins (forming glycoproteins) or to lipids (forming glycolipids). These carbohydrate chains may consist of 2–60 monosaccharide units and can be either straight or branched. Along with peripheral proteins, carbohydrates form specialized sites on the cell surface that allow cells to recognize each other (one of the core functional requirements noted above).
Membrane Fluidity
The mosaic characteristic of the membrane, described in the fluid mosaic model, helps to illustrate its nature. The integral proteins and lipids exist in the membrane as separate molecules and they 'float' in the membrane, moving somewhat with respect to one another. The membrane is not like a balloon, however, that can expand and contract; rather, it can burst if a cell takes in too much water. However, because of its mosaic nature, a very fine needle can easily penetrate a plasma membrane without causing it to burst, and the membrane will flow and self-seal when the needle is extracted. Thus a balloon and a cell membrane actually have very little in common!
The mosaic characteristics of the membrane explain some but not all of its fluidity. There are two other factors that help maintain this fluid characteristic. One factor is the nature of the phospholipids themselves. In their saturated form, the fatty acids are saturated with hydrogen atoms; there are no double bonds between adjacent carbon atoms. This results in the tails that are relatively straight. In contrast, unsaturated fatty acids do not contain a maximal number of hydrogen atoms, but they do contain some double bonds between adjacent carbon atoms; a double bond results in a bend in the string of carbons of approximately 30 degrees.

Any given cell membrane will be composed of a combination of saturated and unsaturated phospholipids. The ratio of the two will influence the permeability and fluidity of the membrane. A membrane composed of completely saturated lipids will be dense and less fluid, and a membrane composed of completely unsaturated lipids will be very loose and very fluid.
Possible discussion
The fluidity of the membrane- the speed with which proteins floating in the membrane can diffuse- is temperature-dependent, with more rapid diffusion at higher temperatures. What types of differences would you expect to see in the lipid composition of plants growing a near-freezing temperatures vs a summer day?
And another...
Looking at the membranes above, which do you think would be more permeable to small molecules?
Saturated fatty acids, with straight tails, are compressed by decreasing temperatures, and they will press in on each other, making a dense and fairly rigid membrane. When unsaturated fatty acids are compressed, the “kinked” tails elbow adjacent phospholipid molecules away, maintaining some space between the phospholipid molecules. This “elbow room” helps to maintain fluidity in the membrane at temperatures at which membranes with high concentrations of saturated fatty acid tails would “freeze” or solidify. The relative fluidity of the membrane is particularly important in a cold environment. A cold environment tends to compress membranes composed largely of saturated fatty acids, making them less fluid and more susceptible to rupturing. Many organisms (fish are one example) are capable of adapting to cold environments by changing the proportion of unsaturated fatty acids in their membranes in response to the lowering of the temperature.
Cholesterol
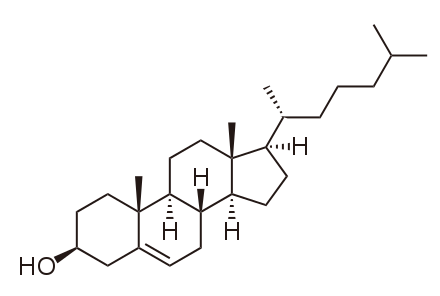 Animals have an additional membrane constituent that assists in maintaining a constant fluidity and in maintaining structural integrity (preventing rips and leaks -partially compensating for the lack of a cell wall). Cholesterol, which lies alongside the phospholipids in the membrane, tends to dampen the effects of temperature on the membrane. Thus, this lipid functions as a fluidity buffer, preventing lower temperatures from inhibiting fluidity and preventing increased temperatures from increasing fluidity too much. Cholesterol also serves other functions, such as organizing clusters of transmembrane proteins into lipid rafts.
Animals have an additional membrane constituent that assists in maintaining a constant fluidity and in maintaining structural integrity (preventing rips and leaks -partially compensating for the lack of a cell wall). Cholesterol, which lies alongside the phospholipids in the membrane, tends to dampen the effects of temperature on the membrane. Thus, this lipid functions as a fluidity buffer, preventing lower temperatures from inhibiting fluidity and preventing increased temperatures from increasing fluidity too much. Cholesterol also serves other functions, such as organizing clusters of transmembrane proteins into lipid rafts.

Cholesterol fits between the phospholipid groups within the membrane.
Review of the Components of the Membrane
| Component | Location |
|---|---|
| Phospholipid | Main fabric of the membrane |
| Cholesterol | -OH group lies near the polar heads, hydrophobic part lies alongside the lipid tails of animal cells |
| Integral proteins (for example, integrins) | Embedded within the phospholipid layer(s). May or may not penetrate through both layers |
| Peripheral proteins | On the inner or outer surface of the phospholipid bilayer; not embedded within the phospholipids |
| Carbohydrates (components of glycoproteins and glycolipids) | Branched polymers attached to proteins or lipids on the membrane |
Archaeal Membranes
One major difference between Archaea and either Eukaryotes or Bacteria is the composition of the archaeal membranes. Unlike eukaryotes and bacteria, archaeal membranes are not made up of fatty acids attached to a glycerol backbone. Instead, the polar lipids consist of isoprenoid (molecules derived from the 5 carbon lipid isoprene) chains of 20–40 carbons in length. These chains, which are usually saturated, are attached by ether bonds to the glycerol carbons at the 2 and 3 positions on the glycerol backbone, instead of the more familier ester linkage found in bacteria and eukaryotes. The polar head groups differ based on the genus or species of the archaea and consist of mixtures of glyco groups (mainly disaccharides), and/or phospho groups primarily phosphoglycerol, phosphoserine, phosphoethanolamine or phosphoinositol. The inherent stability and unique features of archaeal lipids makes them a useful biomarker for Archaea within environmental samples.
A second difference between bacterial and archaeal membranes that is associated with some archaea is the presence of monolayer membranes , as depicted below. Notice that the isoprenoid chain is attached to the glycerol backbones at both ends, forming a single molecule consisting of two polar head gropus attached via 2 isoprenoid chains.
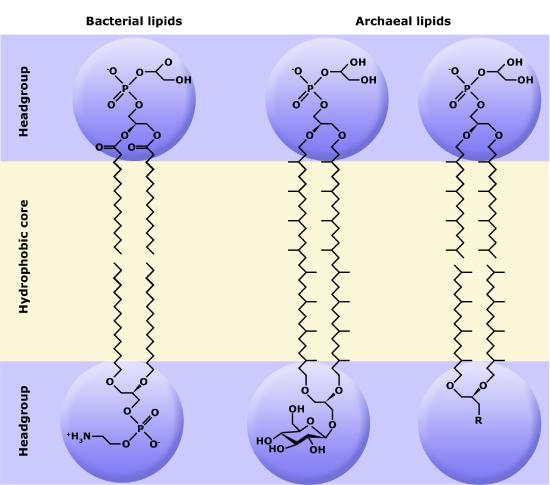
The exterior surface of the archaeal plasma membrane is not identical to the interior surface of the same membrane.
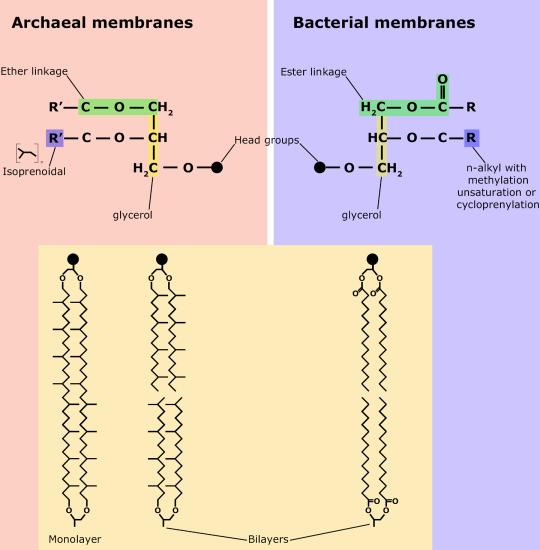
Comparisons of different types of Archaeal lipids and bacterial/eukaryotic lipids
Possible discussion
In many cases - though not all - the archaea are relatively abundant in environments that represent extremes for life (e.g. high temperature, high salt) what possible advantage could monolayered membranes provide?
Membrane Transport Overview
The chemistry of living things occurs in aqueous solutions, and balancing the concentrations of those solutions is an ongoing problem. In living systems, diffusion of substances into and out of cells is mediated by the plasma membrane. The passive forms of transport, diffusion and osmosis, move nonpolar materials of small molecular weight across membranes. Substances diffuse from areas of high concentration to areas of lower concentration, and this process continues until the substance is evenly distributed in a system. In solutions containing more than one substance, each type of molecule diffuses according to its own concentration gradient, independent of the diffusion of other substances. Some materials diffuse readily through the membrane, but others are hindered, and their passage is made possible by specialized proteins, such as channels and transporters.
Transport across the membrane
One of the great wonders of the cell membrane is its ability to regulate the concentration of substances inside the cell. These substances include ions such as Ca2+, Na+, K+, and Cl–; nutrients including sugars, fatty acids, and amino acids; and waste products, particularly carbon dioxide (CO2), which must leave the cell.
Design Challenge Subproblem
Controlling what enters and exits the cell.
The membrane’s lipid bilayer structure provides the first level of control. The phospholipids are tightly packed together, and the membrane has a hydrophobic interior. This structure causes the membrane to be selectively permeable. A membrane that has selective permeability allows only substances meeting certain criteria to pass through it unaided. In the case of the cell membrane, only relatively small, nonpolar materials can move through the lipid bilayer at biologically relevant rates (remember, the lipid tails of the membrane are nonpolar). The rates of transport of various molecules is tabulated in the Membranes section. All substances that move through the membrane do so by one of two general methods, which are categorized based on whether or not the transport process is exergonic or endergonic. Passive transport is the exergonic movement of substances across the membrane. In contrast, active transport is the endergonic movement of substances across the membrane that is coupled to an exergonic reaction.
Selective Permeability
Selective permeability of the cell membrane refers to its ability to differentiate between different types of molecules, only allowing some molecules through while blocking others. Some of this selective property stems from the intrinsic diffusion rates for different molecules across a membrane. A second factor affecting the relative rates of movement of various substances across a biological membrane is activity of various protein-based membrane transporters, both passive and active, that will be discussed in more detail in subsequent sections. First we take on the notion of intrinsic rates of diffusion across the membrane.
Relative Permeability
The fact that different substances might cross a biological membrane at different rates should be relatively intuitive. There are differences in the mosaic composition of membranes in biology and differences in the sizes, flexibility and chemical properties of molecules so it stands to reason that the permeability rates vary. The permeability of a substance across a biological membrane can be measured experimentally and the rate of movement across a membrane reported in what are known as membrane permeability coefficients.
Membrane Permeability Coefficients
Below, a variety of compounds are plotted with respect to their membrane permeability coefficients (MPC) as measured against simple biochemical approximation of a real biological membrane. The reported permeability coefficient for this system is the rate at which simple diffusion through a membrane occurs and is reported in units of centimeters per second (cm/s).
It is important that you are able to read and interpret the diagram below- however- please note the error at the top of the scale! Log scales don't go to zero. What value should this be? Once you have corrected this value, approximately what is the MPC of hexanoic acid? The larger the MPC, the more permeable the membrane is to the solute. For example, acetic acid, water and ethanol have MPC between 0.01 and 0.001, so they are less permeable than hexanoic acid. Whereas ions, such as sodium (Na+) have an MPC of 10-12, and cross the membrane very rarely.
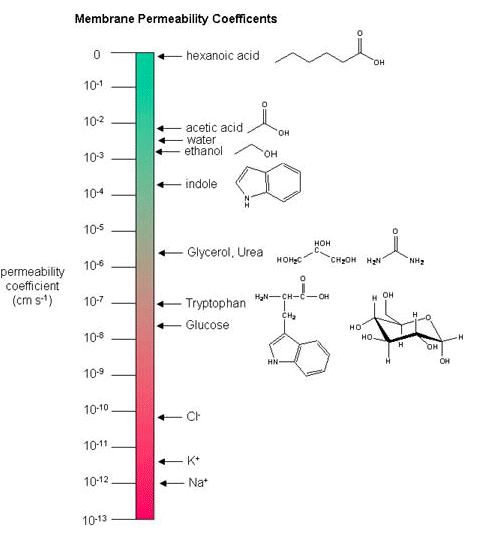
While there are certain trends or chemical properties that can be roughly associated with different compound permeabilities (small thing go through "fast", big things "slowly", charged things not at all etc.) we caution against over-generalizing. The molecular determinants of membrane permeability are complicated and involve numerous factors in addition to the chemical nature of the solute, including the specific composition of the membrane and temperature. So, in this class, rather than try to apply "rules" we will strive to develop a general sense of some properties that can influence permeability and leave the assignment of absolute permeability to experimentally reported rates. In addition, we will also try to minimize the use of vocabulary that depends on a frame of reference. For instance, saying that compound A diffuses "quickly" or "slowly" across a bilayer only means something if the terms "quickly" or "slowly" are numerically defined or the biological context understood.
Passive Transport
Passive transport does not require the cell to expend energy. In passive transport, the net flow of solute is from an area of higher concentration to an area of lower concentration, down the concentration gradient. It is, therefore, energetically favorable. Depending on the chemical nature of the substance, different processes may be associated with passive transport.
Diffusion
Diffusion is one form of passive process of transport. A single substance tends to move from an area of high concentration to an area of low concentration until the concentration is equal across a space. You are familiar with diffusion of substances through the air. For example, think about someone opening a bottle of ammonia in a room filled with people. The ammonia gas is at its highest concentration in the bottle; its lowest concentration is at the edges of the room. The ammonia vapor will diffuse, or spread away, from the bottle, and gradually, more and more people will smell the ammonia as it spreads. Materials move within the cell’s cytosol by diffusion, and certain materials move through the plasma membrane by diffusion.
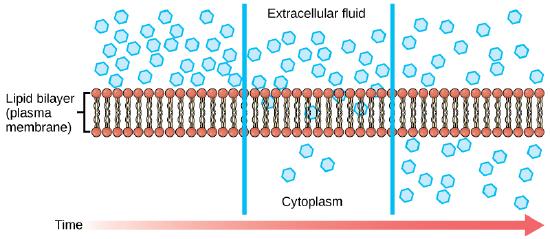
Factors That Affect Diffusion
If unconstrained, molecules will move through and explore space randomly at a rate that depends on their size, their shape, their environment, and their thermal energy. This type of movement underlies the diffusive movement of molecules through whatever medium they are in. The absence of a concentration gradient does not mean that movement will stop, just that there may be no net movement of the number of molecules from one area to another, a condition known as dynamic equilibrium. The diagram immediately above fails to illustrate this- there should still be molecule crossing the membrane, even at equilibrium.
Factors influencing diffusion include:
- Extent of the concentration gradient: The greater the difference in concentration, the more rapid the diffusion. The closer the distribution of the material gets to equilibrium, the slower the rate of diffusion becomes. Remember that by rate, we mean net flux from one side to the other. At equilibrium there is no "net" flux.
- Shape, size and mass of the molecules diffusing: Large and heavier molecules move more slowly; therefore, they diffuse more slowly. The reverse is typically true for smaller, lighter molecules.
- Temperature: Higher temperatures increase the energy and therefore the movement of the molecules, increasing the rate of diffusion. Lower temperatures decrease the energy of the molecules, thus decreasing the rate of diffusion.
- Solvent density: As the density of a solvent increases, the rate of diffusion decreases. The molecules slow down because they have a more difficult time getting through the denser medium. If the medium is less dense, rates of diffusion increase. Since cells primarily use diffusion to move materials within the cytoplasm, any increase in the cytoplasm’s density will decrease the rate at which materials move in the cytoplasm. We tend to think of the cytoplasm as "watery", but a lot of other materials are present in it, some (such as proteins) quite large and massive. Thus the cytoplasm is actually more viscous than water.
- Solubility: As discussed earlier, nonpolar or lipid-soluble materials pass through plasma membranes more easily than polar materials, allowing a faster rate of diffusion.
- Surface area and thickness of the plasma membrane: Increased surface area increases the rate of diffusion, whereas a thicker membrane reduces it.
- Size of the cell: The greater the distance that a substance must travel, the slower the rate of diffusion (the speed is the same, but the molecule needs to cover more space to get from one end of the cell to another). This places an upper limitation on cell size. Therefore, cells must either be small in size, as in the case of many prokaryotes, or be flattened, as with many single-celled eukaryotes. We'll see that in larger eukaryotic cells there are mechanisms that enhance the rate of movement of large molecules within the cell.
Facilitated diffusion
In facilitated diffusion, materials diffuse across the plasma membrane with the help of "transport" proteins. A concentration gradient exists that allows these materials to diffuse into or out of the cell without expending cellular energy. These proteins allow ions or polar molecules, compounds that are repelled by the hydrophobic parts of the cell membrane, to diffuse across through a hydrophylic "tunnel". Transport proteins shield these materials from the repulsive force of the membrane, allowing them to diffuse into the cell.
Note: Possible Discussion
Compare and contrast diffusion and facilitated diffusion.
Channels
The integral proteins involved in facilitated diffusion function as either channels or carriers. In both cases, they are transmembrane proteins, with several "passes" of the polypeptide chain through the membrane. Different channel proteins have different transport properties. The interior "passageway" of channel proteins have evolved to provide a low energetic barrier for passage of substances across the membrane through the complementary arrangement of amino acid functional groups (of both backbone and side-chains). Passage through the channel allows polar compounds to avoid the nonpolar central layer of the plasma membrane that would otherwise slow or prevent their entry into the cell. Channel proteins are often not particularly selective, for example transporting a number of different ions. Channel proteins also different from the "carrier" proteins described below in that- when active- they are simultaneously open at both ends, to both sides of the membrane. They are large enough to allow water to leak through, along with the ion or small molecules whose diffusion they normally facilitate.

Channel proteins are either open at all times or they are “gated.” The latter controls the opening of the channel. Various mechanisms may be involved in the gating mechanism. For instance, the attachment of a specific ion or small molecule to the channel protein may trigger opening. Changes in local membrane "stress" or changes in voltage across the membrane may also be triggers to open or close a channel.
Different organisms and tissues in multicellular species express different sets of channel proteins in their membranes depending on the environments they live in or specialized function they play in an organisms. This provides each type of cell with a unique membrane permeability profile that is evolved to complement its "needs". For example, in some tissues, sodium and chloride ions pass freely through open channels, whereas in other tissues a gate must be opened to allow passage. Cells involved in the transmission of electrical impulses, such as nerve and muscle cells, have gated channels for sodium, potassium, and calcium in their membranes. Opening and closing of these channels changes the relative concentrations on opposing sides of the membrane of these ions, resulting a change in electrical potential across the membrane that lead to message propagation in the case of nerve cells or in muscle contraction in the case of muscle cells.
Carrier Proteins
Another type of protein embedded in the plasma membrane is a carrier protein. This protein binds a substance and, in doing so, triggers a change of its own shape, moving the bound molecule across the membrane. Carrier proteins are typically specific for a single substance. This selectivity adds to the overall selectivity of the plasma membrane. Carrier proteins, unlike channels, are only "open" to one side of the membrane at a time.

Carrier proteins play an important role in the function of kidneys. Glucose, water, salts, ions, and amino acids needed by the body are filtered in one part of the kidney. This filtrate, which includes glucose, is then reabsorbed in another part of the kidney with the help of carrier proteins. Because there are only a finite number of carrier proteins for glucose, if more glucose is present in the filtrate than the proteins can handle, the excess is not reabsorbed and it is excreted from the body in the urine. In a diabetic individual, this is described as “spilling glucose into the urine.” A different group of carrier proteins called glucose transport proteins, or GLUTs, are involved in transporting glucose and other hexose sugars through plasma membranes within the body.
Channel and carrier proteins transport material at different rates. Channel proteins transport much more quickly than do carrier proteins. Channel proteins facilitate diffusion at a rate of tens of millions of molecules per second, whereas carrier proteins work at a rate of a thousand to a million molecules per second. As we'll see below, carrier proteins are often involved in active transport also. Can you see why channel proteins are probably involved only in facilitated diffusion?
Active Transport
Active Transport Summary
In the section above we described the the movement of molecules down a concentration and/or charge gradient- which is an exergonic change in state. Active transport, in contrast, refers to the active concentration of solutes and/or the movement of a solute "up" a gradient of charge and/or concentration. Proteins that perform these tasks are sometimes referred to as "pumps". Just as we can physically pump water uphill, or increase the pressure in a tire, pumping requires work. Therefore, active transport must be paid for via some type of energy exchange. Hopefully ATP springs to mind as a source of energy, and ATP hydrolysis can indeed power these sorts of transactions. However, existing gradients (often of ions, particularly protons) can also be depleted to "pay for" the concentration of a second solute- one "downhill" transaction will pay for another "uphill" transaction. One process may be physically coupled to the other via the transporter protein itself..
Two mechanisms exist for the transport of small-molecular weight material and small molecules. Primary active transport moves ions across a membrane and creates a difference in charge across that membrane, which is directly dependent on ATP. Secondary active transport describes the movement of material that is due to the electrochemical gradient established by primary active transport that does not directly require ATP.
Carrier Proteins for Active Transport
There are three classes of carriers (aka, permeases) for active transport. A uniporter carries one specific ion or molecule. A symporter carries two different ions or molecules, both in the same direction. An antiporter also carries two different ions or molecules, but in different directions. These three types of carrier proteins are also found in facilitated diffusion, but they do not require ATP to work in that process. Some examples of pumps for active transport are Na+-K+ ATPase, which carries sodium and potassium ions, and H+-K+ ATPase, which carries hydrogen and potassium ions. Both of these are antiporter carrier proteins. Two other carrier proteins are Ca2+ATPase and H+ ATPase, which carry only calcium and only hydrogen ions, respectively. Both are pumps.
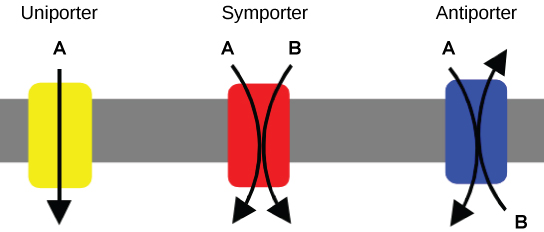
Primary Active Transport
In primary active transport, the energy is derived directly from the breakdown of ATP. Often times, primary active transport such as that shown below which functions to transport sodium and potassium ions allows secondary active transport to occur (discussed in the section below). The second transport method is still considered active because it depends on the use of energy from the primary transport.
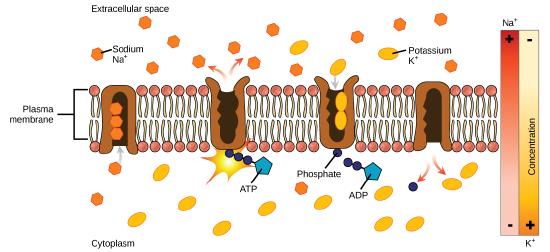
One of the most important pumps in animal cells is the sodium-potassium pump (Na+-K+ ATPase), which maintains the electrochemical gradient (and the correct concentrations of Na+and K+) in living cells. The sodium-potassium pump moves K+ into the cell while moving Na+ out at the same time, at a ratio of three Na+ for every two K+ ions moved in. The Na+-K+ATPase exists in two forms, depending on its orientation to the interior or exterior of the cell and its affinity for either sodium or potassium ions. The process consists of the following six steps. We're describing this process in detail as an illustration of how a "pump" can work.
- With the enzyme oriented towards the interior of the cell, the carrier has a high affinity for sodium ions. Three ions bind to the protein.
- ATP is hydrolyzed by the protein carrier and a low-energy phosphate group attaches to it.
- As a result, the carrier changes shape and re-orients itself towards the exterior of the membrane. The protein’s affinity for sodium decreases and the three sodium ions leave the carrier.
- The shape change increases the carrier’s affinity for potassium ions, and two such ions attach to the protein. Subsequently, the low-energy phosphate group detaches from the carrier.
- With the phosphate group removed and potassium ions attached, the carrier protein repositions itself towards the interior of the cell.
- The carrier protein, in its new configuration, has a decreased affinity for potassium, and the two ions are released into the cytoplasm. The protein now has a higher affinity for sodium ions, and the process starts again.
Several things have happened as a result of this process. At this point, there are more sodium ions outside of the cell than inside and more potassium ions inside than out. For every three ions of sodium that move out, two ions of potassium move in. This results in the interior being slightly more negative relative to the exterior. This difference in charge is important in creating the conditions necessary for the secondary process. The sodium-potassium pump is, therefore, an electrogenic pump (a pump that creates a charge imbalance), creating an electrical imbalance across the membrane and contributing to the membrane potential.
Link to Learning:
Visit the site to see a simulation of active transport in a sodium-potassium ATPase.
Secondary active transport (Co-transport)
Secondary active transport (aka co-transport) can also act as a pump, concentrating a solute (a positive ∆G) by diluting another solute (negative ∆G). In the example below, sodium ions are concentrated outside of the cell (this transport occurs in the kidneys). As sodium ion concentrations build up outside of the plasma membrane, an electrochemical gradient is created. If a Na+ carrier protein exists, the sodium ions can spontaneously pass through this protein and therefore across the membrane. This exergonic dilution is used to transport and concentrate other substances which are also substrates for this transport protein- in this example glucose. In this example, Na+ cannot be transported unless glucose is also bound,a dn transported. This transporter would be called a "Na+-glucose symporter".
For thought-
Would an antiporter also be able to couple a negative ∆G to power a positive ∆G process? Come up with an example.

For Discussion
Compare and contrast chemical reactions mediated by enzymes and membrane transport mediated by proteins. What concepts do these two processes share? And related to this discussion- does a uniporter carry its solute only in one direction (say, only from outside to inside)?
Osmosis
Osmosis is the movement of water through a semipermeable membrane according to the concentration gradient of water across the membrane, which is inversely proportional to the concentration of solutes. While diffusion transports material across membranes and within cells, osmosis transports only water across a membrane and the membrane limits the diffusion of solutes in the water.
Mechanism
Osmosis is a special case of diffusion. Water, like other substances, moves from an area of high concentration to one of low concentration. An obvious question is what makes water move at all? Imagine a beaker with a semipermeable membrane separating the two sides or halves. On both sides of the membrane the water level is the same, but there are different concentrations of a dissolved substance, or solute, that cannot cross the membrane (otherwise the concentrations on each side would be balanced by the solute crossing the membrane). If the volume of the solution on both sides of the membrane is the same, but the concentrations of solute are different, then there are different amounts of water, the solvent, on either side of the membrane.
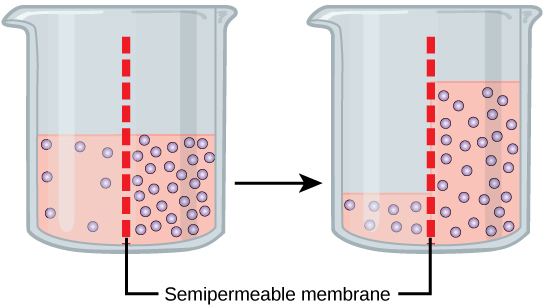
To illustrate this, imagine two full glasses of water. One has a single teaspoon of sugar in it, whereas the second one contains one-quarter cup of sugar. If the total volume of the solutions in both cups is the same, which cup contains more water? Because the large amount of sugar in the second cup takes up much more space than the teaspoon of sugar in the first cup, the first cup has more water in it.
Returning to the beaker example, recall that it has a mixture of solutes on either side of the membrane. A principle of diffusion is that the molecules move around and will spread evenly throughout the medium if they can. However, only the material capable of getting through the membrane will diffuse through it. In this example, the solute cannot diffuse through the membrane, but the water can. Water has a concentration gradient in this system. Thus, water will diffuse down its concentration gradient, crossing the membrane to the side where it is less concentrated. This diffusion of water through the membrane—osmosis—will continue until the concentration gradient of water goes to zero or until the hydrostatic pressure of the water balances the osmotic pressure. Osmosis proceeds constantly in living systems.
Tonicity
Tonicity describes how an extracellular solution can change the volume of a cell by affecting osmosis. A solution's tonicity often directly correlates with the osmolarity of the solution. Osmolarity describes the total solute concentration of the solution. A solution with low osmolarity has a greater number of water molecules relative to the number of solute particles; a solution with high osmolarity has fewer water molecules with respect to solute particles. In a situation in which solutions of two different osmolarities are separated by a membrane permeable to water, though not to the solute, water will move from the side of the membrane with lower osmolarity (and more water) to the side with higher osmolarity (and less water). This effect makes sense if you remember that the solute cannot move across the membrane, and thus the only component in the system that can move—the water—moves along its own concentration gradient. An important distinction that concerns living systems is that osmolarity measures the number of particles (which may be molecules) in a solution. Therefore, a solution that is cloudy with cells may have a lower osmolarity than a solution that is clear, if the second solution contains more dissolved molecules than there are cells.
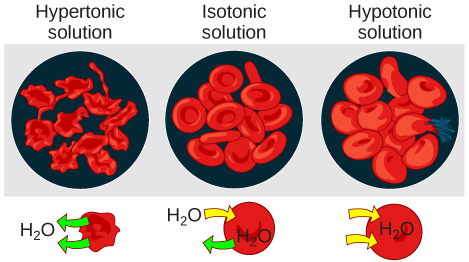
Hypotonic Solutions
Three terms—hypotonic, isotonic, and hypertonic—are used to relate the osmolarity of a cell to the osmolarity of the extracellular fluid that contains the cells. In a hypotonic situation, the extracellular fluid has lower osmolarity than the fluid inside the cell, and water enters the cell. (In living systems, the point of reference is always the cytoplasm, so the prefix hypo- means that the extracellular fluid has a lower concentration of solutes, or a lower osmolarity, than the cell cytoplasm.) It also means that the extracellular fluid has a higher concentration of water in the solution than does the cell. In this situation, water will follow its concentration gradient and enter the cell.
Hypertonic Solutions
As for a hypertonic solution, the prefix hyper- refers to the extracellular fluid having a higher osmolarity than the cell’s cytoplasm; therefore, the fluid contains less water than the cell does. Because the cell has a relatively higher concentration of water, water will leave the cell.
Isotonic Solutions
In an isotonic solution, the extracellular fluid has the same osmolarity as the cell. If the osmolarity of the cell matches that of the extracellular fluid, there will be no net movement of water into or out of the cell, although water will still move in and out. Blood cells and plant cells in hypertonic, isotonic, and hypotonic solutions take on characteristic appearances.
Connection:
A doctor injects a patient with what the doctor thinks is an isotonic saline solution. The patient dies, and an autopsy reveals that many red blood cells have been destroyed. Do you think the solution the doctor injected was really isotonic?
Link to Learning:
For a video illustrating the process of diffusion in solutions, visit this site.
Tonicity in Living Systems
In a hypotonic environment, water enters a cell, and the cell swells. In an isotonic condition, the relative concentrations of solute and solvent are equal on both sides of the membrane. There is no net water movement; therefore, there is no change in the size of the cell. In a hypertonic solution, water leaves a cell and the cell shrinks. If either the hypo- or hyper- condition goes to excess, the cell’s functions become compromised, and the cell may be destroyed.
A red blood cell will burst, or lyse, when it swells beyond the plasma membrane’s capability to expand. Remember, the membrane resembles a mosaic, with discrete spaces between the molecules composing it. If the cell swells, and the spaces between the lipids and proteins become too large, the cell will break apart.
In contrast, when excessive amounts of water leave a red blood cell, the cell shrinks, or crenates. This has the effect of concentrating the solutes left in the cell, making the cytosol denser and interfering with diffusion within the cell. The cell’s ability to function will be compromised and may also result in the death of the cell.
Various living things have ways of controlling the effects of osmosis—a mechanism called osmoregulation. Some organisms, such as plants, fungi, bacteria, and some protists, have cell walls that surround the plasma membrane and prevent cell lysis in a hypotonic solution. The plasma membrane can only expand to the limit of the cell wall, so the cell will not lyse. In fact, the cytoplasm in plants is always slightly hypertonic to the cellular environment, and water will always enter a cell if water is available. This inflow of water produces turgor pressure, which stiffens the cell walls of the plant. In nonwoody plants, turgor pressure supports the plant. Conversely, if the plant is not watered, the extracellular fluid will become hypertonic, causing water to leave the cell. In this condition, the cell does not shrink because the cell wall is not flexible. However, the cell membrane detaches from the wall and constricts the cytoplasm. This is called plasmolysis. Plants lose turgor pressure in this condition and wilt.
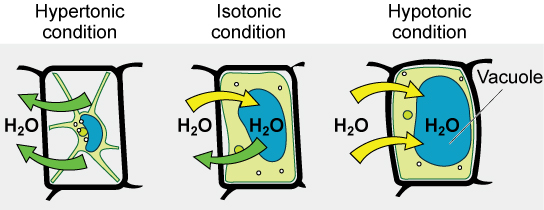
Tonicity is a concern for all living things. For example, paramecia and amoebas, which are protists that lack cell walls, have contractile vacuoles. This vesicle collects excess water from the cell and pumps it out, keeping the cell from bursting as it takes on water from its environment.
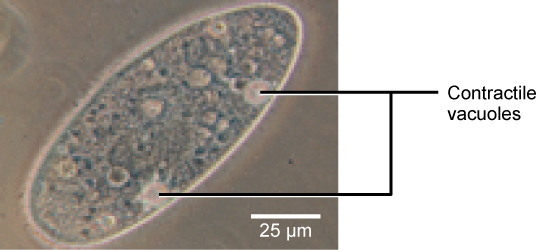
A paramecium’s contractile vacuole, here visualized using bright field light microscopy at 480x magnification, continuously pumps water out of the organism’s body to keep it from bursting in a hypotonic medium. (credit: modification of work by NIH; scale-bar data from Matt Russell)
Many marine invertebrates have internal salt levels matched to their environments, making them isotonic with the water in which they live. Fish, however, must spend approximately five percent of their metabolic energy maintaining osmotic homeostasis. Freshwater fish live in an environment that is hypotonic to their cells. These fish actively take in salt through their gills and excrete diluted urine to rid themselves of excess water. Saltwater fish live in the reverse environment, which is hypertonic to their cells, and they secrete salt through their gills and excrete highly concentrated urine.
In vertebrates, the kidneys regulate the amount of water in the body. Osmoreceptors are specialized cells in the brain that monitor the concentration of solutes in the blood. If the levels of solutes increase beyond a certain range, a hormone is released that retards water loss through the kidney and dilutes the blood to safer levels. Animals also have high concentrations of albumin, which is produced by the liver, in their blood. This protein is too large to pass easily through plasma membranes and is a major factor in controlling the osmotic pressures applied to tissues.


