1.14: Cashing in on Redox
- Page ID
- 8236
Metabolism in Bis2A
Cellular metabolism represents roughly 1/3 of the Bis2A curriculum. While this may seem like a lot, we cover very little of what a classic course in metabolism (like BIS103) will cover and an even smaller (really minuscule) fraction of the metabolism that occurs on the planet. What we do cover, however, is very important foundational knowledge. You will learn about some common chemical transformations that are associated with the transformation of life's molecular building blocks and about different core modes of energy transfer that you will encounter often in biology. The energy story and the design challenges introduced earlier will become increasingly important in these next few modules and beyond.
Metabolism Modules
- Here you will be introduced to an important concept called reduction potential and you will be given the opportunity to use a redox tower. There is also a discussion on redox chemistry in your discussion manual. Make sure to use both resources.
- Here you will be introduced to a second two major player in metabolism, NADH. You will be expected to recognize NADH's structure if shown on an exam.
- The metabolic pathway glycolysis has been covered in detail. Keep in mind that we want you to be able to look at any an illustration of this process, including its individual reactions, and tell us an energy story of that reaction. By no means should you spend time trying to memorize these pathways (though it will help tremendously to remember some big picture things - these will be stressed). Sometimes we will give you the pathway as a figure on the exams. Glycolysis ultimately produces 2 ATP via a process called substrate level phosphorylation, 2 NADH and 2 pyruvate compounds.
- The TCA cycle provides opportunities to create multiple examples of energy stories. The TCA cycle will also produce more ATP, NADH and completely oxidize glucose into CO2.
- We considered an alternative pathway to that of the TCA cycle, fermentation. In fermentation for the first time we saw NADH used as a reactant in a metabolic reaction.
- In our module on respiration, we will follow NADH to the end of its journey, as it donates its electrons to the electron transport chain (ETC). In this module you will need to be able to use a redox tower. The ETC produces a proton gradient. No ATP is directly generated in this process. However, the proton gradient is then used by the cell (among other things) to run an enzyme called ATP synthase which catalyzes the reaction ADP + Pi --> ATP. This method of ATP production (called oxidative respiration) results in additional- many additional- ATPs being produced. Substrate level phosphorylation produces the minority of ATPs generated when glucose is processed via respiration: glycolysis, pyruvate oxidation, the TCA cycle, and formation of ATP via the ETC and ATP synthase.
- And finally, in another module, we will go through the process of photosynthesis.
Reduction/Oxidation Reactions
Brief Overview of Redox Chemistry and the Redox Tower
An oxidation-reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two compounds or atoms. For example, the transfer of an electron from a neutral sodium (Na) to neutral chlorine (Cl) resulting in a positively charged sodium (Na+) ion and negatively charged chloride (Cl-) ion is a redox reaction. An oxidation reaction strips an electron from an atom in a compound, and the addition of this electron to another compound is a reduction reaction. Because oxidation and reduction usually occur together, these pairs of reactions are called oxidation reduction reactions, or redox reactions. Redox reactions are common and vital to some of the basic functions of life, including photosynthesis, respiration, combustion, and corrosion or rusting.
Every red/ox reaction can be thought of as 2 half-reactions, in one half reaction a compound loses electrons and in the second half-reaction a different compound gains electrons. The amount of energy transferred in a redox reaction is associated with the difference between each half reactions' reduction potential, E0'. The electron tower is a tool that ranks different common half reactions based on how likely they are to donate or accept electrons. The lower, more negative, the electrochemical potential for each half reaction, the less likely the compound is to "accept" an electron. Usually, reduced compounds can "donate" electrons to oxidized compounds that have higher reduction potentials. Generally, oxidized compounds can "accept" electrons from reduced compounds that have lower reduction potentials. The use of the electron tower will be more evident as we discuss electron transport chains in a few modules.
Note: Quick discussion
Sometimes a redox tower will list compounds in order of decreasing redox potentials (high values on top and low values on the bottom). Our towers do not- we list (reduced vs. oxidized state) molecule pairs with negative values (highly negative E˚') up top and positive ones (highly positive E˚') towards the bottom. Does presenting the data this way change the redox potential of a compound?
Reduction-Oxidation Reactions
In this class we are going to focus on REDOX reactions that are biologically associated. The majority of the reactions we discuss occur in the context of metabolic pathways (connected sets of metabolic reactions) where compounds may be consumed by the cell, broken down into smaller parts and then reassembled into larger macromolecules.
Lets start with some Generic Reactions
Transferring electrons between two compounds results in one of these compounds loosing an electron, and one of the compounds gaining an electron. For example, look at the figure below. If we use the energy story rubric to look at the overall reaction we can compare the before and after characteristics of the reactants and products. What happens to the matter (stuff) before and after the reaction? Compound A starts as neutral and becomes positively charged. Compound B starts as neutral and becomes negatively charged. Because electrons are negatively charged, we can follow the movement of electrons from compound A to B by looking at the change in charge. A looses an electron (becoming positively charged), and in so doing we say that A has become oxidized. Oxidation is associated with the loss of electron(s). B gains the electron (becoming negatively charged), and we say that B has become reduced. Reduction is associated with gain of electrons. We also know, since something happened that energy must have been either transferred and/or reorganized in this process and we'll consider this shortly.
In Bis2A we expect you to become familiar with this terminology. Try to learn it and learn to use it as soon as possible - we will use the terms frequently.

When an electron(s) is lost, or a molecule is oxidized, the electron(s) must then passed to another molecule. The molecule gaining the electron is said to be reduced. The oxidation and reduction reactions are always paired in what is known as an oxidation-reduction reaction (also called a redox reaction).
Remember the Definitions:
Oxidation of A = A has lost electrons
Reduction of B = B has gained electrons
The Half Reaction
To formalize our common understanding of redox reactions, we introduce the concept of the half reaction. Two half reactions are required to make the full redox reaction. Each half reaction can be thought of as a description of what happens to one of the two molecules involved in the redox reaction. This is illustrated below. In this example compound AH is being oxidized by compound B+; electrons are moving from AH to B+ to generate A+ and BH. Each reaction can be thought of as two half reactions: Where AH is being oxidized and a second reaction where B+ is being reduced.
AH2 --> A + 2H+ + 2e- (half rx 1)
B + 2H+ + 2e- --> BH2 (half rx 2)
AH2 + B --> A + BH2 (sum of the two half rx, a redox reaction, here involving a 2 electron transfer)
Note: Possible discussion
If you consider a generic redox reaction and reflect back on the thermodynamic lectures, what factor will determine whether a redox reaction will proceed in a particular direction spontaneously, and what might determine its rate?
Reduction Potential
By convention we analyze and describe redox reactions with respect to reduction potentials (E0'), a term that quantitatively describes the "ability" of a compound to gain electrons. This value of the reduction potential is determined experimentally. We can anthropomorphize the reduction potential by saying that it is related to the strength with which a compound can “attract” or “pull” or “capture” electrons. Not surprisingly this is is related to but not identical to electronegativity.
Different compounds, based on their structure and atomic composition, have intrinsic and distinct attractions for electrons. This quality is termed reduction potential or E0’ and is a relative quantity (relative by comparison to some “standard” reaction). If a test compound has a stronger "attraction" to electrons than the standard (if the two competed, the test compound would "take" electrons from the standard compound), we say that the test compound has a positive reduction potential whose magnitude is proportional to how much more strongly it can attract and keep electrons than the standard compound. The relative strength of the compound in comparison to the standard is measured and reported in units of Volts (V) or milliVolts (mV).
Note: Possible discussion
Rephrase for yourself: How do you describe or think about the difference between the concept of electronegativity and reduction potential?
Another: What is a volt? Why does reduction potential have the units of volts?
The Redox Tower
All kinds of compounds can participate in redox reactions. A tool has been developed to graphically tabulate redox half reactions based on their E0' values and to help us predict the direction of electron flow between potential electron donors and acceptors. Whether a particular compound will act as an electron donor (reductant, reducing agent) or electron acceptor (oxidant, oxidizing agent) depends on what other compound it is interacting with. The electron tower usually ranks a variety of common compounds (their half reactions) from the most negative E0', compounds that readily get rid of electrons, to the most positive E0', compounds most likely to accept electrons. In addition, each half reaction is written by convention with the oxidized form on the left/followed by the reduced form on the right of the slash.
For example the half reaction for the reduction of NAD+ to NADH is written: NAD+/NADH. In the tower below, the number of electrons that are transferred is also listed. For example the reduction of NAD+ to NADH involves two electrons, written in the table as "2e-".
An electron tower is shown below.
Important study hint:
When you see an electron tower, it's very helpful to orient yourself by looking for O2 . By now you should know- without having to pause to think!- that O2 is a very strong oxidizing agent- that electrons will gravitate towards it. Finding O2 on the tower will help you remember that compounds with lower, more negative, reduction potential are written above O2 in our BIS2A towers (this may not be true for your Chem class tower- it might be flipped- it's the same data, just written in a different order). It'll also remind you that the oxidized compound is written first, and the reduced compound (here water) second. Note also that the table informs you that the splitting of a single water molecule to "form" half an O2 is a 2 electron transfer. This location at the bottom of our table reminds you that compounds at the bottom of the table are strong oxidizing agents and their reduced form (like water) is a weak reducing agent.

For a short video on how to use the electron tower in red/ox problems click here. This video was made by Dr. Easlon for Bis2A students and is very helpful.
What is the relationship between ΔE0' and ΔG?
The question now becomes: how do we know if any given redox reaction is energetically spontaneous or not (exergonic vs. endergonic) and can we calculate that free energy difference? Yes, we can, as the 2 values are related via the following equation:

here n is the number of electrons involved in each transfer, and F is a constant that is a positive number . Note also that once again we, as biologists, are using tables of E˚', not the chemists' more typical E˚. The difference in the reduction potential for the reaction (or ∆E0' for the reaction), is the difference between the E0' for the oxidant (the compound getting the electrons, the final reduction potential of those electrons) and for the reductant (the compound losing the electrons, the original reduction potential of those electrons). In our generic example below, AH is the reductant and B+ is the oxidant: electrons are moving from AH to B+. This happens to be a 2 electron transfer. Using an E0' of -0.32 for the reductant (we got this value from a redox tower of reduction potentials of imaginary compounds) and a strong E0' of 0.82 for the oxidant, the total change in E0' or ΔE0' is 1.14 V*. If a mole of AH reduced a mole of B+, an entire 2 moles of electrons would have been allowed to drop over a voltage of 1.14 V! Using the Nerst equation essentially corrects for the number of electrons per transfer (here n = 2) and puts things into units biologists can use, and copes with the directionality (sign) for us. F has units of kJ*/volt, E has units of (Volts), so we end up with kJ, a unit of energy. This is the energy that is (in this case) released when a mole of AH reduces a mole of B+.
*Are you having issues with getting to this number? Be sure you can do this on your own. Try some practice examples. Remember that to calculate the change in anything over time, we are subtracting the original state from the final state, so... 0.82 - (-0.32) = +1.14 V

Generic redox reaction with half reactions written with reduction potential (E0') of the two half reactions indicated. One of these E˚' values needs to be flipped- which one, and why?
Keep in mind that a positive ΔE0' gives you a negative ΔG. The reaction #1 written above is written with the arrow in the right direction- it is are exergonic, with a negative ∆G.
*F is the Faraday constant of 96.485 kJ/V. Sometimes it is given in units of kcal/V which is 23.062 kcal/V, which is the amount of energy (in kJ or kcal) released when one mole of electrons passes through a potential drop of 1 volt
Note
Again, what you should notice is that ΔG and ΔE have an inverse relationship: When ΔG is positive, ΔE is negative and when ΔG is negative, ΔE is positive. For additional review see the redox discussion in the Bis2A Discussion Manual.
Introduction to Mobile Energy Carriers
Section Summary
Energy is moved around and transferred within the cell in a variety of ways. One critical mechanism that Nature has evolved is the use of recyclable molecular energy carriers. While there are several major recyclable energy carriers, they all share some common functional features:
Properties of Key Cellular Molecular Energy Carriers
- We think of the energy carriers as existing in "pools" of available carriers. One could, by analogy, consider these mobile energy carriers analogous to the delivery vehicles of parcel carriers - the company has a certain "pool" of available vehicles at any one time to pickup and make deliveries.
- Each individual carrier in the pool can exist in one of multiple distinct states: it is either carrying a "load" of energy, a fractional load, or is "empty". The molecule can interconvert between "loaded" and empty and thus can be recycled. Again by analogy, the delivery vehicles can be either carrying packages or be empty and switch between these states.
- The balance or ratio in the pool between "loaded" and "unloaded" carriers is important for cellular function, is regulated by the cell and can often tell us something about the state of a cell. Likewise, a parcel carrier service keeps close tabs on how full or empty their delivery vehicles are - if they are too full there may be insufficient "empty" trucks to pick up new packages; if they are too empty, business must not be going well or it is shut down; there is an appropriate balance for different situations.
In this course we will examine two major types of molecular recyclable energy carriers: (1) nicotinamide adenine dinucleotide (NAD+), a close relative nicotinamide adenine dinucleotide phosphate (NADP+), and flavin adenine dinucleotide (FAD2+) and (2) nucleotide mono-, di- and triphosphates, with particular attention paid to adenosine triphosphate (ATP). Each of these two types of molecules is involved in energy transfer that involve different classes of chemical reactions. It's interesting that while one class of carrier delivers electrons (and energy), the other delivers phosphates (and energy), but both include an adenine nucleotide(s). Perhaps adenine nucleotides were an important part of early life? Check out the "RNA world" theory.
Redox Chemistry and Electron Carriers
The oxidation of, or removal of an electron from, a molecule (whether accompanied with the removal of an accompanying proton or not) results in a change of free energy for that molecule - matter, internal energy, and entropy have all changed in the process. Likewise the reduction of (the gain of electron on) a molecule also changes its free energy. The magnitude of change in free energy and its direction (positive or negative) for a redox reaction dictates the spontaneity of the reaction and how much energy is transferred. In biological systems, where a great deal of energy transfer happens via redox reactions, it is important to understand how these reactions are mediated and begin to start considering ideas or hypotheses for why these reactions are mediated in many cases by a small family of electron carriers.
Note
Possible discussion: Relate the burning of cellulose (a sugar polymer) with the last paragraph above. What does that demonstration have to do with our upcoming discussion on redox carriers. There is some mention above already - can you find it?
Note
Possible discussion: The problem alluded to in the previous discussion question is a great place to start bringing in the design challenge rubric. If you recall, the first step of the rubric asks that you define a problem or question. In this case let's imagine that there is a problem to define for which the mobile electron carriers below helped Nature solve.
***--- Remember evolution DOES NOT forward engineer solutions to problems, but in retrospect we can use our imagination and logic to infer that what we see preserved by natural selection provided a selective advantage because the natural innovation "solved" a problem that limited success. ---***
Design Challenge for Redox Carriers
- What was a problem(s) that the evolution of mobile electron/redox carriers helped solve?
- The next step of the design challenge asks you to identify criteria for successful solutions. What are criteria for success in the problem you've identified?
- Step 3 in the design challenge ask you to identify possible solutions. Well here Nature has identified some for us - we consider three in the reading below. It looks like Nature is happy to have multiple solutions to the problem.
- The penultimate step of the design challenge rubric asks you to evaluate the proposed solutions against the criteria for success. This should make you think/discuss about why there are multiple different electron carriers? Are there different criteria for success? Are they each solving slightly different problems? What do you think? Be on the lookout as we go through metabolism for clues.
NAD+/H and FADH/H2
In living systems, a small class of compounds function as electron shuttles: They bind and carry electrons between compounds in different metabolic pathways. The principal electron carriers we will consider are derived from the B vitamin group and are derivatives of nucleotides. These compounds can be both reduced (that is, they accept electrons) or oxidized (they lose electrons) depending on the reduction potential of a potential electron donor or acceptor that they might transfer electrons to and from. Nicotinamide adenine dinucleotide (NAD+) (the structure is shown below) is derived from vitamin B3, niacin. NAD+ is the oxidized form of the molecule; NADH is the reduced form of the molecule after it has accepted two electrons and a proton (which together are the equivalent of a hydrogen atom with an extra electron).
We are expecting you to know which is the oxidized and which is the reduced form of NAD+/NADH, and be able to recognize either form on-the-spot in the context of a chemical reaction.
NAD+ can accept electrons from an organic molecule according to the general equation:

A bit of vocabulary review: When electrons are added to a compound, the compound is said to have been reduced. A compound that reduces another (donates electrons) is called a reducing agent. In the above equation, RH is a reducing agent, and NAD+ is reduced to NADH. When electrons are removed from a compound, it becomes oxidized. A compound that oxidizes another is called an oxidizing agent. In the above equation, NAD+ is an oxidizing agent, and RH is oxidized to R.
You need to get this down! We will (a) test specifically on your ability to do so - as "easy" questions and (b) we will use the terms with the expectation that you know what they mean and can relate them to biochemical reactions correctly (in class and on tests).
You will also encounter a second variation of NAD+, NADP+. It is structurally very similar to NAD+ but it contains an extra phosphate group and plays an important role in anabolic reactions such as photosynthesis. Another nucleotide-based electron carrier that you will also encounter in this course and beyond, flavin adenine dinucleotide (FAD+) is derived from vitamin B2, also called riboflavin. Its reduced form is FADH2. Learn to recognize these molecules as electron carriers as well.

The oxidized form of the electron carrier (NAD+) is shown on the left and the reduced form (NADH) is shown on the right. The nitrogenous base in NADH has one more hydrogen ion and two more electrons than in NAD+.
NAD+ is used by the cell to "pull" electrons off of compounds and to carry them to other locations within the cell, thus they are called electron carriers. NAD+/H compounds are used in many of the metabolic processes we will discuss in this class. For example, in its oxidized form NAD+ is used as a reactant in glycolysis and the TCA cycle, whereas in its reduced form (NADH) it is a reactant in fermentation and the electron transport chain (ETC). Each of these processes will be discussed in later modules.
Energy Story for a Redox Reaction
***As a rule of thumb, when we see NAD+/H as a reactant or product we know we are looking at a redox reaction.***
When NADH is a product and NAD+ is a reactant we know that NAD+ has become reduced (forming NADH) therefore the other reactant must have been the electron donor and become oxidized. The vice versa is also true. If NADH has become NAD+, then the other reactant must have gained electrons from NADH and become reduced.
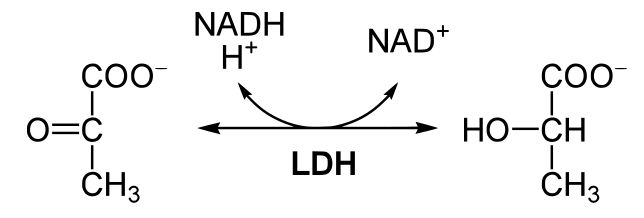
This reaction shows the conversion of pyruvate to lactic acid coupled with the conversion of NADH to NAD+. Source: en.wikibooks.org/wiki/Structural_Biochemistry/Enzyme/sequential_reactions
In the figure above we see the reaction of pyruvate becoming lactic acid, coupled with the conversion of NADH into NAD+. This reaction is catalyzed by LDH. Using our 'rule of thumb' above, we categorize this reaction as a redox reaction. NADH is the reduced form of the electron carrier and NADH is converted into NAD+. This half of the reaction results in the oxidation of the electron carrier. Pyruvate is converted into lactic acid in this reaction. Both of these sugars are negatively charged so it would be difficult to see which compound is more reduced using the charges of the compounds. However, we know that pyruvate has become reduced to form lactic acid because this conversion is coupled to the oxidation of NADH into NAD+. But how can we tell that lactic acid is more reduced than pyruvate? The answer is to look at the carbon-hydrogen bonds in both compounds. As electrons are transferred, they are often accompanied by a hydrogen atom. There are a total of 3 C-H bonds in pyruvate and there are a total of 4 C-H bonds in lactic acid. When we compare these two compounds in the before and after state, we see that lactic acid has one more C-H bond, therefore, lactic acid is more reduced than pyruvate. Note also lactic acid it has picked up 2 complete hydrogen atoms (and the source of these was....?). This holds true for multiple compounds. For example, the figure below, you should be able to rank the compounds from most to least reduced using the C-H bonds as your guide.

Above are a series of compounds than can be ranked or reorganized from most to least reduced. Compare the number of C-H bonds in each compound. Carbon dioxide has no C-H bonds and is the most oxidized form of carbon we will discuss in this class. Answer: Most reduced is methane (compound 3), then methanol (4), formaldehyde (1), carboxylic acid (2), and finally carbon dioxide (5).

This reaction shows the conversion of G3P, Pi, NAD+ into NADH and 1,3-BPG. This reaction is catalyzed by Glyceraldehyde-3-phosphate dehydrogenase.
Energy story for the reaction catalyzed by Glyceraldehyde-3-phosphate dehydrogenase:
Let's make an energy story for the reaction above.
First, let's characterize the reactants and products. The reactants are Glyceraldehyde-3-phosphate (a carbon compound), Pi (inorganic phosphate) and NAD+. These three reactants enter into a chemical reaction to produce two products, NADH and 1,3-Bisphosphoglycerate. If you look closely you can see that the 1,3-BPG contains two phosphates. This is important when we are double checking that no mass has been lost. There are two phosphates in the reactants so there need to be two phosphates in the products (conservation of mass!). You can double check that all the other atoms are also accounted for. The enzyme that catalyzes this reaction is called Glyceraldehyde-3-phosphate dehydrogenase. The standard free energy change of this reaction is ~6.3 kJ/mol so under standard conditions we can say that the free energy of the products is higher than that of the reactants and that this reaction is not spontaneous under standard conditions.
What can we say about this reaction catalyzed by glyceraldehyde-3-phosphate dehydrogenase?
This is a redox reaction. We know that because we have produced a reduced electron carrier (NADH) as a product and NAD+ is a reactant. Where did the electron come from to make NADH? The electron must have come from the other reactant (the carbon compound).
Note
Recommended discussion: We will spend some time examining the reaction catalyzed by glyceraldehyde-3-phosphate dehydrogenase in more detail as we move through the lectures and text. The first thing to discuss here is that the figure above is a highly simplified or condensed version of the steps that take place - one could in fact break that reaction above into TWO conceptual reactions. Can you imagine what those two "subreactions" might be? Discuss amongst yourselves.
Note
Recommended discussion: The text above notes that the standard change in free energy for this complex reaction is ~+6.3 kJ/mol. Under standard conditions this reaction is NOT spontaneous. However, this is one of the key reactions in the oxidation of glucose. It needs to GO in the cell. The questions are: why is it important to note things like "standard change of free energy" or "under standard conditions" when reporting that ΔG°? What could possibly be going on in the cell to make what is under standard conditions an endergonic reaction "go" under real-life conditions?

