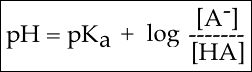2020_Winter_Bis2a_Facciotti_Lecture_04
- Page ID
- 27180
Learning Objectives Associated with 2020_Winter_Bis2a_Facciotti_Lecture_04
|
pKa
The most common acid we will talk about in BIS2A is the carboxylic acid functional group. These acids are weak acids, meaning that they only partially dissociate (into H+ cations and RCOO-
Note that the key difference in the figure below between a strong acid or base and a weak acid or base is the single arrow (strong) versus a double arrow (weak). In the
- both forms of the conjugate acid or base (that is what we call the molecule that "holds" the proton - i.e. CH3OOH and CH3OO-, respectively in the figure) are present at the same time and
- the ratio of those two quantities can change easily by moving the reaction in either direction.
Figure 1. An example of strong acids and strong bases in their protonation and
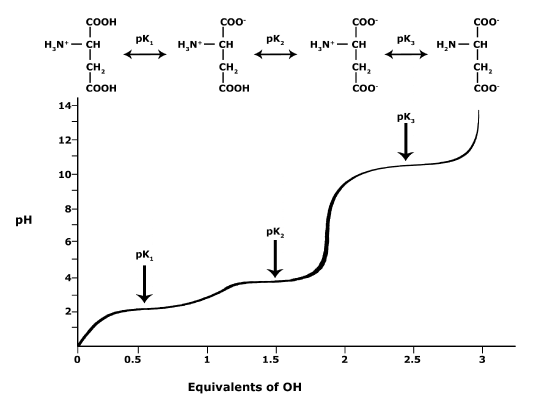
In Bis2A you we ask you to relate pH and pKa to each other when discussing the protonation state of an acid or base, for example, in amino acids. How can we use the information given in this module to answer the question: Will the functional groups on the amino acid Glutamate be
To answer this question, we need to create a relationship between pH and pKa. The relationship between pKa and pH
Figure 2. The Henderson-Hasselbach equation
A solution to this equation
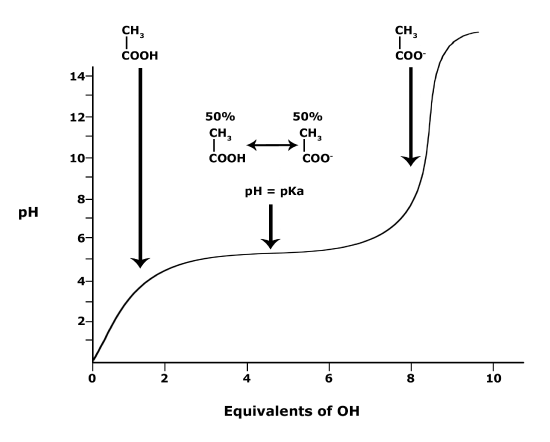
Figure 3. This graph depicts the protonation state of acetic acid as the pH changes. At a pH below the pKa, the acid is
In BIS2A, we will look at the protonation state and
Figure 4. This graph depicts the protonation state of glutamate as the pH changes. At a pH below the pKa for each functional group on the amino acid, the functional group is
Attribution: Ivy Jose
Possible NB Discussion  Point
Point
What is the overall charge of free Glutamate at a pH of 5? What is the overall charge of free Glutamate at a pH of 10? In as much detail as you can, using the terms you've learned thus far in this course, describe the chemical change of glutamate’s predominant form as the pH changes from 5 to 10.
Lipids
Lipids are a diverse group of hydrophobic compounds that include molecules like fats, oils, waxes, phospholipids, and steroids. Most lipids are at their core hydrocarbons, molecules that include many nonpolar carbon-carbon or carbon-hydrogen bonds. The abundance of nonpolar functional groups give lipids a degree of hydrophobic (
Fats and oils
A common fat molecule or triglyceride. These types of molecules are hydrophobic and, while they have many functions, are probably best known for their roles in body fat and plant oils. A triglyceride molecule derived from two types of molecular components—a polar "head" group and a nonpolar "tail" group. The "head" group of a triglyceride
The models of the triglycerides shown above depict the relative positions of the atoms in the molecule. If you Google for images of

Natural fats like butter, canola oil, etc.,
- The number of carbons in the hydrocarbon chains;
- The number of desaturations, or double bonds, in the hydrocarbon chains.
The first factor influences how these molecules interact with each other and with water, while the second factor dramatically influences their shape. Introducing a double bond causes a "kink" in the otherwise relatively "straight" hydrocarbon, depicted in a slightly exaggerated was in Figure 3.
Based on what you can understand from this brief description, are you able to explain why butter is solid at room temperature while vegetable oil is liquid?
Here is an additional piece of information that could help you answer question: butter has a greater percentage of longer and saturated hydrocarbons in its triglycerides than does vegetable oil.

Real-Life connection:
Ever thought about how lipids are important to vision? Read more here.
Sterols
Steroids are lipids with a fused ring structure. Although they do not resemble the other lipids discussed here,
| Cholesterol | Cortisol |
Phospholipids
Phospholipids are major constituents of the cell membrane, the outermost layer of cells. Like fats,
Note
Note in Figure 5 that the phosphate group has an R group linked to one of the oxygen atoms. R is a variable commonly used in these types of diagrams to show some other atom or molecule bound at that position. That part of the molecule can be different in different phospholipids—and will impart some different chemistry to the whole molecule. At the moment, however, you are
| 1,2-Dioleoyl-sn-glycero-3-phospho-L-serine |
In the presence of water, some phospholipids will spontaneously arrange themselves into a micelle (Figure 6). The lipids arrange such that their polar groups are on the outside of the micelle, and the nonpolar tails are on the inside. Under other conditions, a lipid bilayer can also form. This structure, only a few nanometers thick,
As mentioned above, if you were to take some pure phospholipids and drop them into water that some phospholipid would spontaneously form into micelles. This sounds like a process that an Energy Story could describe. Go back to the Energy Story rubric and see if you can try to create an Energy Story for this process — I expect that the steps involving the description of energy might be difficult at this point (we'll come back to that later) but you should be able to do at least the first three steps.
We discuss the phospholipid membrane in a later module. It is important to remember the chemical properties associated with the functional groups in the phospholipid to understand the function of the cell membrane.
Carbohydrates
Carbohydrates are one of the four main classes of macromolecules that make up all cells and are an essential part of our diet; grains, fruits, and vegetables are all natural sources. While we may be most familiar with the role carbohydrates play in nutrition, they also have a variety of other essential functions in humans, animals, plants, and bacteria. In this section, we will discuss and review basic concepts of carbohydrate structure and nomenclature, and a variety of functions they play in cells.
Molecular structures
In their simplest form, carbohydrates can be represented by the stoichiometric formula (CH2O
Nomenclature
One issue with carbohydrate chemistry is the nomenclature. Here are a few quick and simple rules:
- Simple carbohydrates, such as glucose, lactose, or dextrose, end with an "-ose."
Simple carbohydrates can be classified based on the number of carbon atoms in the molecule, as with triose (three carbons), pentose (five carbons), or hexose (six carbons).Simple carbohydrates can be classified based on the functional group found in the molecule,i.e ketose (contains a ketone) or aldose (contains an aldehyde).- Polysaccharides
are often organized by the number of sugar molecules in the chain, such as in a monosaccharide, disaccharide, or trisaccharide.
For a short video on carbohydrate classification, see the 10-minute Khan Academy video by clicking here.
Monosaccharides
Monosaccharides ("mono-" = one; "

Figure 1. Monosaccharides are classified based on the position of their carbonyl group and the number of carbons in the backbone. Aldoses have a carbonyl group (
Glucose versus galactose
Galactose (part of
Fructose versus both glucose and galactose
In glucose and galactose, the carbonyl group is on the C1 carbon, forming an aldehyde group. In fructose, the carbonyl group is on the C2 carbon, forming a ketone group. The former sugars
Figure 2. Glucose, galactose, and fructose are all hexoses. They are structural isomers, meaning they have the same chemical formula (C6H12O6) but a different arrangement of atoms.
Linear versus ring form of the monosaccharides
Monosaccharides can exist as a linear chain or as ring-shaped molecules. In aqueous solutions, monosaccharides are usually found in ring form (Figure 3). Glucose in a ring form can have two different arrangements of the hydroxyl group (OH) around the anomeric carbon (C1 that becomes asymmetric in the process of ring formation). If the hydroxyl group is below C1 in the sugar, it is said to be in the alpha (α) position, and if it is above C1 in the sugar, it is said to be in the beta (β) position.
Figure 3. Five- and six-carbon monosaccharides exist in equilibrium between linear and ring form. When the ring forms, the side chain it closes on is locked into an α or β position. Fructose and ribose also form rings, although they form five-membered rings as opposed to the six-membered ring of glucose.
Disaccharides
Disaccharides ("di-" = two) form when two monosaccharides undergo a dehydration reaction (also known as a condensation reaction or dehydration synthesis). During this process, the hydroxyl group of one monosaccharide combines with the hydrogen of another monosaccharide, releasing a molecule of water and forming a covalent bond. A covalent bond formed between a carbohydrate molecule and another molecule (in this case, between two monosaccharides) is known as a glycosidic bond. Glycosidic bonds (also called glycosidic linkages) can be of the alpha or the beta type.
Figure 4. Sucrose is formed when a monomer of glucose and a monomer of fructose are joined in a dehydration reaction to form a glycosidic bond. In the process, a water molecule is lost. By convention, the carbon atoms in a monosaccharide are numbered from the terminal carbon closest to the carbonyl group. In sucrose, a glycosidic linkage is formed between the C1 carbon in glucose and the C2 carbon in fructose.
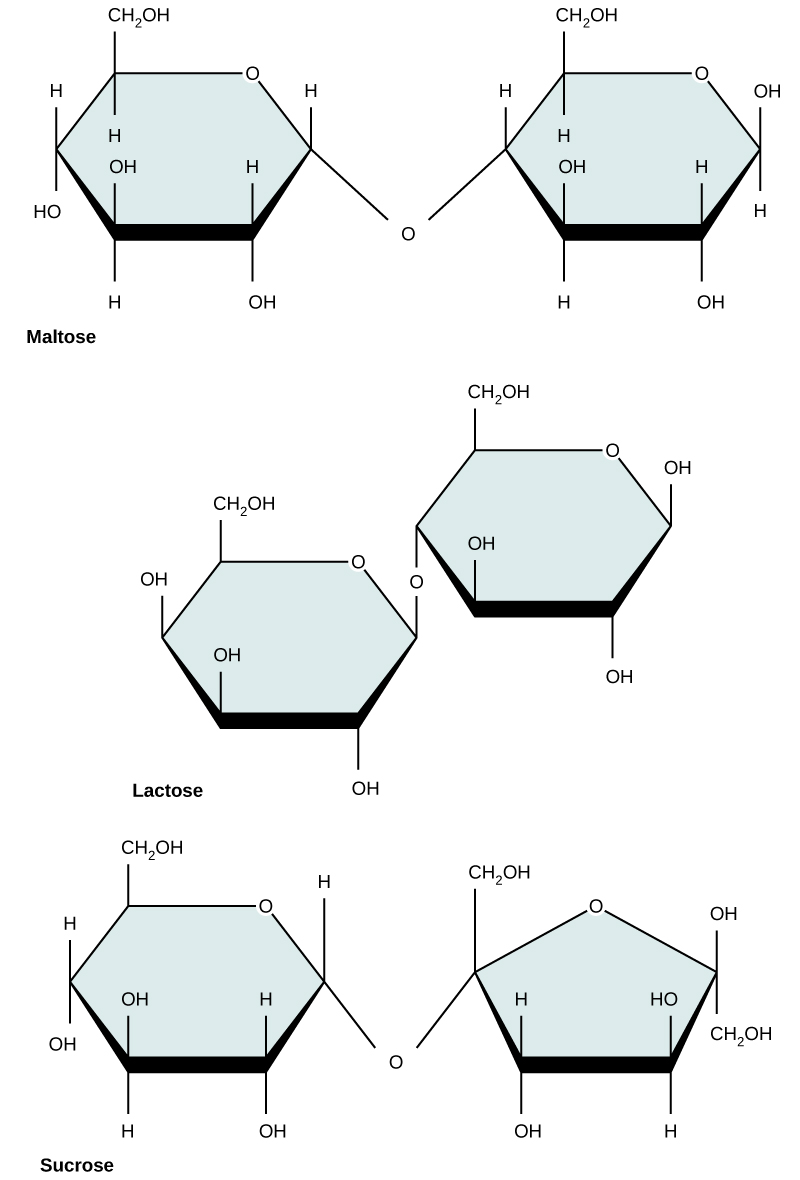
Common disaccharides include lactose, maltose, and sucrose (Figure 5). Lactose is a disaccharide consisting of the monomers glucose and galactose. It is found naturally in milk. Maltose, or malt/grain sugar, is a disaccharide formed by a dehydration reaction between two glucose molecules. The most common disaccharide is sucrose, or table sugar, which is composed of the monomers glucose and fructose.
Figure 5. Common disaccharides include maltose (grain sugar), lactose (milk sugar), and sucrose (table sugar).
| Sucrose | Lactose | Maltose |
Polysaccharides
A long chain of monosaccharides linked by glycosidic bonds is known as a polysaccharide ("poly-" = many). The chain may be branched or unbranched, and it may contain different types of monosaccharides. The molecular weight may be 100,000 Daltons or more, depending on the number of monomers joined. Starch, glycogen, cellulose, and chitin are primary examples of polysaccharides.
Starch is the stored form of sugars in plants and is made up of a mixture of amylose and amylopectin; both are polymers of glucose. Plants are able to synthesize glucose. Excess glucose, the amount synthesized that is beyond the plant’s immediate energy needs, is stored as starch in different plant parts, including roots and seeds. The starch in the seeds provides food for the embryo as it germinates and can also act as a source of food for humans and animals who may eat the seed. Starch that is consumed by humans is broken down by enzymes, such as salivary amylases, into smaller molecules, such as maltose and glucose.
Starch is made up of glucose monomers that are joined by 1-4 or 1-6 glycosidic bonds; the numbers 1-4 and 1-6 refer to the carbon number of the two residues that have joined to form the bond. As illustrated in Figure 6, amylose is starch formed by unbranched chains of glucose monomers (only 1-4 linkages), whereas amylopectin is a branched polysaccharide (1-6 linkages at the branch points).
Figure 6. Amylose and amylopectin are two different forms of starch. Amylose is composed of unbranched chains of glucose monomers connected by 1-4 glycosidic linkages. Amylopectin is composed of branched chains of glucose monomers connected by 1-4 and 1-6 glycosidic linkages. Because of the way the subunits are joined, the glucose chains have a helical structure. Glycogen (not shown) is similar in structure to amylopectin but more highly branched.
Glycogen
Glycogen is a common stored form of glucose in humans and other vertebrates. Glycogen is the animal equivalent of starch and is a highly branched molecule usually stored in liver and muscle cells. Whenever blood glucose levels decrease, glycogen is broken down to release glucose in a process known as glycogenolysis.
| Glycogen |
Cellulose
Cellulose is the most abundant natural biopolymer. The cell wall of plants is mostly made of cellulose, which provides structural support to the cell. Wood and paper are mostly cellulosic in nature. Cellulose is made up of glucose monomers that are linked by β 1-4 glycosidic bonds.
Figure 7. In cellulose, glucose monomers are linked in unbranched chains by β 1-4 glycosidic linkages. Because of the way the glucose subunits are joined, every glucose monomer is flipped relative to the next one, resulting in a linear, fibrous structure.
As shown in the figure above, every other glucose monomer in cellulose is flipped over, and the monomers are packed tightly as extended, long chains. This gives cellulose its rigidity and high tensile strength—which is so important to plant cells. While the β 1-4 linkage cannot be broken down by human digestive enzymes, herbivores such as cows, koalas, buffalos, and horses are able, with the help of the specialized flora in their stomach, to digest plant material that is rich in cellulose and use it as a food source. In these animals, certain species of bacteria and protists reside in the rumen (part of the digestive system of herbivores) and secrete the enzyme cellulase. The appendix of grazing animals also contains bacteria that digest cellulose, giving it an important role in the digestive systems of ruminants. Cellulases can break down cellulose into glucose monomers that can be used as an energy source by the animal. Termites are also able to break down cellulose because of the presence of other organisms in their bodies that secrete cellulases.
Interactions with carbohydrates
We have just discussed the various types and structures of carbohydrates found in biology. The next thing to address is how these compounds interact with other compounds. The answer to that is that it depends on the final structure of the carbohydrate. Because carbohydrates have many hydroxyl groups associated with the molecule, they are therefore excellent H-bond donors and acceptors. Monosaccharides can quickly and easily form H-bonds with water and are readily soluble. All of those H-bonds also make them quite "sticky". This is also true for many disaccharides and many short-chain polymers. Longer polymers may not be readily soluble.
Finally, the ability to form a variety of H-bonds allows polymers of carbohydrates or polysaccharides to form strong intramolecular and intermolocular bonds. In a polymer, because there are so many H-bonds, this can provide a lot of strength to the molecule or molecular complex, especially if the polymers interact. Just think of cellulose, a polymer of glucose, if you have any doubts.
Possible NB Discussion  Point
Point
Lipids and carbohydrates are not just classes of macromolecules that we discuss in BIS 2A but are also two of the essential macronutrients that we can obtain from eating various foods. Some popularized diet programs (e.g., Atkins, ketogenic) suggest limiting carbohydrates and/or fats. As you learn more about biomolecules and their roles in living systems, are you refining your perspective on foods and diets? What have you learned so far? Do you think there is anything missing in your understanding? Are you able to better understand and evaluate certain diets, such as the aforementioned?