Traditionally, when cellular levels of a protein were known to change in response to a chemical effector, molecular studies focused on control of the transcription of its gene. These studies often revealed that the control of gene expression was at the level of transcription, turning a gene on or off through interactions of transcription factors with DNA. However, protein levels are also controlled post-transcriptionally, by regulating the rate of mRNA translation or degradation. Studies of transcriptional and posttranscriptional regulation mechanisms are seminal to our understanding of how the correct protein is made in the right amounts at the right time.
We may have suspected, but now know that control of gene expression and cellular responses can be more complex than increasing or decreasing the transcription of a single gene or translation of a single protein. Whole genome sequences and new techniques make possible the study of the expression of virtually all genes in a cell at the same time, a field of investigation called genomics. Genomic studies reveal networks of regulated genes that must be understood to more fully explain the developmental and physiological changes in an organism. When you can ‘see’ all of the RNAs being transcribed from active genes in a cell, you are looking at a cell’s transcriptome. By analogy to genomics, transcriptomics defines studies of ‘webs’ of interactive RNAs. Again, by analogy to genomics and transcriptomics, the broad study of active and inactive proteins in cells or tissues, how they are modified (processed) before use and how they interact is called proteomics. The technologies applied to proteomic studies include protein microarrays, immunochemical techniques and others uniquely suited to protein analysis (click Proteomics Techniques-Wikipedia for more information). Protein Microarrays are increasingly being used to identify protein-protein interactions, as well as the different states of proteins under different cellular conditions. Read even more about these exciting developments and their impact on basic and clinical research at Protein Microarrays from ncbi.
Finally think about this: creating a proteomic library analogous to a genomic library would seem a daunting prospect. But efforts are underway. Check out A stab at mapping the Human Proteome for original research leading to the sampling of a tissue-specific human proteome, and click Strategies for Approaching the Proteome for more general information. Let’s look at some uses of DNA microarrays. This technology involves ‘spotting’ DNA (e.g., cloned DNA from a genomic or cDNA library, PCR products, oligonucleotides…) on a glass slide, or chip. In the language of microarray analysis, the slides are the probes. Spotting a chip is a robotic process. Because the DNA spots are microscopic, a cellspecific transcriptome (cDNA library) can fit on a single chip. A small genome microarray might also fit on a single chip, while larger genomes might need several slides. A primary use of DNA microarrays is transcriptional profiling. A genomic microarray can probe a mixture of fluorescently tagged target cDNAs made from mRNAs, in order to identify many (if not all) of the genes expressed in the cells at a given moment (i.e., its transcriptome). cDNA microarray probes can also probe quantitative differences in gene expression in cells or tissues during normal differentiation or in response to chemical signals. They are also valuable for genotyping, (i.e. characterizing the genes in an organism). Microarrays are so sensitive that they can even distinguish between two genes or regions of DNA that differ by a single nucleotide. Click Single Nucleotide Polymorphisms, or SNPs to learn more. In the microarray below, each colored spot (red, yellow, green) is a different fluorescently tagged molecule hybridizing to target sequences on the microarray. In the fluorescence microscope, the spots fluoresce different colors in response to UV light.
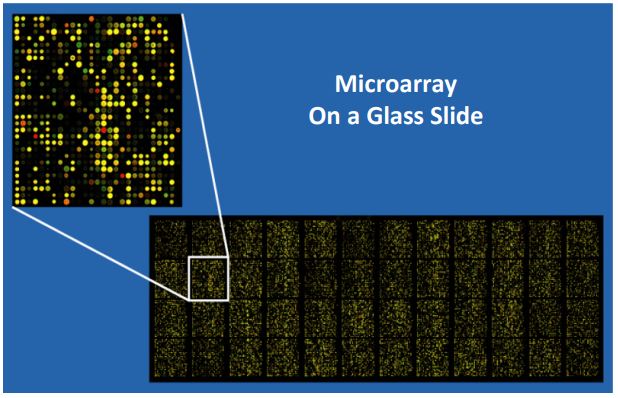
With quantitative microarray methods, the brightness (intensity) of the signal from each probe can be measured. In this way, we can compare the relative amounts of cDNA (and thus, different RNAs) in the transcriptome of different tissues or resulting from different tissue treatments. A table of different applications of microarrays (adapted from Wikipedia) is shown on the next page.
| Application of Technology |
Synopsis |
| Gene Expression Profiling |
In a transcription (mRNA or gene expression) profiling experiment the expression levels of thousands of genes are simultaneously monitored to study the effects of certain treatments, diseases, and developmental stages on gene expression. |
| Comparative genomic hybridization |
Assessing genome content in different cells or closely related organisms, where one organism’s genome is the probe for a target genome from a different species. |
| GeneID |
Small microarrays to check IDs of organisms in food and feed for genetically modified organisms (GMOs), mycoplasmas in cell culture, or pathogens for disease detection. These detection protocols often combine PCR and microarray technology. |
| CHIP; Chromatin immunoprecipitation |
DNA sequences bound to a particular protein can be isolated by immunoprecipitating the protein. The fragments can be hybridized to a microarray (such as a tiling array) allowing the determination of protein binding site occupancy throughout the genome. |
| DamID |
Analogously to ChIP, genomic regions bound by a protein of interest can be isolated and used to probe a microarray to determine binding site occupancy. Unlike ChIP, DamID does not require antibodies but makes use of adenine methylation near the protein's binding sites to selectively amplify those regions, introduced by expressing minute amounts of protein of interest fused to bacterial DNA adenine methyltransferase. |
| SNP detection |
Identifying single nucleotide polymorphism among alleles within or between populations. Some microarray applications make use of SNP detection, including Genotyping, forensic analysis, measuring predisposition to disease, identifying drug-candidates, evaluating germline mutations in individuals or somatic mutations in cancers, assessing loss of heterozygosity, or genetic linkage analysis. |
| Alternative splicing protection |
An exon junction array design uses probes specific to the expected or potential splice sites of predicted exons for a gene. It is of intermediate density, or coverage, to a typical gene expression array (with 1-3 probes per gene) and a genomic tiling array (with hundreds or thousands of probes per gene). It is used to assay the expression of alternative splice forms of a gene. Exon arrays have a different design, employing probes designed to detect each individual exon for known or predicted genes, and can be used for detecting different splicing isoforms |
| Tiling array |
Genome tiling arrays consist of overlapping probes designed to densely represent a genomic region of interest, sometimes as large as an entire human chromosome. The purpose is to empirically detect expression of transcripts or alternatively spliced forms which may not have been previously known or predicted. |
The Power of Microarrays. https://youtu.be/88rzbpclscM
If you like world records, check out the salamander with the largest genome, 10X bigger than our own: The HUGE Axolotl Genome. What do they do with all that DNA? And can our current technologies figure it out? For the original report, click on the following link: here.


