19.4: Materials and Procedures
- Page ID
- 40277
Materials
- Student Stock Cultures
- Glass bowls with Sanisol
- Glass Slides
- Stain trays
- Water bottles (partially fill with DI water)
- Slide holder or clothespin
- Gram Stain kit
- Lens cleaner/lens cleaning kits
- Streptococcus mutans culture
Procedures
Suggested bacteria for staining, however, your group should perform stains on and share as many different organisms as possible (all from Student Stock Collection, except Streptococcusmutans—1 broth culture per table):
- Moraxella catarrhalis
- Streptococcusmutans
- Escherichia coli
- Bacillus subtilis
- Staphylococcus saprophyticus
- Mycobacterium smegmatis
One person at each table should do a mixed smear of E. coli and S. saprophyticus which should be observed by everyone. This combination will help you distinguish the colors of G+ versus G-.
Refer to Figure \(\PageIndex{1}\) as you prepare and while you stain your slides.
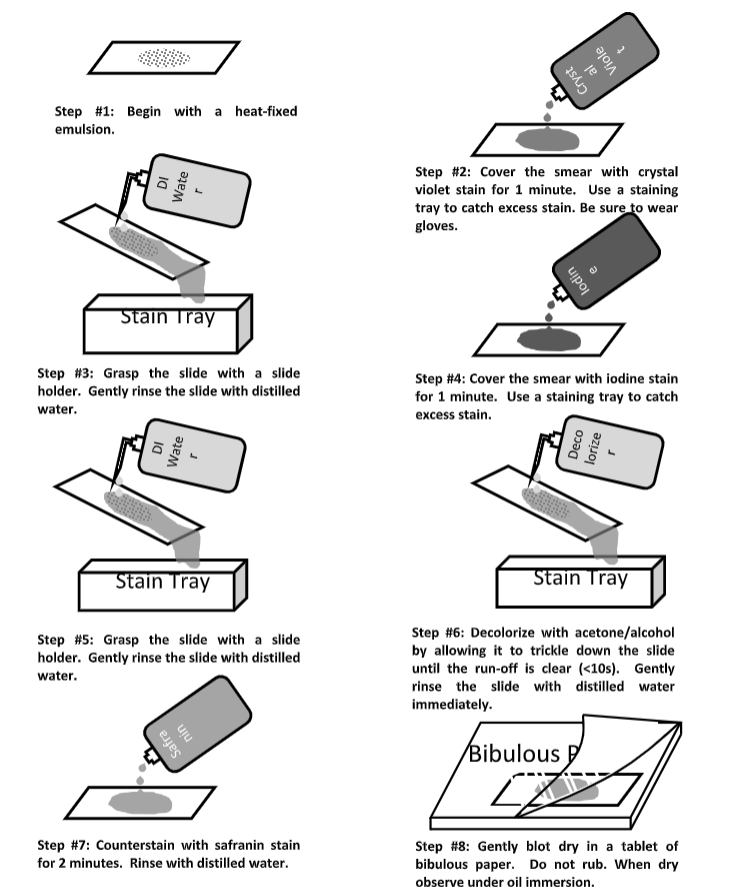
- Prepare an air-dried, heat-fixed smear. Tip: Be very careful to prepare a thin smear. A smear that is too thick will not stain correctly! This can cause a fair amount of grief and frustration when evaluating your stain, and may cause you to miss-identify the gram reaction. Prepare several slides at a time.
- Cover the prepared slide with Crystal Violet for 1 minute.
- Gently, but completely, rinse the slide with DI water.
- Cover the slide with Gram’s Iodine for 1 minute.
- Gently, but completely, rinse the slide with DI water.
- Decolorize the slide with Ethanol/Acetone. Slowly DRIP the decolorizer down the slide until it runs clear of the slide. This should not take more than about 10 seconds. Do not flush the slide with a lot of the ethanol/acetone as this may over decolorize your specimen and cause you to misinterpret the results.
- Immediately after decolorizing, gently rinse the slide with DI water.
- Counterstain with the secondary stain, Safranin for 2 minutes.
- Gently, but completely, rinse the slide with DI water.
- Blot slide gently with bibulous paper. Tap gently; do not rub the paper as this may smear your prep.
- Observe at under oil immersion, 100x objective.
Cautions
- Thick smear-
- Over heat fixing-
- Under decolorizing-
- Over decolorizing-
- Old cultures-
- Gram variable bacteria-
- Acid-Fast bacteria-
Results
- Draw and color (or indicate colors of) the bacteria you Gram stained:

- Fill in the table with the organisms you stained, including the mixed slide.
|
Organism |
Color of cells |
Gram Reaction(+ or -) |
Cell Morphology |
Cell Arrangement |
Size (\(\mu m\)) |
|---|---|---|---|---|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contributors and Attributions
Kelly C. Burke (College of the Canyons)


