10.4: Materials and Procedures
- Page ID
- 40214
Materials
2 Groups per Table:
- 5mL test tube culture of E. coli
- 2- 9.9 mL Water Blanks (Yellow Dots)
- 3- 9.0 mL Water Blanks
- 4- TSA plates (1 plate for ea. DF 10-5 – 10-8)
- 1 mL pipettes
- 4- Sterile yellow spreaders -1 pkg (dispose into pipette container on your bench)
Procedures
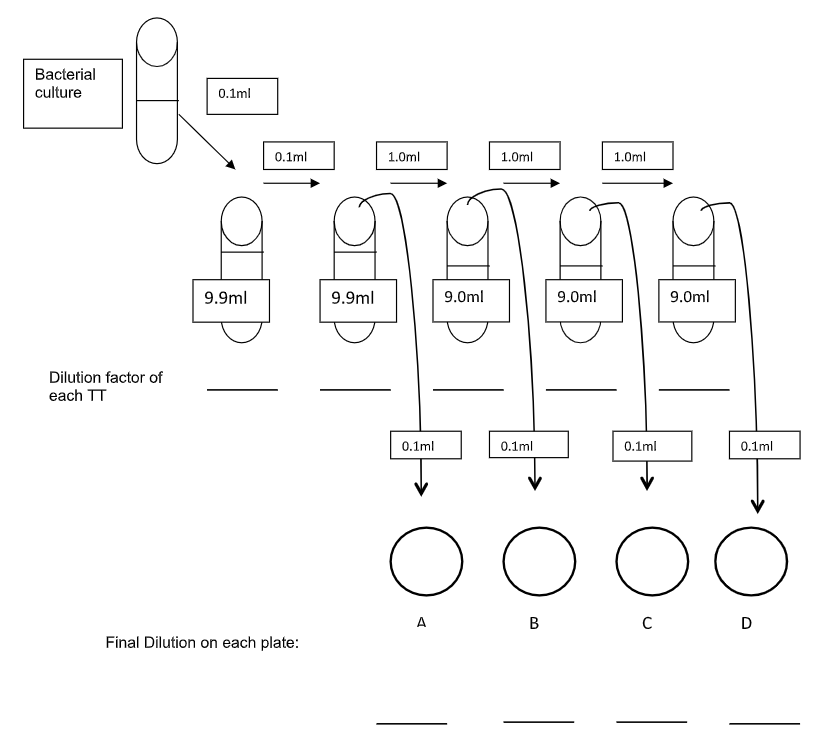
The Standard Plate Count- Refer to the diagram below. The first thing your group should do is gather all the materials needed and work through the diagram until you are confident you know how to perform the dilution series. Then make a plan and begin the procedure. This will save you time in the long run, and will go a long way to prevent mistakes and poor results.
You will use the spread plate technique. First practice the technique with a non-sterile spreader and petri plate. After transferring the dilution sample to the plate, with one hand, open the petri plate, keeping the lid over the plate. With the other hand move the spreader back and forth across the plate while turning the plate at the same time. Spread and turn several times in order to evenly distribute the sample over the entire plate. Close the plate, let it dry, and then invert and incubate. Dispose of the spreader as instructed.
- Mix the tube of E. coli well.
- Use a sterile 1.0ml pipette to transfer 0.1ml of the E. coli culture to the first 9.9ml dilution tube. Mix well. Discard pipette.
- Use a new sterile 1.0ml pipette to transfer 0.1ml from the first dilution tube to the second 9.9ml dilution tube. Mix well. Discard pipette.
- Use a new sterile 1.0ml pipette to remove 1.0ml from the second tube to the third tube, this time a 9.0ml tube. Mix well. Discard pipette. Repeat for the 4th and 5th tubes, mixing each tube along the way.
Now begin to plate your samples. Dispense and spread the sample onto the plates one at a time. Do not dispense the samples and then go back to spread them. The plate will absorb the liquid and thus the sample will not be able to be spread across the plate. This will result in most of the bacteria clumping in the middle of the plate and accurate counts will not be achieved. One person can dispense the sample and another can spread the sample.
- From the third test tube transfer 0.1ml, with a sterile 1.0ml pipette, to the first plate A. Spread the sample.
- From the fourth test tube transfer 0.1ml, with a sterile 1.0ml pipette, to the second plate B. Spread the sample.
- From the fifth test tube transfer 0.1ml, with a sterile 1.0ml pipette, to the third plate C. Spread the sample.
- From the sixth test tube transfer 0.1ml, with a sterile 1.0ml pipette, to the last plate D. Spread the sample.
Keep your dilutions, and your original E.c. broth for the next part of the lab.
- Set the plates aside to allow the liquid to be absorbed. Invert and incubate.
Results:
- After incubation determine which plate is the countable plate (30-300 CFU). There should only be one, correct? Enter the information in the chart below.
Plate counts and determination of original cell density:
|
Plate |
A |
B |
C |
D |
|---|---|---|---|---|
|
DF of plate |
|
|
|
|
|
# of Colonies* |
|
|
|
|
|
Original cell Density** |
|
|
|
|
* Give the colony count on each plate or indicate if plates are TFTC or TNTC
**Calculate based on the countable plate
- Explain your results and/or issues that you had.
Contributors and Attributions
Kelly C. Burke (College of the Canyons)


