7.18: Regulation of Cellular Respiration - Regulatory Mechanisms for Cellular Respiration
- Page ID
- 13191
- Explain the mechanisms that regulate cellular respiration.
Regulatory Mechanisms
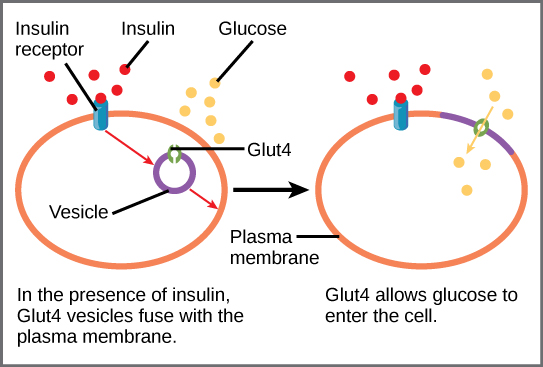
Various mechanisms are used to control cellular respiration. As such, some type of control exists at each stage of glucose metabolism. Access of glucose to the cell can be regulated using the GLUT proteins that transport glucose. In addition, different forms of the GLUT protein control passage of glucose into the cells of specific tissues.
Some reactions are controlled by having two different enzymes: one each for the two directions of a reversible reaction. Reactions that are catalyzed by only one enzyme can go to equilibrium, stalling the reaction. In contrast, if two different enzymes (each specific for a given direction) are necessary for a reversible reaction, the opportunity to control the rate of the reaction increases and equilibrium is not reached.
A number of enzymes involved in each of the pathways (in particular, the enzyme catalyzing the first committed reaction of the pathway) are controlled by attachment of a molecule to an allosteric (non-active) site on the protein. This site has an effect on the enzyme’s activity, often by changing the conformation of the protein. The molecules most commonly used in this capacity are the nucleotides ATP, ADP, AMP, NAD+, and NADH. These regulators, known as allosteric effectors, may increase or decrease enzyme activity, depending on the prevailing conditions, altering the steric structure of the enzyme, usually affecting the configuration of the active site. This alteration of the protein’s (the enzyme’s) structure either increases or decreases its affinity for its substrate, with the effect of increasing or decreasing the rate of the reaction. The attachment of a molecule to the allosteric site serves to send a signal to the enzyme, providing feedback. This feedback type of control is effective as long as the chemical affecting it is bound to the enzyme. Once the overall concentration of the chemical decreases, it will diffuse away from the protein, and the control is relaxed.
Key Points
- Varying forms of the GLUT protein control the passage of glucose into the cells of specific tissues, thereby regulating cellular respiration.
- Reactions that are catalyzed by only one enzyme can go to equilibrium, which can cause the reaction to stall.
- If two different enzymes are necessary for a reversible reaction, there is greater opportunity to control the rate of the reaction and, as a result, equilibrium is reached less often.
- Enzymes are often controlled by binding of a molecule to an allosteric site on the protein.
Key Terms
- enzyme: a globular protein that catalyses a biological chemical reaction
- allosteric: a compound that binds to an inactive site, affecting the activity of an enzyme by changing the conformation of the protein (can activate or deactivate)
- metabolism: the complete set of chemical reactions that occur in living cells


