7.13: Oxidative Phosphorylation - ATP Yield
- Page ID
- 13155
ese atoms were originally part of a glucose molecule. At the end of the pathway, the electrons are used to reduce an oxygen molecule to oxygen ions. The extra electrons on the oxygen attract hydrogen ions (protons) from the surrounding medium and water is formed.
ATP Yield
The amount of energy (as ATP) gained from glucose catabolism varies across species and depends on other related cellular processes.
LEARNING OBJECTIVES
Describe the origins of variability in the amount of ATP that is produced per molecule of glucose consumed
KEY TAKEAWAYS
Key Points
- While glucose catabolism always produces energy, the amount of energy (in terms of ATP equivalents) produced can vary, especially across different species.
- The number of hydrogen ions the electron transport chain complexes can pump through the membrane varies between species.
- NAD+ provides more ATP than FAD+ in the electron transport chain and can lead to variance in ATP production.
- The use of intermediates from glucose catabolism in other biosynthetic pathways, such as amino acid synthesis, can lower the yield of ATP.
Key Terms
- catabolism: Destructive metabolism, usually including the release of energy and breakdown of materials.
ATP Yield
In a eukaryotic cell, the process of cellular respiration can metabolize one molecule of glucose into 30 to 32 ATP. The process of glycolysis only produces two ATP, while all the rest are produced during the electron transport chain. Clearly, the electron transport chain is vastly more efficient, but it can only be carried out in the presence of oxygen.
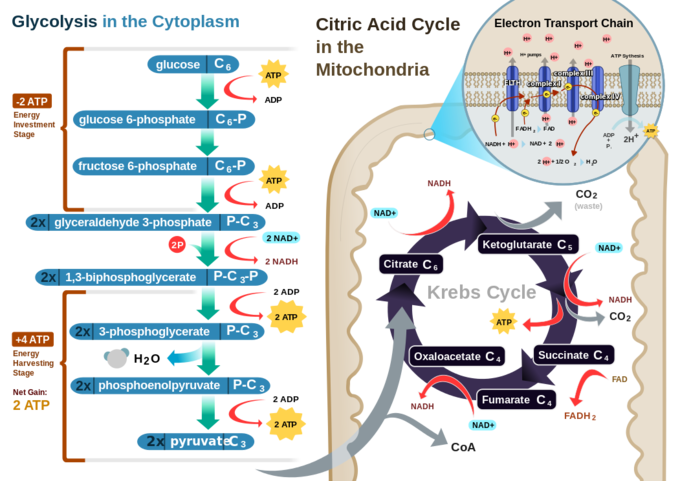
The number of ATP molecules generated via the catabolism of glucose can vary substantially. For example, the number of hydrogen ions the electron transport chain complexes can pump through the membrane varies between species. Another source of variance occurs during the shuttle of electrons across the membranes of the mitochondria. The NADH generated from glycolysis cannot easily enter mitochondria. Thus, electrons are picked up on the inside of mitochondria by either NAD+ or FAD+. These FAD+ molecules can transport fewer ions; consequently, fewer ATP molecules are generated when FAD+ acts as a carrier. NAD+ is used as the electron transporter in the liver, and FAD+ acts in the brain.
Another factor that affects the yield of ATP molecules generated from glucose is the fact that intermediate compounds in these pathways are used for other purposes. Glucose catabolism connects with the pathways that build or break down all other biochemical compounds in cells, but the result is not always ideal. For example, sugars other than glucose are fed into the glycolytic pathway for energy extraction. Moreover, the five-carbon sugars that form nucleic acids are made from intermediates in glycolysis. Certain nonessential amino acids can be made from intermediates of both glycolysis and the citric acid cycle. Lipids, such as cholesterol and triglycerides, are also made from intermediates in these pathways, and both amino acids and triglycerides are broken down for energy through these pathways. Overall, in living systems, these pathways of glucose catabolism extract about 34 percent of the energy contained in glucose.


