15.8: RNA Processing in Eukaryotes - mRNA Processing
- Page ID
- 13307
Eukaryotic pre-mRNA receives a 5′ cap and a 3′ poly (A) tail before introns are removed and the mRNA is considered ready for translation.
- Outline the steps of pre-mRNA processing
Key Points
- A 7-methylguanosine cap is added to the 5′ end of the pre-mRNA while elongation is still in progress. The 5′ cap protects the nascent mRNA from degradation and assists in ribosome binding during translation.
- A poly (A) tail is added to the 3′ end of the pre-mRNA once elongation is complete. The poly (A) tail protects the mRNA from degradation, aids in the export of the mature mRNA to the cytoplasm, and is involved in binding proteins involved in initiating translation.
- Introns are removed from the pre-mRNA before the mRNA is exported to the cytoplasm.
Key Terms
- intron: a portion of a split gene that is included in pre-RNA transcripts but is removed during RNA processing and rapidly degraded
- moiety: a specific segment of a molecule
- spliceosome: a dynamic complex of RNA and protein subunits that removes introns from precursor mRNA
Pre-mRNA Processing
The eukaryotic pre-mRNA undergoes extensive processing before it is ready to be translated. The additional steps involved in eukaryotic mRNA maturation create a molecule with a much longer half-life than a prokaryotic mRNA. Eukaryotic mRNAs last for several hours, whereas the typical E. coli mRNA lasts no more than five seconds.
Pre-mRNAs are first coated in RNA-stabilizing proteins; these protect the pre-mRNA from degradation while it is processed and exported out of the nucleus. The three most important steps of pre-mRNA processing are the addition of stabilizing and signaling factors at the 5′ and 3′ ends of the molecule, and the removal of intervening sequences that do not specify the appropriate amino acids. In rare cases, the mRNA transcript can be “edited” after it is transcribed.
5′ Capping
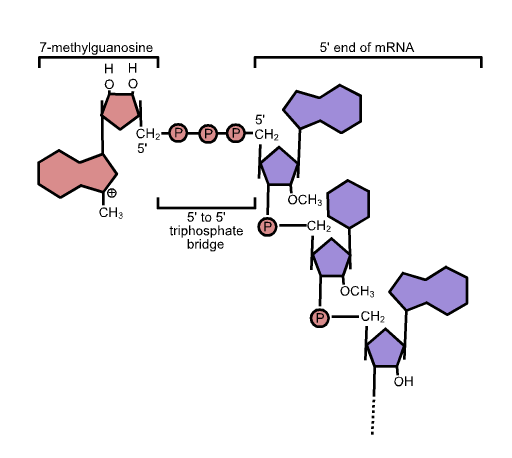
While the pre-mRNA is still being synthesized, a 7-methylguanosine cap is added to the 5′ end of the growing transcript by a 5′-to-5′ phosphate linkage. This moiety protects the nascent mRNA from degradation. In addition, initiation factors involved in protein synthesis recognize the cap to help initiate translation by ribosomes.
3′ Poly-A Tail
While RNA Polymerase II is still transcribing downstream of the proper end of a gene, the pre-mRNA is cleaved by an endonuclease-containing protein complex between an AAUAAA consensus sequence and a GU-rich sequence. This releases the functional pre-mRNA from the rest of the transcript, which is still attached to the RNA Polymerase. An enzyme called poly (A) polymerase (PAP) is part of the same protein complex that cleaves the pre-mRNA and it immediately adds a string of approximately 200 A nucleotides, called the poly (A) tail, to the 3′ end of the just-cleaved pre-mRNA. The poly (A) tail protects the mRNA from degradation, aids in the export of the mature mRNA to the cytoplasm, and is involved in binding proteins involved in initiating translation.

Pre-mRNA Splicing
Eukaryotic genes are composed of exons, which correspond to protein-coding sequences (ex-on signifies that they are expressed), and intervening sequences called introns (int-ron denotes their intervening role), which may be involved in gene regulation, but are removed from the pre-mRNA during processing. Intron sequences in mRNA do not encode functional proteins.
Discovery of Introns
The discovery of introns came as a surprise to researchers in the 1970s who expected that pre-mRNAs would specify protein sequences without further processing, as they had observed in prokaryotes. The genes of higher eukaryotes very often contain one or more introns. While these regions may correspond to regulatory sequences, the biological significance of having many introns or having very long introns in a gene is unclear. It is possible that introns slow down gene expression because it takes longer to transcribe pre-mRNAs with lots of introns. Alternatively, introns may be nonfunctional sequence remnants left over from the fusion of ancient genes throughout evolution. This is supported by the fact that separate exons often encode separate protein subunits or domains. For the most part, the sequences of introns can be mutated without ultimately affecting the protein product.
Intron Processing
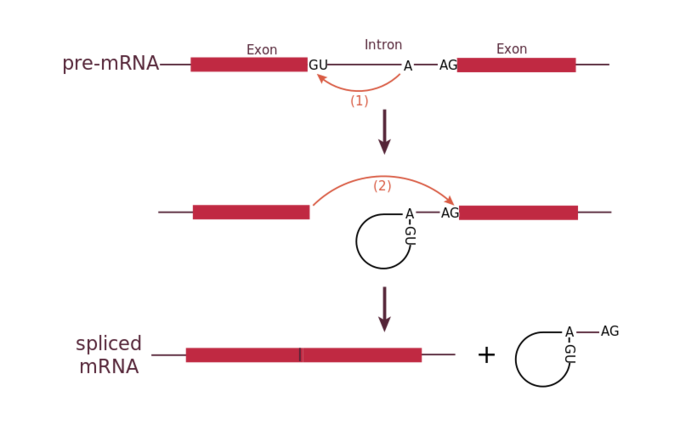
All introns in a pre-mRNA must be completely and precisely removed before protein synthesis. If the process errs by even a single nucleotide, the reading frame of the rejoined exons would shift, and the resulting protein would be dysfunctional. The process of removing introns and reconnecting exons is called splicing. Introns are removed and degraded while the pre-mRNA is still in the nucleus. Splicing occurs by a sequence-specific mechanism that ensures introns will be removed and exons rejoined with the accuracy and precision of a single nucleotide. The splicing of pre-mRNAs is conducted by complexes of proteins and RNA molecules called spliceosomes.
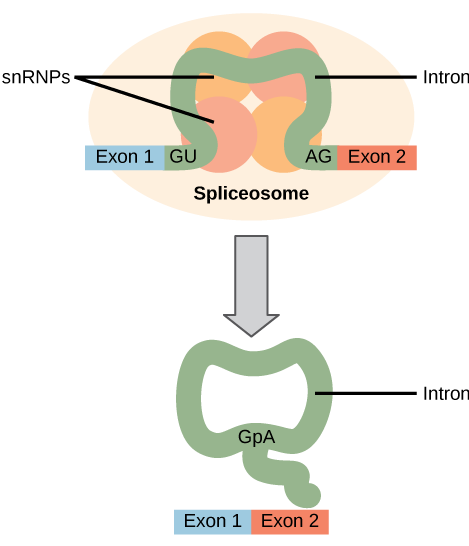
Each spliceosome is composed of five subunits called snRNPs (for small nuclear ribonucleoparticles, and pronounced “snurps”.) Each snRNP is itself a complex of proteins and a special type of RNA found only in the nucleus called snRNAs (small nuclear RNAs). Spliceosomes recognize sequences at the 5′ end of the intron because introns always start with the nucleotides GU and they recognize sequences at the 3′ end of the intron because they always end with the nucleotides AG. The spliceosome cleaves the pre-mRNA’s sugar phosphate backbone at the G that starts the intron and then covalently attaches that G to an internal A nucleotide within the intron. Then the spliceosme connects the 3′ end of the first exon to the 5′ end of the following exon, cleaving the 3′ end of the intron in the process. This results in the splicing together of the two exons and the release of the intron in a lariat form.