3.23: Diffusion, Active Transport and Membrane Channels
- Page ID
- 4070
Transport Across Cell Membranes
All cells acquire the molecules and ions they need from their surrounding extracellular fluid (ECF). There is an unceasing traffic of molecules and ions in and out of the cell through its plasma membrane (Examples: glucose, \(Na^+\), \(Ca^{2+}\)). In eukaryotic cells, there is also transport in and out of membrane-bounded intracellular compartments such as the nucleus, endoplasmic reticulum, and mitochondria (Examples: proteins, mRNA, \(Ca^{2+}\), and ATP).
The following problems can occur during transport:
1. Relative concentrations
Molecules and ions move spontaneously down their concentration gradient (i.e., from a region of higher to a region of lower concentration) by diffusion. Molecules and ions can be moved against their concentration gradient, but this process, called active transport, requires the expenditure of energy (usually from ATP).
2. Lipid bilayers are impermeable to most essential molecules and ions.
The lipid bilayer is permeable to water molecules and a few other small, uncharged, molecules like oxygen (O2) and carbon dioxide (CO2). These diffuse freely in and out of the cell. The diffusion of water through the plasma membrane is of such importance to the cell that it is given a special name - osmosis. Lipid bilayers are not permeable to ions such as K+, Na+, Ca2+ (called cations because when subjected to an electric field they migrate toward the cathode [the negatively-charged electrode]) and Cl-, HCO3- (called anions because they migrate toward the anode [the positively-charged electrode]). They are also not permeable to small hydrophilic molecules like glucose and macromolecules like proteins and RNA. The cells solve the problem of transporting ions and small molecules across their membranes with the help of the following two mechanisms:
- Facilitated diffusion: Transmembrane proteins create a water-filled pore through which ions and some small hydrophilic molecules can pass by diffusion. The channels can be opened (or closed) according to the needs of the cell.
- Active transport: Transmembrane proteins, called transporters, use the energy of ATP to force ions or small molecules through the membrane against their concentration gradient.
Facilitated Diffusion of Ions
Facilitated diffusion of ions takes place through proteins, or assemblies of proteins, embedded in the plasma membrane. These transmembrane proteins form a water-filled channel through which the ion can pass down its concentration gradient. The transmembrane channels that permit facilitated diffusion can be opened or closed. They are said to be "gated"; some types of gated ion channels:
- ligand-gated
- mechanically-gated
- voltage-gated
- light-gated
Ligand-gated ion channels
Many ion channels open or close in response to binding a small signaling molecule or "ligand". Some ion channels are gated by extracellular ligands; some by intracellular ligands. In both cases, the ligand is not the substance that is transported when the channel opens.
External ligands
External ligands (shown here in green) bind to a site on the extracellular side of the channel.
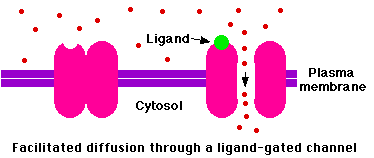
Examples:
- Acetylcholine (ACh). The binding of the neurotransmitter acetylcholine at certain synapses opens channels that admit Na+ and initiate a nerve impulse or muscle contraction.
- Gamma amino butyric acid (GABA). Binding of GABA at certain synapses — designated GABAA — in the central nervous system admits Cl- ions into the cell and inhibits the creation of a nerve impulse
Internal ligands
Internal ligands bind to a site on the channel protein exposed to the cytosol. Examples:
- "Second messengers", like cyclic AMP (cAMP) and cyclic GMP (cGMP), regulate channels involved in the initiation of impulses in neurons responding to odors and light respectively.
- ATP is needed to open the channel that allows chloride (Cl-) and bicarbonate (HCO3-) ions out of the cell. This channel is defective in patients with cystic fibrosis. Although the energy liberated by the hydrolysis of ATP is needed to open the channel, this is not an example of active transport; the ions diffuse through the open channel following their concentration gradient.
Mechanically-gated ion channels
Sound waves bending the cilia-like projections on the hair cells of the inner ear open up ion channels leading to the creation of nerve impulses that the brain interprets as sound. Mechanical deformation of the cells of stretch receptors opens ion channels leading to the creation of nerve impulses.
Voltage-gated ion channels
In so-called "excitable" cells like neurons and muscle cells, some channels open or close in response to changes in the charge (measured in volts) across the plasma membrane. For example, as an impulse passes down a neuron, the reduction in the voltage opens sodium channels in the adjacent portion of the membrane. This allows the influx of \(Na^+\) into the neuron and thus the continuation of the nerve impulse. Some 7000 sodium ions pass through each channel during the brief period (about 1 millisecond) that it remains open. This was learned by use of the patch clamp technique.
The Patch Clamp Technique
The properties of ion channels can be studied by means of the patch clamp technique. A very fine pipette (with an opening of about 0.5 µm) is pressed against the plasma membrane of either an intact cell or the plasma membrane can be pulled away from the cell and the preparation placed in a test solution of desired composition. Current flow through a single ion channel can then be measured.
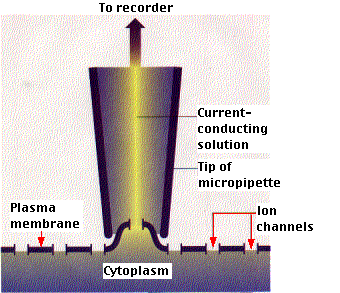
Such measurements reveal that each channel is either fully open or fully closed; that is, facilitated diffusion through a single channel is "all-or-none". This technique has provided so much valuable information about ion channels that its inventors, Erwin Neher and Bert Sakmann, were awarded a Nobel Prize in 1991.
Facilitated Diffusion of Molecules
Some small, hydrophilic organic molecules, like sugars, can pass through cell membranes by facilitated diffusion. Once again, the process requires transmembrane proteins. In some cases, these — like ion channels — form water-filled pores that enable the molecule to pass in (or out) of the membrane following its concentration gradient.
Example:
Maltoporin. This homotrimer in the outer membrane of E. coli forms pores that allow the disaccharide maltose and a few related molecules to diffuse into the cell.
Example: The plasma membrane of human red blood cells contain transmembrane proteins that permit the diffusion of glucose from the blood into the cell.
Note that in all cases of facilitated diffusion through channels, the channels are selective; that is, the structure of the protein admits only certain types of molecules through. Whether all cases of facilitated diffusion of small molecules use channels is yet to be proven. Perhaps some molecules are passed through the membrane by a conformational change in the shape of the transmembrane protein when it binds the molecule to be transported.
In either case, the interaction between the molecule being transported and its transporter resembles in many ways the interaction between an enzyme and its substrate.
Active Transport
Active transport is the pumping of molecules or ions through a membrane against their concentration gradient. It requires a transmembrane protein (usually a complex of them) called a transporter and energy. The source of this energy is ATP.
The energy of ATP may be used directly or indirectly.
- Direct Active Transport. Some transporters bind ATP directly and use the energy of its hydrolysis to drive active transport.
- Indirect Active Transport. Other transporters use the energy already stored in the gradient of a directly-pumped ion. Direct active transport of the ion establishes a concentration gradient. When this is relieved by facilitated diffusion, the energy released can be harnessed to the pumping of some other ion or molecule.
Direct Active Transport
The Na+/K+ ATPase
The cytosol of animal cells contains a concentration of potassium ions (K+) as much as 20 times higher than that in the extracellular fluid. Conversely, the extracellular fluid contains a concentration of sodium ions (Na+) as much as 10 times greater than that within the cell. These concentration gradients are established by the active transport of both ions. And, in fact, the same transporter, called the Na+/K+ ATPase, does both jobs. It uses the energy from the hydrolysis of ATP to
- actively transport 3 Na+ ions out of the cell
- for each 2 K+ ions pumped into the cell.
This accomplishes several vital functions:
- It helps establish a net charge across the plasma membrane with the interior of the cell being negatively charged with respect to the exterior. This resting potential prepares nerve and muscle cells for the propagation of action potentials leading to nerve impulses and muscle contraction.
- The accumulation of sodium ions outside of the cell draws water out of the cell and thus enables it to maintain osmotic balance (otherwise it would swell and burst from the inward diffusion of water).
- The gradient of sodium ions is harnessed to provide the energy to run several types of indirect pumps.
The crucial roles of the Na+/K+ ATPase are reflected in the fact that almost one-third of all the energy generated by the mitochondria in animal cells is used just to run this pump.
The H+/K+ ATPase
The parietal cells of your stomach use this pump to secrete gastric juice. These cells transport protons (H+) from a concentration of about 4 x 10-8 M within the cell to a concentration of about 0.15 M in the gastric juice (giving it a pH close to 1). Small wonder that parietal cells are stuffed with mitochondria and uses huge amounts of ATP as they carry out this three-million fold concentration of protons.
The Ca2+ ATPases
A Ca2+ ATPase is located in the plasma membrane of all eukaryotic cells. It uses the energy provided by one molecule of ATP to pump one Ca2+ ion out of the cell. The activity of these pumps helps to maintain the ~20,000-fold concentration gradient of Ca2+ between the cytosol (~ 100 nM) and the ECF (~ 20 mM). In resting skeletal muscle, there is a much higher concentration of calcium ions (Ca2+) in the sarcoplasmic reticulum than in the cytosol. Activation of the muscle fiber allows some of this Ca2+ to pass by facilitated diffusion into the cytosol where it triggers contraction.
After contraction, this Ca2+ is pumped back into the sarcoplasmic reticulum. This is done by another Ca2+ ATPase that uses the energy from each molecule of ATP to pump 2 Ca2+ ions.
Pumps 1. - 3. are designated P-type ion transporters because they use the same basic mechanism: a conformational change in the proteins as they are reversibly phosphorylated by ATP. And all three pumps can be made to run backward. That is, if the pumped ions are allowed to diffuse back through the membrane complex, ATP can be synthesized from ADP and inorganic phosphate.
ABC Transporters
ABC ("ATP-Binding Cassette") transporters are transmembrane proteins that
- expose a ligand-binding domain at one surface and a
- ATP-binding domain at the other surface.
The ligand-binding domain is usually restricted to a single type of molecule.
The ATP bound to its domain provides the energy to pump the ligand across the membrane.
The human genome contains 48 genes for ABC transporters. Some examples:
- CFTR — the cystic fibrosis transmembrane conductance regulator
- TAP, the transporter associated with antigen processing
- The transporter that liver cells use to pump the salts of bile acids out into the bile.
- ABC transporters that pump chemotherapeutic drugs out of cancer cells thus reducing their effectiveness.
ABC transporters must have evolved early in the history of life. The ATP-binding domains in archaea, eubacteria, and eukaryotes all share a homologous structure, the ATP-binding "cassette".
Indirect Active Transport
Indirect active transport uses the downhill flow of an ion to pump some other molecule or ion against its gradient. The driving ion is usually sodium (Na+) with its gradient established by the Na+/K+ ATPase.
Symport Pumps
In this type of indirect active transport, the driving ion (Na+) and the pumped molecule pass through the membrane pump in the same direction. Examples:
- The Na+/glucose transporter. This transmembrane protein allows sodium ions and glucose to enter the cell together. The sodium ions flow down their concentration gradient while the glucose molecules are pumped up theirs. Later the sodium is pumped back out of the cell by the Na+/K+ ATPase. The Na+/glucose transporter is used to actively transport glucose out of the intestine and also out of the kidney tubules and back into the blood.
- All the amino acids can be actively transported, for example out of the kidney tubules and into the blood, by sodium-driven symport pumps.
- Sodium-driven symport pumps also return neurotransmitters to the presynaptic neuron.
- The Na+/iodide transporter. This symporter pumps iodide ions into the cells of the thyroid gland (for the manufacture of thyroxine) and also into the cells of the mammary gland (to supply the baby's need for iodide).
- The permease encoded by the lac operon of E. coli that transports lactose into the cell.
Antiport Pumps
In antiport pumps, the driving ion (again, usually sodium) diffuses through the pump in one direction providing the energy for the active transport of some other molecule or ion in the opposite direction. Example:
Ca2+ ions are pumped out of cells by sodium-driven antiport pumps. Antiport pumps in the vacuole of some plants harness the outward facilitated diffusion of protons (themselves pumped into the vacuole by a H+ ATPase) to the active inward transport of sodium ions. This sodium/proton antiport pump enables the plant to sequester sodium ions in its vacuole. Transgenic tomato plants that overexpress this sodium/proton antiport pump are able to thrive in saline soils too salty for conventional tomatoes. Antiport pumps to the active inward transport of nitrate ions (NO3−)
Some inherited ion-channel diseases
A growing number of human diseases have been discovered to be caused by inherited mutations in genes encoding channels.
Examples:
- Chloride-channel diseases
- cystic fibrosis
- inherited tendency to kidney stones (caused by a different kind of chloride channel than the one involved in cystic fibrosis)
- Potassium-channel diseases
- the majority of cases of long QT syndrome, an inherited disorder of the heartbeat
- a rare, inherited tendency to epileptic seizures in the newborn
- several types of inherited deafness
- Sodium-channel diseases
- inherited tendency to certain types of muscle spasms
- Liddle's syndrome. Inadequate sodium transport out of the kidneys, because of a mutant sodium channel, leads to elevated osmotic pressure of the blood and resulting hypertension (high blood pressure)
Osmosis
Osmosis is a special term used for the diffusion of water through cell membranes. Although water is a polar molecule, it is able to pass through the lipid bilayer of the plasma membrane. Aquaporins — transmembrane proteins that form hydrophilic channels — greatly accelerate the process, but even without these, water is still able to get through. Water passes by diffusion from a region of higher to a region of lower concentration. Note that this refers to the concentration of water, NOT the concentration of any solutes present in the water. Water is never transported actively; that is, it never moves against its concentration gradient. However, the concentration of water can be altered by the active transport of solutes and in this way the movement of water in and out of the cell can be controlled. Example: the reabsorption of water from the kidney tubules back into the blood depends on the water following behind the active transport of \(Na^+\).
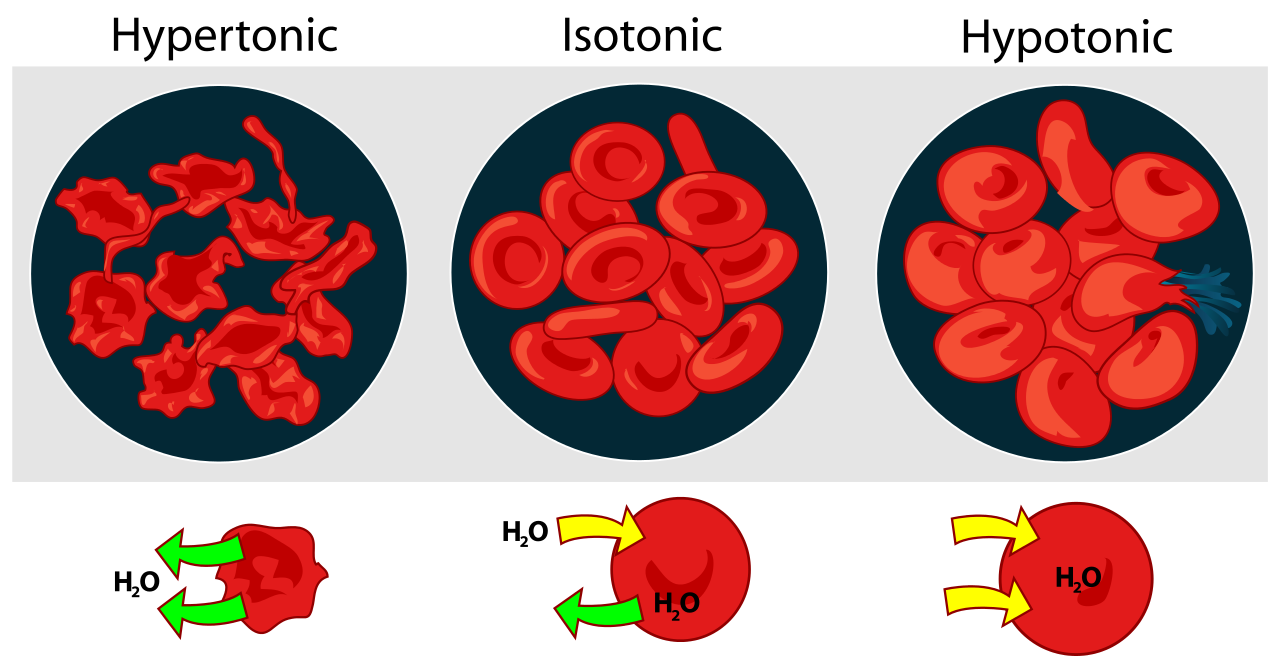
- Hypotonic solutions: If the concentration of water in the medium surrounding a cell is greater than that of the cytosol, the medium is said to be hypotonic. Water enters the cell by osmosis. A red blood cell placed in a hypotonic solution (e.g., pure water) bursts immediately ("hemolysis") from the influx of water. Plant cells and bacterial cells avoid bursting in hypotonic surroundings by their strong cell walls. These allow the buildup of turgor within the cell. When the turgor pressure equals the osmotic pressure, osmosis ceases.
- Isotonic solutions: When red blood cells are placed in a 0.9% salt solution, they neither gain nor lose water by osmosis. Such a solution is said to be isotonic. The extracellular fluid (ECF) of mammalian cells is isotonic to their cytoplasm. This balance must be actively maintained because of the large number of organic molecules dissolved in the cytosol but not present in the ECF. These organic molecules exert an osmotic effect that, if not compensated for, would cause the cell to take in so much water that it would swell and might even burst. This fate is avoided by pumping sodium ions out of the cell with the Na+/K+ ATPase.
- Hypertonic solutions: If red cells are placed in sea water (about 3% salt), they lose water by osmosis and the cells shrivel up. Sea water is hypertonic to their cytosol. Similarly, if a plant tissue is placed in sea water, the cell contents shrink away from the rigid cell wall. This is called plasmolysis. Sea water is also hypertonic to the ECF of most marine vertebrates. To avoid fatal dehydration, these animals (e.g., bony fishes like the cod) must continuously drink sea water and then desalt it by pumping ions out of their gills by active transport.
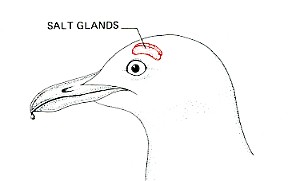
Marine birds, which may pass long periods of time away from fresh water, and sea turtles use a similar device. They, too, drink salt water to take care of their water needs and use metabolic energy to desalt it. In the herring gull, shown here, the salt is extracted by two glands in the head and released (in a very concentrated solution — it is saltier than the blood) to the outside through the nostrils. Marine snakes use a similar desalting mechanism.