6.4: The Translation of RNA into Proteins
- Page ID
- 4836
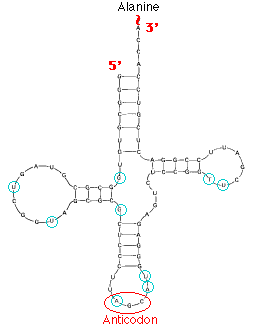
This image shows the structure of alanine transfer RNA (tRNAala) from yeast. It consists of a single strand of 77 ribonucleotides. The chain is folded on itself, and many of the bases pair with each other forming four helical regions. Loops are formed in the unpaired regions of the chain. (The bases circled in blue have been chemically-modified following synthesis of the molecule.)
At least one kind of tRNA is present for each of the 20 amino acids used in protein synthesis. (Some amino acids employ the services of two or three different tRNAs, so most cells contain as many as 32 different kinds of tRNA.) The amino acid is attached to the appropriate tRNA by an activating enzyme (one of 20 aminoacyl-tRNA synthetases) specific for that amino acid as well as for the tRNA assigned to it.
Each kind of tRNA has a sequence of 3 unpaired nucleotides — the anticodon — which can bind, following the rules of base pairing, to the complementary triplet of nucleotides — the codon — in a messenger RNA (mRNA) molecule. Just as DNA replication and transcription involve base pairing of nucleotides running in opposite direction, so the reading of codons in mRNA (5' -> 3') requires that the anticodons bind in the opposite direction.
Anticodon: 3' CGA 5'
Codon: 5' GCU 3'
The RNA Codons
| U | C | A | G | ||
|---|---|---|---|---|---|
| U | UUU Phenylalanine (Phe) | UCU Serine (Ser) | UAU Tyrosine (Tyr) | UGU Cysteine (Cys) | U |
| UUC Phe | UCC Ser | UAC Tyr | UGC Cys | C | |
| UUA Leucine (Leu) | UCA Ser | UAA STOP | UGA STOP | A | |
| UUG Leu | UCG Ser | UAG STOP | UGG Tryptophan (Trp) | G | |
| C | CUU Leucine (Leu) | CCU Proline (Pro) | CAU Histidine (His) | CGU Arginine (Arg) | U |
| CUC Leu | CCC Pro | CAC His | CGC Arg | C | |
| CUA Leu | CCA Pro | CAA Glutamine (Gln) | CGA Arg | A | |
| CUG Leu | CCG Pro | CAG Gln | CGG Arg | G | |
| A | AUU Isoleucine (Ile) | ACU Threonine (Thr) | AAU Asparagine (Asn) | AGU Serine (Ser) | U |
| AUC Ile | ACC Thr | AAC Asn | AGC Ser | C | |
| AUA Ile | ACA Thr | AAA Lysine (Lys) | AGA Arginine (Arg) | A | |
| AUG Methionine (Met) or START | ACG Thr | AAG Lys | AGG Arg | G | |
| G | GUU Valine Val | GCU Alanine (Ala) | GAU Aspartic acid (Asp) | GGU Glycine (Gly) | U |
| GUC (Val) | GCC Ala | GAC Asp | GGC Gly | C | |
| GUA Val | GCA Ala | GAA Glutamic acid (Glu) | GGA Gly | A | |
| GUG Val | GCG Ala | GAG Glu | GGG Gly | G |
Note:
- Most of the amino acids are encoded by synonymous codons that differ in the third position of the codon.
- In some cases, a single tRNA can recognize two or more of these synonymous codons.
- Example: phenylalanine tRNA with the anticodon 3' AAG 5' recognizes not only UUC but also UUU.
- The violation of the usual rules of base pairing at the third nucleotide of a codon is called "wobble"
- The codon AUG serves two related functions
- It begins every message; that is, it signals the start of translation placing the amino acid methionine at the amino terminal of the polypeptide to be synthesized.
- When it occurs within a message, it guides the incorporation of methionine.
- Three codons, UAA, UAG, and UGA, act as signals to terminate translation. They are called STOP codons.
The Steps of Translation
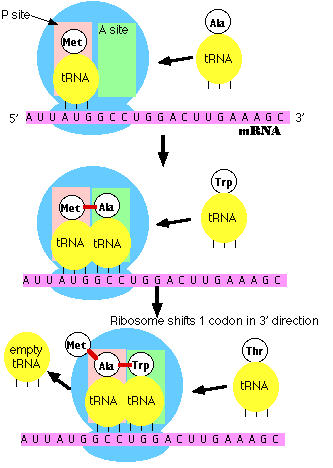
Initiation
- The small subunit of the ribosome binds to a site "upstream" (on the 5' side) of the start of the message.
- It proceeds downstream (5' -> 3') until it encounters the start codon AUG. (The region between the mRNA cap and the AUG is known as the 5'-untranslated region [5'-UTR].)
- Here it is joined by the large subunit and a special initiator tRNA.
- The initiator tRNA binds to the P site (shown in pink) on the ribosome.
- In eukaryotes, initiator tRNA carries methionine (Met). (Bacteria use a modified methionine designated fMet.)
Elongation
- An aminoacyl-tRNA (a tRNA covalently bound to its amino acid) able to base pair with the next codon on the mRNA arrives at the A site (green) associated with:
- an elongation factor (called EF-Tu in bacteria; EF-1 in eukaryotes)
- GTP (the source of the needed energy)
- The preceding amino acid (Met at the start of translation) is covalently linked to the incoming amino acid with a peptide bond (shown in red).
- The initiator tRNA is released from the P site.
- The ribosome moves one codon downstream.
- This shifts the more recently-arrived tRNA, with its attached peptide, to the P site and opens the A site for the arrival of a new aminoacyl-tRNA.
- This last step is promoted by another protein elongation factor (called EF-G in bacteria; EF-2 in eukaryotes) and the energy of another molecule of GTP.
Note: the initiator tRNA is the only member of the tRNA family that can bind directly to the P site. The P site is so-named because, with the exception of initiator tRNA, it binds only to a peptidyl-tRNA molecule; that is, a tRNA with the growing peptide attached.
The A site is so-named because it binds only to the incoming aminoacyl-tRNA; that is the tRNA bringing the next amino acid. So, for example, the tRNA that brings Met into the interior of the polypeptide can bind only to the A site.
Termination
- The end of translation occurs when the ribosome reaches one or more STOP codons (UAA, UAG, UGA). (The nucleotides from this point to the poly(A) tail make up the 3'-untranslated region [3'-UTR] of the mRNA.)
- There are no tRNA molecules with anticodons for STOP codons.
- However, protein release factors recognize these codons when they arrive at the A site.
- Binding of these proteins —along with a molecule of GTP — releases the polypeptide from the ribosome.
- The ribosome splits into its subunits, which can later be reassembled for another round of protein synthesis.
Polysomes
A single mRNA molecule usually has many ribosomes traveling along it, in various stages of synthesizing the protein. This complex is called a polysome.
Codon Bias
All but two of the amino acids (Met and Trp) can be encoded by from 2 to 6 different codons. However, the genome of most organisms reveals that certain codons are preferred over others. In humans, for example, alanine is encoded by GCC four times as often as by GCG. This probably reflects a greater translation efficiency by the translation apparatus for certain codons over their synonyms.
- At the start of translation, two or more of a set of synonymous codons (e.g., the 6 codons that incorporate leucine in the growing protein) are used alternately. The need to locate first one and then another tRNA for that amino acid slows down the rate of translation.
- This may aid in keeping ribosomes from bumping into each other on the polysome.
- It may also provide more time for the nascent protein to begin to fold correctly as it emerges from the ribosome.
- Once translation is well underway (after 30–50 amino acids have been added), one particular codon tends to be chosen each time its amino acid is called for. Presumably this now increases the efficiency (speed) of translation.
- Most organisms have more than the 61 genes needed to encode a tRNA for each of the 61 codons (we have 270 tRNA genes). The presence of multiple genes for tRNAs with an identical anticodon increases the concentration of tRNAs able to bind a particular codon. Messenger RNAs — especially those of active genes — tend to favor codons that correspond to abundant tRNAs carrying the anticodon.
Codon bias even extends to pairs of codons: wherever a human protein contains the amino acids Ala-Glu, the gene encoding those amino acids is seven times as likely to use the codons GCAGAG rather than the synonymous GCCGAA. Codon bias is exploited by the biotechnology industry to improve the yield of the desired product. The ability to manipulate codon bias may also usher in a era of safer vaccines.
Quality Control
Defective mRNA molecules can be produced by mutations in the gene as well as errors introduced during transcription (albeit at a remarkably low rate). In addition to producing mRNAs with incorrect codons for amino acids, these errors can produce mRNA molecules that have
- Premature Termination Codons (PTCs); that is, the introduction of a STOP codon before the normal end of the message. Translation of these mRNAs produces a truncated protein that is probably ineffective and may be harmful. The problem can sometimes be solved by Nonsense-Mediated mRNA Decay (NMD).
- no STOP codon. These produce "nonstop" transcripts. The problem can be solved by Nonstop mRNA Decay.
Nonsense-Mediated mRNA Decay (NMD)
Premature termination codons (PTCs) may be generated by "nonsense" mutations, frameshifts, and RNA processing (intron removal) errors. They are also an inevitable consequence of creating antigen receptors on B cells and T cells.
Mechanisms
- During RNA processing within the nucleus, protein complexes are added at each spot where adjacent exons are spliced together. (These are important signals for exporting the mRNA to the cytoplasm.)
- In the cytoplasm, as the ribosome moves down the mRNA, these complexes are removed (and sent back to the nucleus for reuse).
- If the ribosome encounters a premature termination codon, the final exon-exon tag(s) are not removed, and this marks the defective mRNA for destruction (in P bodies).
Mutations that introduce premature termination codons are responsible for some cases of such inherited human diseases as cystic fibrosis and Duchenne muscular dystrophy (DMD).
A drug, designated PTC124 or ataluren, causes the ribosome to skip over PTCs while still enabling normal termination of translation. PTC124 has shown promise in animal models of cystic fibrosis and DMD and phase II clinical trials are now being conducted on humans.
Nonstop mRNA Decay
Nonstop transcripts occur when there is no STOP codon in the message. As a result the ribosome is unable to recruit the release factors needed to leave the mRNA. Nonstop transcripts are formed during RNA processing, e.g., by having the poly(A) tail put on before the STOP codon is reached.
Mechanisms
Eukaryotes and bacteria handle the problem of no STOP codon differently.
- In eukaryotes, when the ribosome stalls at the end of the poly(A) tail, proteins are recruited to release the ribosome for reuse and to degrade the faulty message.
- In bacteria, a special RNA molecule — called tmRNA saves the day. It is called tmRNA because it has the properties of both a transfer RNA and a messenger RNA. The transfer part adds alanine to the A site on the ribosome. The ribosome then moves on to the messenger part which encodes 10 amino acids that target the molecule for destruction (and releases the ribosome for reuse).
Regulation of Translation
The expression of most genes is controlled at the level of their transcription. Transcription factors (proteins) bind to promoters and enhancers turning on (or off) the genes they control. However, gene expression can also be controlled at the level of translation.
By General RNA-Degradation Machinery
P bodies
The cytosol of eukaryotes contains protein complexes that compete with ribosomes for access to mRNAs. As these increase their activity, they sequester mRNAs in larger aggregates called P bodies (for "processing bodies", but this processing should not be confused with the processing of pre-mRNA to mature mRNA that occurs in the nucleus).
These protein complexes break down the mRNA by
- removing its "cap"
- removing its poly(A) tail
- degrading the remaining message (nibbling away in the 5' -> 3' direction)
What controls the dynamic balance between ribosomes and P bodies for access to mRNAs remains to be learned. But this mechanism provides for
- destruction of "bad" mRNAs (e.g., those with premature STOP codons
- turnover of mRNAs thus increasing the flexibility of gene expression in the cell
Exosomes
These are hollow macromolecular complexes with two openings. They take in unfolded RNA molecules and degrade them in the 3' -> 5' direction. (In neither structure nor function do these exosomes resemble the exosomes involved in antigen presentation that unfortunately share the same name.)
By MicroRNAs (miRNAs)
Here small RNA molecules bind to a complementary portion in the 3'-UTR of the mRNA and prevent it from being translated by ribosomes and/or trigger its destruction. Both these activities take place in P bodies.
By Riboswitches
It turns out that the regulation of the level of certain metabolites is controlled by riboswitches. A riboswitch is a part of a molecule of messenger RNA (mRNA) with a specific binding site for the metabolite (or a close relative).
Examples:
- If thiamine pyrophosphate (the active form of thiamine [vitamin B1]) is available in the culture medium of E. coli,
- It binds to a messenger RNA whose protein product is an enzyme needed to synthesize thiamine from the ingredients in minimal medium.
- Binding induces an allosteric shift in the structure of the mRNA so that it can no longer bind to a ribosome and thus cannot be translated into the enzyme.
- E. coli no longer wastes resources on synthesizing a vitamin that is available preformed.
- If vitamin B12 is present in the cell, it binds to the mRNA which encodes a protein needed to import the vitamin from the culture medium. This, too, induces an allosteric shift in the mRNA that prevents it from binding a ribosome. E. coli no longer wastes resources on synthesizing a transporter for a vitamin that it already has enough of.
- Some Gram-positive bacteria (E. coli is Gram-negative) control the level of a sugar needed to synthesize their cell wall with a riboswitch. In this case, as the concentration of the sugar builds up, it binds to the messenger RNA (mRNA) whose product is the enzyme that makes the sugar. This causes the mRNA to self-destruct so production of the enzyme — and thus the sugar — ceases.
It has been suggested that these regulatory mechanisms, which do not involve any protein, are a relict from an "RNA world".


