4.14: Secondary Messengers
- Page ID
- 4657
Second messengers are molecules that relay signals received at receptors on the cell surface — such as the arrival of protein hormones, growth factors, etc. — to target molecules in the cytosol and/or nucleus. But in addition to their job as relay molecules, second messengers serve to greatly amplify the strength of the signal. Binding of a ligand to a single receptor at the cell surface may end up causing massive changes in the biochemical activities within the cell.
There are 3 major classes of second messengers:
- cyclic nucleotides (e.g., cAMP and cGMP)
- inositol trisphosphate (IP3) and diacylglycerol (DAG)
- calcium ions (Ca2+)
Cyclic Nucleotides
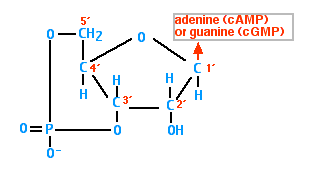
Cyclic AMP (cAMP)
Some of the hormones that achieve their effects through cAMP as a second messenger:
- adrenaline
- glucagon
- luteinizing hormone (LH)
Cyclic AMP is synthesized from ATP by the action of the enzyme adenylyl cyclase.
- Binding of the hormone to its receptor activates
- a G protein which, in turn, activates
- adenylyl cyclase.
- The resulting rise in cAMP turns on the appropriate response in the cell by either (or both):
- changing the molecular activities in the cytosol, often using Protein Kinase A (PKA) — a cAMP-dependent protein kinase that phosphorylates target proteins
- turning on a new pattern of gene transcription
Cyclic GMP (cGMP)
Cyclic GMP is synthesized from the nucleotide GTP using the enzyme guanylyl cyclase. Cyclic GMP serves as the second messenger for
- atrial natriuretic peptide (ANP)
- nitric oxide (NO)
- the response of the rods of the retina to light
Some of the effects of cGMP are mediated through Protein Kinase G (PKG) — a cGMP-dependent protein kinase that phosphorylates target proteins in the cell.
Inositol trisphosphate (IP3) and diacylglycerol (DAG)
Peptide and protein hormones like vasopressin, thyroid-stimulating hormone (TSH), and angiotensin and neurotransmitters like GABA bind to G protein-coupled receptors (GPCRs) that activate the intracellular enzyme phospholipase C (PLC).
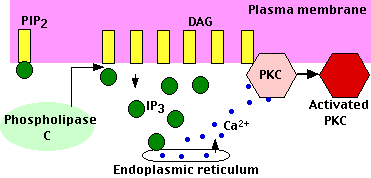
As its name suggests, it hydrolyzes phospholipids — specifically phosphatidylinositol-4,5-bisphosphate (PIP2) which is found in the inner layer of the plasma membrane. Hydrolysis of PIP2 yields two products:
- diacylglycerol (DAG): DAG remains in the inner layer of the plasma membrane. It recruits Protein Kinase C (PKC) — a calcium-dependent kinase that phosphorylates many other proteins that bring about the changes in the cell. As its name suggests, activation of PKC requires calcium ions. These are made available by the action of the other second messenger — IP3.
- inositol-1,4,5-trisphosphate (IP3): This soluble molecule diffuses through the cytosol and binds to receptors on the endoplasmic reticulum causing the release of calcium ions (Ca2+) into the cytosol. The rise in intracellular calcium triggers the response.
Example:
The calcium rise is needed for NF-AT (the "nuclear factor of activated T cells") to turn on the appropriate genes in the nucleus.
The remarkable ability of tacrolimus and cyclosporine to prevent graft rejection is due to their blocking this pathway.
The binding of an antigen to its receptor on a B cell (the BCR) also generates the second messengers DAG and IP3.
Calcium ions (Ca2+)
As the functions of IP3 and DAG indicate, calcium ions are also important intracellular messengers. In fact, calcium ions are probably the most widely used intracellular messengers.
In response to many different signals, a rise in the concentration of Ca2+ in the cytosol triggers many types of events such as
- muscle contraction
- exocytosis, e.g.
- release of neurotransmitters at synapses (and essential for the long-term synaptic changes that produce Long-Term Potentiation (LTP) and Long-Term Depression (LTD);
- secretion of hormones like insulin
- activation of T cells and B cells when they bind antigen with their antigen receptors (TCRs and BCRs respectively)
- adhesion of cells to the extracellular matrix (ECM)
- apoptosis
- a variety of biochemical changes mediated by Protein Kinase C (PKC).
Normally, the level of calcium in the cell is very low (~100 nM). There are two main depots of Ca2+ for the cell:
- The extracellular fluid (ECF — made from blood), where the concentration is ~ 2 mM or 20,000 times higher than in the cytosol;
- the endoplasmic reticulum ("sarcoplasmic" reticulum in skeletal muscle).
However, its level in the cell can rise dramatically when channels in the plasma membrane open to allow it in from the extracellular fluid or from depots within the cell such as the endoplasmic reticulum and mitochondria.
Getting Ca2+ into (and out of) the cytosol
- Voltage-gated channels
- open in response to a change in membrane potential, e.g. the depolarization of an action potential
- are found in excitable cells:
- skeletal muscle
- smooth muscle (These are the channels blocked by drugs, such as felodipine [Plendil®], used to treat high blood pressure. The influx of Ca2+ contracts the smooth muscle walls of the arterioles, raising blood pressure. The drugs block this.)
- neurons. When the action potential reaches the presynaptic terminal, the influx of Ca2+ triggers the release of the neurotransmitter.
- the taste cells that respond to salt.
- allow some 106 ions to flow in each second following the steep concentration gradient.
- Receptor-operated channels
These are found in the post-synaptic membrane and open when they bind the neurotransmitter. Example: NMDA receptors. - G-protein-coupled receptors (GPCRs). These are not channels but they trigger a release of Ca2+ from the endoplasmic reticulum as described above. They are activated by various hormones and neurotransmitters (as well as bitter substances on taste cells in the tongue).
Ca2+ ions are returned
- to the ECF by active transport using
- an ATP-driven pump called a Ca2+ ATPase;
- two Na+/Ca2+ exchangers. These antiport pumps harness the energy of
- 3 Na+ ions flowing DOWN their concentration gradient to pump one Ca2+ against its gradient and
- 4 Na+ ions flowing down to pump 1 Ca2+ and 1 K+ ion up their concentration gradients.
- to the endoplasmic (and sarcoplasmic) reticulum using another Ca2+ ATPase.
How can such a simple ion like Ca2+ regulate so many different processes? Some factors at work:
- localization within the cell (e.g., released at one spot — the T-system is an example — or spread throughout the cell)
- by the amount released (amplitude modulation, "AM")
- by releasing it in pulses of different frequencies (frequency modulation, "FM")


