3. Butterfly Wings Lab: Boundaries and Pattern Formation
- Page ID
- 14909
Butterfly Wings Lab
Nipam Patel, the author of the picture below, is interested in using gynandromorphic Morpho butterflies to understand wing development in insects. Much of his research focuses on developmental mechanisms of boundary formation across evolution. Boundary formation (like we see in brain lamination or in segmentation) is a key process in compartmentalizing an embryo. As we read before, compartmentalization can increase evolvability by separating cellular functions and reducing pleiotropy.

In Figure 1, a series of Blue Morpho butterflies is pictured. The top left butterfly is a wild-type male and the top center butterfly is a wild-type female. All of the rest of the butterflies are gynandromorphic with some male cells and some female cells in their wings. We will use these natural sports to make a hypothesis about boundary formation in butterfly wings.
Genetic mosaics and eyespots
Genetic mosaics are classical models for studying developmental processes. Mutations in key developmental genes often results in early embryonic lethality. Sometimes, a genetic mosaic organism can be generated that has only a small subset of cells or a single tissue/organ that exhibits the mutation. In these cases, the role of the gene in just that tissue or organ can be studied much later in development1. Drosophila wing compartments and the finding that the transcription factor Engrailed is involved in boundary formation were both discovered using genetic mosaics2,3. Drosophila wings tend to be fairly simple in terms of wing vein patterning and pigmentation, compared to butterfly wings (see Figure 3 below). However both wings undergo very similar early development and share a basic "ground plan", the set of instructions that both defines the major regions of the wing (veins and margin) as well as sets the boundaries for the initiation of color pattern elements4. These pigmented pattern elements (notably eyespots) can be repeated across the wing with each element exhibiting a similar morphology due to similar developmental patterning5. Because of this, eyespots are considered to be serial homologs like limbs or ectodermal appendages.
Color elements are a useful assessor of boundaries since they are easy to see and vary both within a population and between species. Since the color elements are serially homologous, a single wing offers multiple opportunities to observe the effect of boundaries on pattern formation. By creating or finding genetic mosaics that exhibit abnormal wing patterning, we can start to understand how compartments both restrict individual phenotypic variance (by providing a ground plan) and allow for greater population variance (by reducing inductive effects).
The wing ground plan
The wing ground plan develops along 3 axes: the D/V axis (the front and the back of the wing), the A/P axis (the top/bottom axis on the figures below), and the proximal-distal axis. For this lab, we will only consider the dorsal sides of the wings. Each side (dorsal or ventral) is patterned on a warped X-Y coordinate grid with the veins making up the horizontal boundaries and a series of bands (colored in Figure 2) making up the ventral boundaries.
|
Figure 2: The nymphalid ground plan The vertical boundaries of the nymphalid ground plan were first proposed by Fred Nijhout to explain color variation in the Nymphalidae, a large group of spectacularly patterned butterflies6. These bands, from distal to proximal, are the wing margin (red), the border band (orange), the central band (yellow), and the basal band (green). The horizontal boundaries are formed by the wing veins. The intervein region within a band is a compartment called a "cell" where pattern formation can be initiated semi-independently from other cells6. |
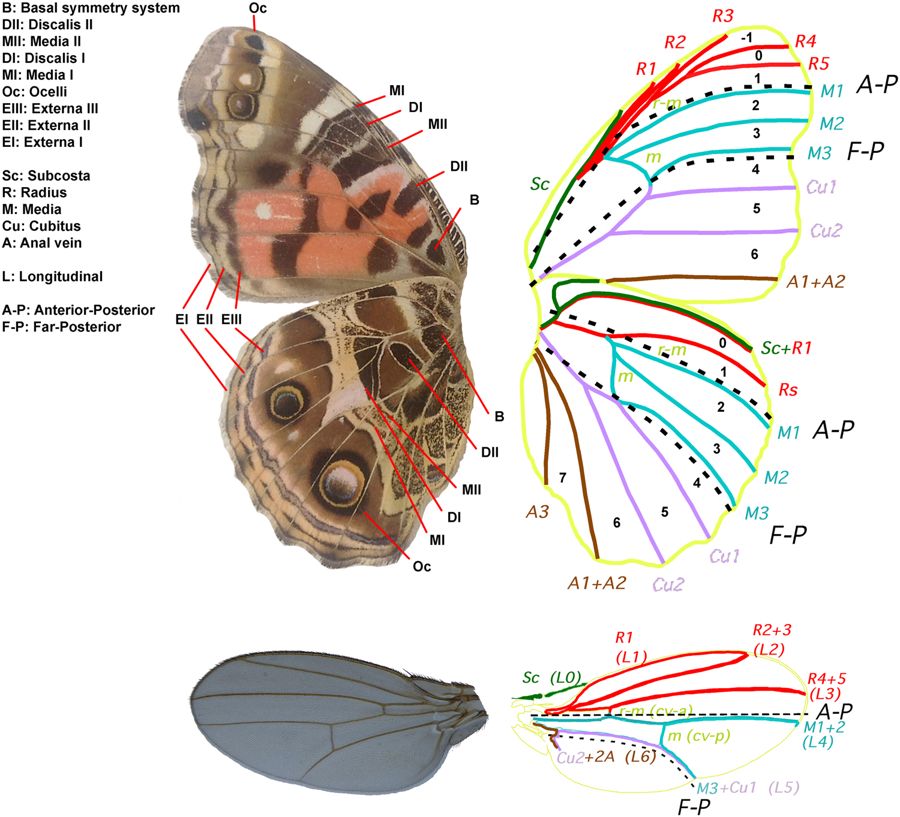
In the last 3 decades as gene expression, gene sequencing, and genetic manipulation techniques have become more reliable, a wealth of knowledge regarding butterfly wing patterning has emerged. For example, Abbassi and Marcus (2017) define a new developmental compartment boundary shared between butterflies and Drosophila (named Far-Posterior) based on genetic mosaic and gynandromorph data (Figure 3)
Lab instructions
1. Using Figure 1 above, draw the Blue Morpho ground plan. Write up a short justification for your ground plan based on the picture and the information
2. Do you think this ground plan is homologous to the Drosophila and V. braziliensis ground plans? Why or why not?
3. Imagine you are a butterfly researcher trying to justify a collecting trip to an exotic location to collect butterflies. How would you pitch your research to the greater scientific/heathcare community?
Class Discussion Readings and QuestionsRead an overview of Gynandromorphism in animals Read an overview of tetragametism(genetic gynandromorphism) Read a case study of a genetic "gynandromorphic" human , you can skip the Materials, Methods, and Results sections, please focus on the Introduction, Case Report, and Discussion. Choose one or more of the questions below to answer 1. What is gynandromorphism, how can we tell when an organism is gynandromorphic? How did doctors know that a phenotypically normal girl was gynandromorphic (in the third reading)? 2. Why do scientists and doctors seek out gynandromorphic individuals to study? How are they useful for understanding biology? Can scientists create gynandromorphs or other types of mosaic/chimeric animals? 3. What are the different mechanisms that can give a 46XY/46XX karyotype in an amniocentesis? Be sure to describe both the false positive as well as different developmental mechanisms for generating true positives. 4. Gynandromorphic (46XX/46XY) humans often have abnormal genitalia and gonads that are either a mix of ovarian and testicular tissue (called ovotestes) or one ovary and one testis. What information does this give us regarding the development of gonads and genitalia in humans? |
References
1. Beatrice Mintz "Formation of genetically mosaic mouse embryos, and early development of “lethal (t12/t12)–normal” mosaics" 1964. Journal of Experimental Zoology 157(2):273-291 doi.org/10.1002/jez.1401570210
2. Garcia-Bellido, A., Ripoll, P. & Morata, G. "Developmental compartmentalisation of the wing disk of Drosophila." 1973, Nature New Biol. 245, 251–253 https://www.nature.com/articles/newbio245251a0
3. Brower, D. L. engrailed gene expression in Drosophila imaginal discs.1986. EMBO J. 5, 2649–2656. PMID: 3536480
4. Martin, A. & Reed, R. D. "wingless and aristaless2 define a developmental ground plan for moth and butterfly wing pattern evolution." 2010 Mol. Biol. Evol. 27: 2864–2878. https://doi.org/10.1093/molbev/msq173
5. Abbasi, R. & Marcus, J. M. "Colour pattern homology and evolution in Vanessa butterflies (Nymphalidae: Nymphalini): Eyespot characters." 2015. J. Evol. Biol. 28, 2009–2026, doi.org/10.1111/jeb.12716
6. Roohollah Abbasi & Jeffrey M. Marcus "A new A-P compartment boundary and organizer in holometabolous insect wings " 2017. Nature. https://doi.org/10.1038/s41598-017-16553-5


