8.4: History of Extinctions in Sub-Saharan Africa
- Page ID
- 26806
Many people hold on to the romanticised belief that historical human societies lived in harmony with nature. Accumulated evidence however indicates that this is not true; early humans have caused extensive ecosystem changes and species extinctions since Homo sapiens appeared on Earth about 300,000 years ago. In fact, even before the arrival of humans, our ancestors had made a mark, by driving species to extinction as early as during the Pleistocene period, which started about 2.5 million years ago (Box 8.1). The impact of early humans was particularly devastating to the wildlife of North America, South America, and Australia, which saw the demise of nearly all their large ( 100 kg) mammals, most famously megaherbivores such as the mammoths (Mammuthus spp.). The Pleistocene extinctions were somewhat less devastating to wildlife in Africa, Europe, and Asia, possibly because large mammals on these continents evolved with human predators, allowing them to develop appropriate defence/escape mechanisms. Nevertheless, Africa’s wildlife did not completely escape the Pleistocene extinctions, as increasingly-sophisticated human activities during that time ensured the demise of as many as 28 large mammal groups, which included Africa’s sabre-toothed cats (Barbourofelidae), nearly all the elephant relatives (Proboscidae), as well as giant hartebeests (Megalotragus spp.), giant buffaloes (Pelorovis spp.), giant hyenas (Pachycrocuta spp.), and giant giraffes (Sivatherium spp.).
Early humans have caused extensive ecosystem changes and species extinctions even before Homo sapiens appeared on Earth about 300,000 years ago.
David H.M. Cumming1,2
1FitzPatrick Institute of African Ornithology,
University of Cape Town, South Africa.
2Tropical Resource Ecology Programme, University of Zimbabwe,
Harare, Zimbabwe.
Many scientists consider the present rapid loss of biodiversity to be the start of the 6th mass extinction following five previous extinction episodes (see Figure 8.1), each of which led to large-scale restructuring of Earth’s biodiversity. The 5th global mass extinction took place 65 million years ago (Ma) when a massive meteorite collided with the Earth, and resulted in the extinction of all non-avian dinosaurs, and much else besides. This 5th mass extinction event also marked the transition from the Cretaceous to the Tertiary epoch (65 to 2.5 Ma). The Tertiary epoch was followed by the Quaternary, which includes the Pleistocene period (2.5 million to ~ 12 thousand years ago) and more recently the Holocene period—marked by the development of agriculture, and the subsequent domination of the Earth’s resources by Homo sapiens. The Pleistocene is known for a mini mass-extinction of sorts, which saw the demise of species such as mammoths and sabre-tooth cats. However, unlike previous comprehensive mass extinctions, the Pleistocene was characterized by the extinction of mostly large mammals and very large island birds.
Attempts by scientists to explain these Pleistocene extinctions have been characterized by two centuries of controversy over whether they were caused by climate change or by predatory hominins—the evolutionary line of primates that gave rise to modern humans. The four main hypotheses advanced to account for the loss of Pleistocene fauna are: (i) climate change with minimal if any hominin influence (e.g. Faith et al., 2018); (ii) climate change together with some hominin influence (e.g. Barnosky et al., 2004); (iii) selective hominin predation aided by climate change (e.g. Bartlett et al., 2015), and (iv) hominin predation helped by other large predators without the influence of climate change (e.g. Janzen, 1983, Ripple and Van Valkenburgh, 2010).
The very close relationship between the dispersal of hominins out of Africa, the timing of their arrival elsewhere in the world, and the subsequent extinction of large mammals and birds, provided the primary (if challenged) evidence for human agency in non-African Late- Pleistocene extinctions (e.g. Surovell et al., 2005; Johnson, 2009; Ripple and Van Valkenburgh, 2010). As Haynes (2018) has remarked, “…the proponents of climate change as the only cause of the Late Pleistocene extinctions have not clearly explained how or why so many of the extinct megafaunal genera had survived numerous earlier climate changes.”
Similarly, Faith et al. (2018) have stated that the failure of Pleistocene megaherbivores to adapt to the emergence of C4 grasses was the primary driver of their extinction. However, this claim ignores evidence that many extinct herbivore genera and species previously survived changes in diet over time (Ripple and Van Valkenburgh, 2010), that the diets of particular species were known to vary with location (Ferranec, 2004), and that many large species, which are typically highly mobile generalists, would have had little trouble adapting their ranges and diets to changing climates.
Research and debate on Pleistocene extinctions have tended to focus on the demise of non-African large mammals in the Late Pleistocene, which coincided with the period when hominins (Homo erectus and later also H. sapiens) dispersed across the globe starting at about 2 Ma. In Africa, however, earliest hominins appeared some 7 Ma. It didn’t take long for these early African hominins to develop the skills necessary to manipulate the environment to their advantage. Setting the pace were the Australopithecines, who used stone tools to butcher mammalian carcasses between 4–3 Ma. The Australopithecines and the rest of a diverse group of large predators were joined by H. erectus at the beginning of the Pleistocene, about 2 Ma. This new, qualitatively unique, hunter was able to hunt collaboratively in bands, and was anatomically adapted to throw projectiles forcefully and accurately at large prey (Lombardo and Deaner, 2018). A large brain also placed high nutritional and energetic demands on H. erectus, that could best be met by obtaining meat and bone marrow from proboscidians, the elephant relatives (Surovell et al., 2005; Boschian et al., 2019). Early African hominins were thus well adapted to hunt large prey and contribute to the demise of wildlife, particularly megaherbivores (those over 1,000 kg), through the Early and Middle Pleistocene (Figure 8.A). Other large carnivores at the time may very well have helped hominins drive many Pleistocene herbivores to extinction (Janzen, 1983, Ripple and Van Valkenburgh, 2010; Van Valkenburgh et al., 2016). But this combination may ironically also have led to demise of many of the large Pleistocene predators, through co-extinctions, after their main prey base disappeared (Werdelin and Lewis, 2013).

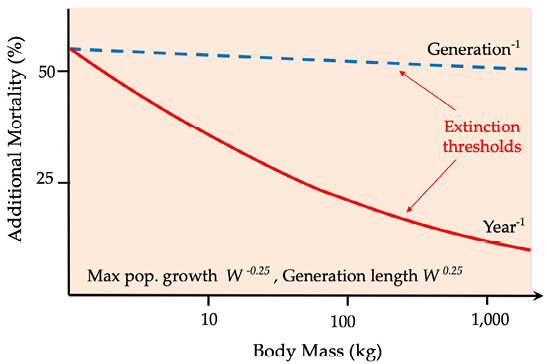
It thus seems likely that the emergence of a novel and increasingly effective predator during the Early Pleistocene, rather than climate change, was the ultimate factor that tipped the balance against the iconic species that disappeared soon after hominins appeared on Earth. It is worth noting that there is a clear relationship between body size and extinction risk (Figure 8.B), the result of large animals’ relatively long generation lengths, long gestation periods, long periods of caring for young, and an abundance of meat presented to eager hunters. Consequently, even a small increase in mortality may very well result in a large animal’s annual mortality exceeding its generational mortality, the end result being extinction. This relationship also partly explains why present-day elephant populations are unable to withstand poaching in many parts of Africa (Box 7.2).
The lessons from the Pleistocene extinctions are relevant also today. As explained above, accumulated evidence indicates that early humans have caused extensive ecosystem changes and species extinctions extending over more than a million years. Homo sapiens had emerged in Africa about 300,000 years ago (Callaway, 2017). As early humans mastered the use of fire, poison-tipped spears and arrows, pitfall traps, and a host of additional hunting techniques, this unique apex predator proceeded to influence the structure and composition of African (and global) landscapes, and the plant and vertebrate assemblages of the continent (Smith et al., 2019). For the last two million years our ancestors have set in motion a series of trophic cascades that continue to this day and are resulting in increasing loss of diversity of the flora and fauna of the African continent and the rest of the world.
While early extinctions were generally isolated and selective, extinction rates increased rapidly after the rise of agriculture, and especially after European settlers started colonising Africa from the 17th century onward. By no coincidence, the area where most of the extinctions and extirpations during colonialism occurred was on the southwestern tip of Africa, the location of the earliest intensive European settlements on the continent. For example, by 1700, hunting caused the extirpation of every single land animal over 50 kg within 200 km of Cape Town (Rebelo, 1992). As hunters moved further afield in search of targets, Africa saw its first post-colonial large mammal extinctions, namely the bluebuck (Figure 8.4), quagga (Equus quagga quagga, EX), and Cape warthog (Phacochoerus aethiopicus aethiopicus, EX).

Following the demise of many of the Cape Floristic Province’s large animals, humans have driven African species to extinction at an increasing pace. Today, at least 84 Sub-Saharan African species have been confirmed Extinct (Figure 8.5), nine species are Extinct in the Wild, and as many as 202 species are considered possibly Extinct (IUCN, 2019). Among the extinct species are two wildflowers (Acalypha dikuluwensis, EX; Basananthe cupricola, EX) wiped out by mining activities in the DRC; and from Seychelles, an endemic parakeet (Psittacula wardi, EX) that was hunted to extinction. Among the species that persist only in captivity is the Kihansi spray toad (Nectophrynoides asperginis, EW), whose population crashed from more than 20,000 individuals in June 2003 to only five individuals in January 2004 after the establishment of a hydropower plant in eastern Tanzania (Channing et al., 2006). While some species that are Extinct in the Wild may be released back into the wild at some point in future, the four (or possibly seven) cycad species that persist only in captivity will probably not be reintroduced due to ongoing concerns about poaching by plant collectors (Okubamichael et al., 2016).

While most of Africa’s extinctions—at least until now—were isolated events involving one or two species at a time, the region also provides one of the best-studied examples of a recent man-made mass-extinction event. In the mid-1950s, the Uganda Game and Fisheries Department introduced the predatory Nile perch (Lates niloticus) to Lake Victoria to bolster the local fishery industry (Pringle, 2005). An ecological and economic disaster followed, pushing the entire ecosystem to the brink of collapse. First, the local people continued to prefer smaller endemic cichlids—which they could preserve by drying in the sun—to the perch with its oily flesh. This allowed the predatory perch’s population to grow unchecked which, in turn, reduced over 500 endemic cichlid species’ populations by 80% in just a few years (Witte et al., 1992). As the cichlid populations crashed, some local people started consuming perch for protein; however, they preferred smoking the perch over wood fires. To obtain firewood and charcoal, trees were logged around the lake, which in turn increased eutrophication, as well as erosion and siltation. Despite this array of emerging ecological threats, the local fishery continued to harvest the rapidly declining cichlid population. Consequently, as many as 200 cichlid species may have been driven to extinction in the decade following the perch introduction (Goldschmidt et al., 1993).
Judging by the number of extirpations over the last few decades, Africa will undoubtedly see more species pushed to extinction in the coming decades. Of particular concern is West and Southern Africa, which have lost over 75% of its large mammal populations over the past few decades; losses across Sub-Saharan Africa as a whole generally amount to over 50% (Ceballos et al., 2017). Some species will hopefully be spared this fate with the help of people and organizations fighting for their continued survival (Box 8.2). Many species will not be so lucky. The world’s last western black rhinoceros (Diceros bicornis longipes, EX) died in Cameroon in 2011; the northern white rhinoceros (Ceratotherium simum cottoni, CR) may follow this fate within the next few years (see Box 11.4). Lions (Panthera leo, EN) have been extirpated from as many as 16 African nations (Bauer et al., 2015), while cheetahs (Figure 8.6) occur in less than 9% of their historic range (Durant et al., 2017).
Jeremy Shelton
Freshwater Research Centre (FRCSA),
Kommetji, South Africa.
Lesotho’s iconic Maloti minnow (Pseudobarbus quathlambae, EN) (Figure 8.C) is a small, stream-dwelling cyprinid, and is the only freshwater fish species endemic to the country. Historically, the species was widespread, but its distribution has become increasingly restricted and fragmented in recent times due to interactions with non-native fishes and habitat degradation (Skelton et al., 2001), leading to it being classified as Endangered by the IUCN (Chakona and Kubheka, 2018). Genetic research has revealed that what was previously considered a single widespread species comprises two genetically distinct lineages: a “Mohale lineage” found in the Mohale catchment, and an “Eastern lineage” which includes populations in five catchments east of the Mohale catchment (Skelton et al., 2001). The genetic divergence between the two Maloti minnow lines is a result of a long period of geographic isolation and warrants that they be conserved as separate evolutionary significant units (ESU). Furthermore, the Mohale lineage, which comprises 77% of the species’ known distribution, is of critical importance for continued survival of the species (Skelton et al., 2001).

Past surveys (e.g. Steyn et al., 1996) have revealed that the Maloti minnow was the only fish species inhabiting the rivers flowing into the Mohale Reservoir. Situated 4 km below the Reservoir, the 20 m high Semongkoaneng waterfall has historically prevented larger fish species from moving upstream into the upper catchment. Following the filling of the Mohale Reservoir in 2003, an inter-basin transfer (IBT) tunnel linking it to Katse Reservoir was opened. Biologists working in the catchment subsequently expressed concern that non-native fishes might colonise the Mohale Reservoir via the IBT tunnel, and from there invade the influent rivers (Rall and Sephaka, 2008). Because the Maloti minnow evolved in the absence of large-bodied fishes, it would not have had an opportunity to evolve adaptations to cope with competition from and predation by larger species and may, therefore, be particularly sensitive to the arrival of other fish.
In 2006, the smallmouth yellowfish (Labeobarbus aeneus, LC), a larger, more aggressive cyprinid, was recorded in Mohale Reservoir (Rall and Sephaka, 2008), suggesting that it had dispersed from Katse Reservoir through the IBT tunnel. By 2013, it had spread into the major influent rivers in that system and coinciding with this was a virtual disappearance of the Maloti minnow from this former stronghold for the Mohale lineage. To illustrate this, surveys in previous decades described healthy populations of several thousand fish (e.g. Steyn et al., 1996), while only five individuals were recorded from the same sites in 2013 (Shelton et al., 2017).
Interestingly, the saving grace for this lineage may have originated from the same source that landed them in this predicament in the first place: human intervention. Prompted by the opening of the Kaste-Mohale IBT, a small team of passionate conservation scientists translocated several Maloti minnows to sections of stream above tall waterfalls, upstream of their natural distribution range (Rall and Sephaka, 2008). These sections, they knew, would be unreachable by larger species swimming upstream from Mohale Reservoir. This assisted colonisation approach has been viewed as controversial, but it may also have saved a tiny minnow from almost certain extinction in the wild. The prospect of losing a charismatic species like the Maloti minnow showcases how projects like the Lesotho Highlands Water Project can easily damage sensitive ecosystems that were not consider in development plans. In order to save the Maloti minnow from extinction, the next step will be to assess the success of translocation efforts and develop a rescue plan for the species.



