6.6: Fatty Acid Synthesis
- Page ID
- 16126
This anabolic process is accomplished using a different set of enzymes than the catabolism of fatty acids discussed earlier. Fatty acid synthesis (Figure \(\PageIndex{11}\)) starts with the formation of palmitic acid (C16) from acetyl-CoA and malonyl-CoA (which is itself a 3-carbon molecule formed from acetyl-CoA). Another difference between the catabolic and anabolic reactions for fatty acids is the location: whereas we saw that catabolism occurs largely in the mitochondria, fatty acid synthesis is run from a single large cytoplasmic enzyme complex. The fatty acid synthase system is comprised of seven enzymes linked together with an acyl carrier protein (ACP). As mentioned, this complex is found in the cytoplasm, so its substrates must be as well. The acetyl-CoA in the cytoplasm is primarily derived from the mitochondrial acetyl-CoA via a citrate-malate shuttle that couples deacetylation in the mitochondrion with acetylation in the cytosol.
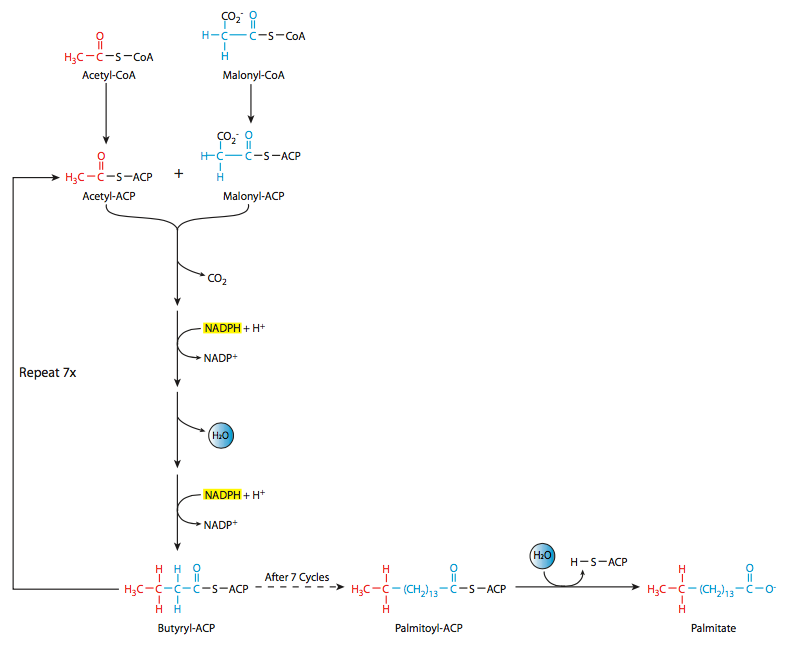
The acetyl-CoA and malonyl-CoA are linked to the synthase and ACP, then there is a sequence of acetyl group transfers that runs a total of seven times to form palmitoyl-ACP, from which the palmitic acid is finally released. Palmitic acid is the precursor for variety of long-chain fatty acids such as stearic acid, palmitoleic acid, and oleic acid. Generally, there is either an elongation or sometimes a desaturation step. However, desaturation is a tricky process for vertebrates. The desaturation at C9 to form oleic acid from stearic acid can occur; however, other desaturations such as desaturation at C-12 to generate linoleic acid are not possible in vertebrates. Interestingly, they can be carried out in plant species. Furthermore, even though linoleic acid cannot be synthesized by vertebrates, it is nevertheless needed by vertebrates, which build arachidonic acid, prostaglandins, and other molecules from it. Linoleic acid is therefore considered an essential fatty acid, since it must be ingested by the animal.
These fatty acids are then used to form the triacylglycerols that form the bulk of the energy storage molecules in most animals. Triacylglycerols are synthesized by the reaction of fatty acyl-CoA chains with glycerol-3-phosphate. Two rounds of this reaction yields diacylglygerol-3-phosphate (phosphatidic acid). After the action of phosphatidate phosphatase, the phosphatidic acid is converted to 1,2-diacylglycerol. This reacts with fatty acyl-CoA to form the final triacyglycerol.
Each of the fatty acyl chain additions generates an ester bond, which requires a significant energy input: that energy comes from a linked ATP hydrolysis reaction for each chain addition.


