6.2: Glycolysis (from the Greek glyco (sugar) lysis (Separation), or Sugar Breakdown
- Page ID
- 16444
One of the properties of life is that living things require energy. The pathways of energy flow through life are shown below.

To begin with, the most common intracellular energy currency with which livings things “pay” for cellular work is ATP. The energy to make ATP on planet earth ultimately comes from the sun via photosynthesis. Recall that light energy fuels the formation of glucose and O2 from CO2 and water in green plants, algae, cyanobacteria and a few other bacteria. Photosynthesis even produces some ATP directly, but not enough to fuel all cellular and organismic growth and metabolism. So all cells, even plant cells, use fermentation or respiration (anaerobic or aerobic processes respectively) to capture nutrient free energy (mostly) as ATP.
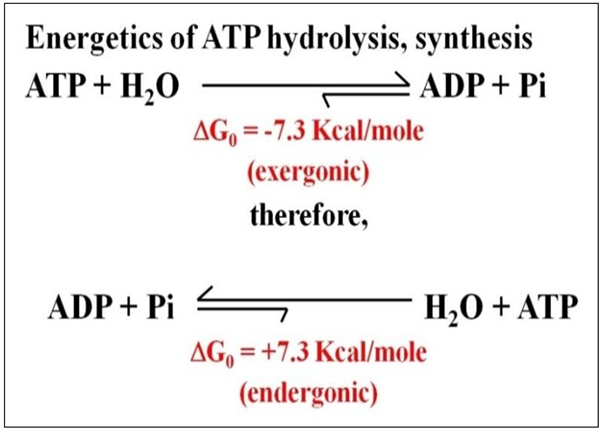
ATP is called a high-energy intermediate because its hydrolysis releases a large amount to free energy. In the condensation reactions that make ATP, it takes about 7.3 Kcal of free energy to link a phosphate to ADP in a phosphate ester linkage. Having captured nutrient free energy in a form that cells can use, ATP hydrolysis releases that free energy to fuel cellular work, including bending cilia, whipping flagella, contracting muscles, transmitting neural information, building polymers from monomers, and more. The energetics of ATP hydrolysis and synthesis are summarized below.
The free energy needed to make ATP in animal cells comes exclusively from nutrients (sugars, fats, proteins). As noted, plants get free energy directly from sunlight, but they mobilize nutrient free energy they make in much the same way as the rest of us get it from what we eat! Glucose oxidation releases a considerable amount of free energy, enough to synthesize many molecules of ATP, as shown below.

Cellular respiration, the oxidation of glucose, starts with glycolysis. Otto Myerhoff and Archibald V. Hill shared a Nobel Prize in Physiology or Medicine with in 1923 for isolating enzymes of glucose metabolism from muscle cells. Thanks to the efforts of others (e.g., Gustav Embden, Otto Meyerhof, Otto Warburg, Gerty Cori, Carl Cori), all the enzymes and reactions of the glycolyitic pathway were known by 1940, and the pathway became known as the Embden-Myerhoff Pathway. As we will see, glycolysis is an evolutionarily conserved biochemical pathway used by all organisms to capture a small amount of nutrient free energy. For more detail, check out Fothergill-Gilmore LA [(1986) The evolution of the glycolytic pathway. Trends Biochem. Sci. 11:47-51]. The glycolytic pathway occurs in the cytosol of cells where it breaks down each molecule of glucose (C6H12O6) into two molecules of pyruvic acid (pyruvate; CH3COCOOH). This occurs in two stages, capturing nutrient free energy in two ATP molecules per glucose molecule that enters the pathway.
Glycolytic reactions are summarized below, highlighting the two stages of the pathway.

Stage 1 of glycolysis actually consumes ATP. Phosphates are transferred from ATP first to glucose and then to fructose-6-phosphate, reactions catalyzed by hexokinase and phosphofructokinase respectively. So, these Stage 1 phosphorylations consume free energy. Later, in Stage 2 of glycolysis, nutrient free energy is captured in ATP and NADH (reduced nicotinamide adenine dinucleotide). NADH forms in redox reactions in which NAD+ is reduced as some metabolite is oxidized. In Stage 2, it is glyceraldehyde- 3-phosphate that is oxidized…, but more later!
In fact, by the end of glycolysis, four molecules of ATP and two of NADH have been formed and a single starting glucose molecule has been split into two molecules of pyruvate. Pyruvate will be metabolized either anaerobically or aerobically.
The alternate fates of pyruvate are summarized below.

Anaerobic (complete) glycolysis is a fermentation pathway. In anaerobic glycolysis the electrons in NADH produced in Stage 2 of glycolysis are used to reduce pyruvate, so that in the end, there is no consumption of O2 and no net oxidation of nutrient (i.e., glucose). A familiar anaerobic glycolytic pathway is the production of alcohol by yeast in the absence of oxygen. Another one is the muscle fatigue you might have experienced after especially vigorous and prolonged exercise. This results from a fermentation that produces an anaerobic build-up of lactic acid in skeletal muscle cells. In anaerobic glycolysis, the reduction of pyruvate can lead to one of several other fermentation end products, along with a net yield of two ATPs per glucose fermented.
Aerobic (incomplete) glycolysis also produced two ATPs, and is the first step in the complete oxidation of glucose, the respiration pathway oxidizing glucose to CO2 and H2O, leaving no carbohydrates behind. Pyruvate is completely oxidized in mitochondria. As we look at the reactions of glycolysis and the Krebs cycle, watch for redox reactions in both pathways.
Along the way, we’ll also consider Gluconeogenesis, a pathway that essentially reverses the glycolysis and results in glucose synthesis. Gluconeogenesis occurs both under normal conditions, during in high-protein/low carb diets, and during fasting or starvation. In another chapter, we’ll look at electron transport and oxidative phosphorylation, the pathways that complete the oxidation of glucose. Here, we begin with a closer look at glycolysis, focusing on the enzyme-catalyzed reactions and free energy transfers between pathway components. We will consider the energetics and enzymatic features of each reaction.


