11.2: Light and Photosynthesis
- Page ID
- 93187
- Understand the meaning of photoautotroph in reference to plants.
- Explain how the energy from light is converted into carbon-based chemical energy and building blocks in plants.
- Identify where in the plant the various photosynthetic reactions take place.
- Explain how the carbon-based building blocks move to other parts of the plant and are used for energy, storage, and structures.
Photoautotrophs
Plants are autotrophs, meaning that they are self-nourishing (Greek autos = self and trophe = nutrition). Specifically, plants are photoautotrophs, because they use the energy from light to produce organic molecules with which they build their cells and store energy.
Organic molecules are compounds associated with living organisms that contain carbon atoms. It was once thought that organic molecules could only be synthesized in nature by living organisms through the intervention of a “life force.” This hypothesis was disproved in 1828 when urea, a simple organic compound, was synthesized in a laboratory. Since that time, a major branch of chemistry, organic chemistry, has arisen to study and synthesize organic molecules. In contrast to organic compounds, inorganic compounds were historically defined as those lifeless minerals that are dug up from the ground.
Note that this chemical definition of organic (containing carbon atoms) has little or no relationship to the contemporary use of the word to describe a method of producing food. Organic food production, by regulation, relies strictly on inputs of organic molecules that come from life (like manure) and also on inorganic compounds like minerals, and eschews the use of organic molecules that have been synthesized by humans.
The organic molecules that a plant produces must be:
- Storable within the plant.
- Metabolized by the plant to yield energy for use in growth, maintenance, and producing other required organic molecules.
- Reasonably compact so that enough energy can be stored for growth.
- Transportable within the plant.
- Stable and non-toxic to the plant.
Since plants are photoautotrophs, they must have a mechanism for capturing energy from the sun or other sources of light and using that energy to produce organic molecules with the characteristics noted above. Photosynthesis is the process on which photoautotrophs rely to capture that light energy and to produce carbon-based organic molecules. The carbon used to make these molecules comes from the carbon dioxide (CO2) in the atmosphere. Because photosynthesis removes carbon from the atmosphere and incorporates it into organic molecules which eventually become the plant’s leaves, stems, roots, and fruits, photosynthesis is sometimes said to fix carbon. Fix, in this sense, means to secure or sequester rather than to repair.
If you follow the public discourse on climate change, you are aware that global warming is accelerated by the accumulation of greenhouse gasses in the atmosphere which trap and re-radiate sunlight and heat back to the earth. CO2 is one of these greenhouse gasses. Removal of CO2 from the atmosphere, for instance by planting trees that photosynthesize, fix carbon, and store the carbon-rich product as wood, is one method of carbon sequestration. An emerging and increasingly popular strategy for remediating greenhouse gas emissions is through the buying and selling of carbon credits, where industries that discharge CO2 into the atmosphere purchase credits from organizations whose activities (such as tree planting) sequester carbon. Photosynthesis and sequestration of carbon by trees is one tool used to offset the industrial release of CO2.
- In what sense does photosynthesis fix carbon?
- Where does the carbon come from that is used by photosynthesis, and where does it go within the plant?
Light reaction
Let’s start with light, because that’s where the plant gets the energy for photosynthesis. Here are some characteristics of light:
- Light travels in waves.
- The length of the wave is measured from one peak to the next and is called the wavelength, which differs for different colors of light.

Visible spectrum. Philip Ronan. CC BY-SA 3.0 - Within the visible wavelengths of light, the longest wavelengths are red light; outside the visible range of wavelengths, even longer wavelengths include infrared radiation, microwaves, and radio waves.
- Shorter visible wavelengths include blue and purple light, and beyond the visible range even shorter wavelengths include UV light, X-rays, and Gamma rays
- Light also has a particulate nature, and those particles are called photons.
The photons in light provide the energy that drives photosynthesis. This energy is used to incorporate carbon found in CO2 from the atmosphere into organic molecules and, in particular, into simple sugars used by the plant. The chemical formula is the same for the two types of simple sugars produced by photosynthesis: glucose and fructose: C6H12O6. The equation that summarizes photosynthesis is:
water + carbon dioxide -> oxygen, water, and simple sugars
12H20 + 6CO2 -> 6O2 + 6H2O + C6H12O6
This balanced equation tells us that 12 molecules of water plus 6 molecules of carbon dioxide, in the presence of chlorophyll, accessory pigments, and light, produces 6 molecules of oxygen gas, returns 6 molecules of water back to the cell, and produces one molecule of a simple sugar like glucose or fructose.
Two reactions make up photosynthesis: the Light Reaction (abbreviated LR) and the Light Independent Reaction (abbreviated LIR). As the names suggest, the LR requires light while the LIR does not. The LR uses light energy to split water, which transforms the energy from the sun into hydrogen ions and electrons. The LIR uses that energy to grab the carbon from carbon dioxide and use the carbon to build simple sugars.
Let’s start with the light reaction. You’ve heard of chlorophyll, and may recognize this molecule as a green pigment that captures light for photosynthesis. There are two chlorophyll pigments in plants that are critical for absorbing light: Chlorophyll a and Chlorophyll b.
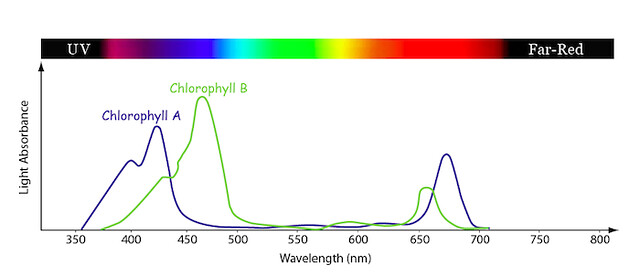
The graph above shows % absorbance of different wavelengths by these two chlorophylls. The Y axis (the vertical one) shows the percentage of the light that is absorbed (rather than reflected). High levels of absorption mean that the chlorophyll molecule uses that wavelength of light for energy. Low absorption means that the molecule does not use that wavelength, and is thus reflected away. The X axis indicates the wavelength of light in nanometers (nm) (the wavelength of green light, roughly 500 nm). The bar at the top represents the color of the light at the wavelength shown. The blue line is a typical absorption curve for chlorophyll a, while the green line shows chlorophyll b.
High absorbance at a particular wavelength means that pigment is collecting that light at that wavelength to harvest energy. Low absorbance means that the plant is reflecting that light back. Both chlorophyll a and b absorb blue and red light wavelengths and reflect green. Chlorophyll a has a peak in the violet and red regions and chlorophyll b in the blue and orange regions. Notice how their absorbance is very low in the green region. That’s why we think of chlorophyll as green, and why we perceive leaves, which have chlorophyll as the predominant pigment, as green. Also notice that chlorophyll reflects some yellow wavelengths, but when the yellow and deep green wavelengths are mixed, we see the green leaf color.

The graph above shows the absorbance of carotenoid pigments, which are present throughout the growing season. Carotenoids are called an accessory pigment in photosynthesis. They assist chlorophyll in light capture and energy transfer, and contribute to the regulation and moderation of excessive excitation of pigment molecules during intense sunlight, including exposure to UV light. Carotenoids absorb light in the green range, but reflect in yellow and red. We don’t see these pigments during the growing season because they are much lower in concentration than the chlorophylls, so the green reflected light overwhelms the orange, and we see green. But when the chlorophyll fades in the fall, due to decomposition of chlorophyll, the orange can be seen in beautiful fall leaf colors.
Chlorophyll a and b, as well as the accessory pigments, are found in the chloroplasts, which are membrane-bound organelles within cells. The highest concentration of chloroplasts is most commonly found in the palisade mesophyll cells of the leaf.
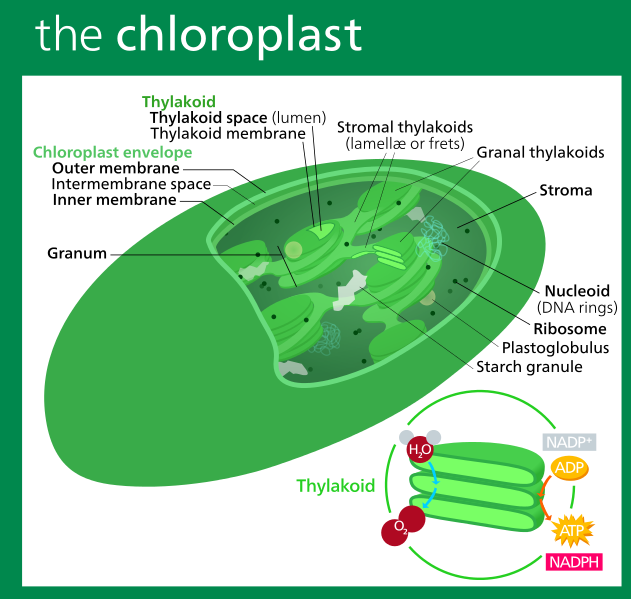
The above illustration of a chloroplast labels the internal structures. The chloroplast has a double membrane. The interior of the chloroplast is called the stroma. Within the stroma are coin-like thylakoids. The stacks of thylakoids are called grana. The thylakoids are also surrounded by a membrane, called the thylakoid membrane. The green chlorophyll pigment that you associate with photosynthesis, as well as the accessory pigments, are embedded in the thylakoid membranes and arranged in a structure called the antenna complex — given this name because it captures and routes the energy from sunlight to a collector called a reaction center.
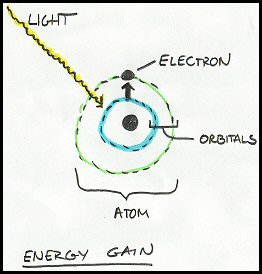
As shown above, when light hits a pigment molecule in the antenna complex, the energy from the light photon promotes (pushes up) an electron in one of the pigment’s atoms to a higher orbital as seen in the cartoon and energy is gained.

The electron can’t stay in that higher orbital indefinitely, and when it drops back to its home orbital it releases the energy it absorbed from light, denoted as energy loss. This released energy can be passed to another pigment molecule. This process of one pigment capturing the photon’s energy and passing that energy onto adjacent pigment molecules is the crucial step in energy transformation that takes place in photosynthesis. This is the step that takes light energy and converts it into chemical energy — one of the only known biological processes that allows this type of energy transformation.

A light photon excites an electron of one pigment molecule in the antenna complex, or light harvesting complex, and by resonance this energy is transferred from pigment molecule to pigment molecule The energy transfer makes its way to the reaction center, where the first major chemical reaction in photosynthesis — splitting water — takes place. This reaction is called the light reaction or light-dependent reaction because it requires light. Water is split when the reaction center grabs electrons from water, which separates water into oxygen gas (O2), hydrogen ions (H+), and electrons (e–).
To reiterate, the light is captured by the light harvesting complex (antenna complex) where electrons in the chlorophyll atoms are excited and jump up to a higher orbital. When the electron drops back, the energy is transferred to an adjacent pigment atom. This resonance energy travels down the antenna complex to the reaction center, where the captured energy pulls electrons out of water molecules, and water is split into oxygen gas, hydrogen ions, and electrons. The energy that was present in the photons of light has been transferred to the hydrogen ions and the electrons. We’ll see more of how that energy is used in the next section.
- What wavelength(s) of light does chlorophyll a absorb? Chlorophyll b? What wavelengths do these two molecules reflect?
- What pigments make up the antenna complex?
- How is the energy in light transformed in the Light Reaction?
Recall that the overall equation for photosynthesis is:
water + carbon dioxide -> oxygen, water, and simple sugars
12H20 + 6CO2 -> 6O2 + 6H2O + C6H12O6
This equation is made up of two parts called half-reactions. The first half-reaction is an equation summarizing the Light Reaction, where energy from sunlight is used to split water molecules into oxygen gas, some electrons, and some hydrogen ions. The energy from sunlight is transferred from the pigments to these hydrogen ions and electrons. The half-reaction for the Light Reaction is as follows:
12H2O -> 6O2 + 24e– + 24H+
Light independent reaction
The Light-Independent Reaction (LIR) is the second part of photosynthesis. It takes place in the stroma of the chloroplast. Unlike the Light Reaction, it does not require light. In the LIR, two compounds, NADPH and ATP, carry the energy from light that was originally transformed into hydrogen ions and electrons through the splitting of water. The NADPH and ATP, along with carbon dioxide from the atmosphere, enter a process called the Calvin Cycle, where the energy is used to fix carbon into a molecule abbreviated G3P. This process requires the help of an important protein abbreviated RuBisCO (Ribulose-1,5-bisphosphate carboxylase/oxygenase) that catalyzes the step in the process where the carbon from atmospheric CO2 is incorporated into an organic molecule. RuBisCO is the most abundant protein in leaves and, given the number of leaves in the world, likely the most abundant protein on the planet. The G3P produced by the carbon fixation process is called a triose phosphate, meaning it is a 3-carbon sugar (triose) with phosphorus and oxygen atoms (phosphate) attached. Triose phosphate moves out of the chloroplast into the mesophyll cell’s cytoplasm, where two of these three-carbon molecules are combined to produce the 6-carbon molecules glucose and fructose. The glucose and fructose molecules then combine to form sucrose, a 12-carbon organic molecule. Sucrose is important because it is the sugar that is transported by the phloem throughout the plant to provide energy and building blocks for other organic molecules like starch and cellulose.
The half-reaction for the LIR is:
24H+ + 24e– + 6CO2 -> C6H12O6 + 6H2O
- Does the Light Independent Reaction require darkness?
- What sugar is moved throughout the plant through the phloem?
Photosynthesis summary
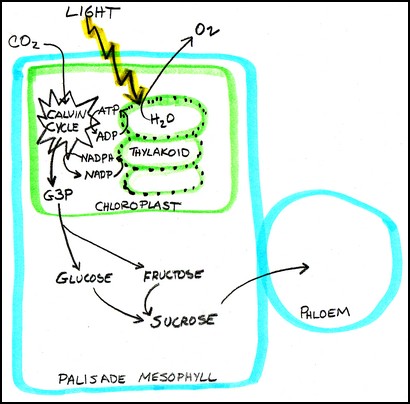
When we add the two half-reaction equations for LR and LIR together, we get back to the summary equation for photosynthesis:
12H2O + 6CO2 -> 6O2 + 6H2O + C6H12O6
The illustration above is a summary of what happens in a mesophyll cell. The rectangular blue outline represents a palisade mesophyll cell in a leaf. Inside the cell is a green rectangle, representing a chloroplast. Inside the chloroplast is a stack of green ovals with black dots. These ovals are the thylakoids, and the stacks are grana. The black dots in the green thylakoid membrane represent the antenna complexes. Light hits the antenna complex and transfers its energy to pigments, and the energy is funneled to the reaction center where water (H2O) is split in the light reaction to form the energy carriers ATP and NADPH. This is the Light Reaction. The waste product formed at this stage is oxygen, which might be waste for the plant, but is quite useful for us.
In the Light Independent Reaction the energy is carried to the Calvin Cycle, represented by the multi-pointed star in the chloroplast, which uses the energy in ATP, the NADPH, and CO2 from the atmosphere to form the three-carbon G3P triose phosphate with the help of RuBisCO. Triose phosphate leaves the chloroplast and passes into the cytoplasm of the mesophyll cell to be transformed into glucose and fructose, which are combined into sucrose that is exported from the mesophyll cell to the phloem.
Cellulose and starch
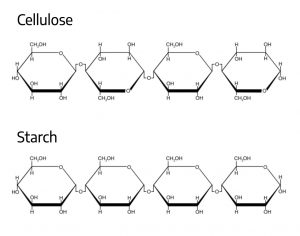
Within a plant, the regions of photosynthesis and sugar production are called the source. Leaves are typically the main source within the plant, since that is where most photosynthesis takes place. Those regions that do not support photosynthesis (like roots), but that still need organic molecules to survive, are called sinks. Movement of solutes (molecules dissolved in water) like sucrose from source to sink through the phloem is called translocation. Translocation of sucrose through the phloem to the sink provides cells with a source of stored energy, and also building blocks for organic molecules, as noted earlier. Sucrose can be broken down to glucose and fructose, building blocks used to form other extremely useful organic compounds. Two particularly useful compounds result from the production of long glucose chains: starch, a key energy storage compound in plant cells, and cellulose, the main constituent of the cell wall and key to a plant’s structural integrity. Wood, for instance, is primarily made up of the cellulose-rich cell walls of dead xylem. Both starch and cellulose are long chains of glucose, but they differ in the way the glucose molecules are linked together.
Cellulose is the molecule into which carbon extracted from atmospheric CO2 is sequestered for long-term storage. Starch sequesters carbon for a much shorter period of time because it is either eaten, used by the plant for new growth, or decomposed by bacteria and fungi that can utilize starch for energy.
- Define translocation — what molecules are being transported?
To review
- In the light reaction, pigments in the thylakoid membrane capture energy from sunlight.
- The energy is used to split water, which releases oxygen to the atmosphere.
- The energy used to split water is transferred into electrons and hydrogen atoms, and eventually to ATP and NADPH.
- In the light independent reaction, the ATP and NADPH power the Calvin cycle that captures carbon from atmospheric CO2 and incorporates it into simple sugar molecules.
- These simple sugars can be translocated to sinks, where they are used for energy, converted into energy storage compounds, or converted into structural molecules.
Some of the explanations and pictures used in this lecture came from Introduction to photosynthesis and other pages at that McDaniel College site. For additional explanations, an overview of the chemical reactions of photosynthesis, and another description of the properties of light and pigments, navigate to the pages listed at the top of that web page.


