2.3: Extrachromosomal elements, plasmids, selectable markers
- Page ID
- 18128
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)
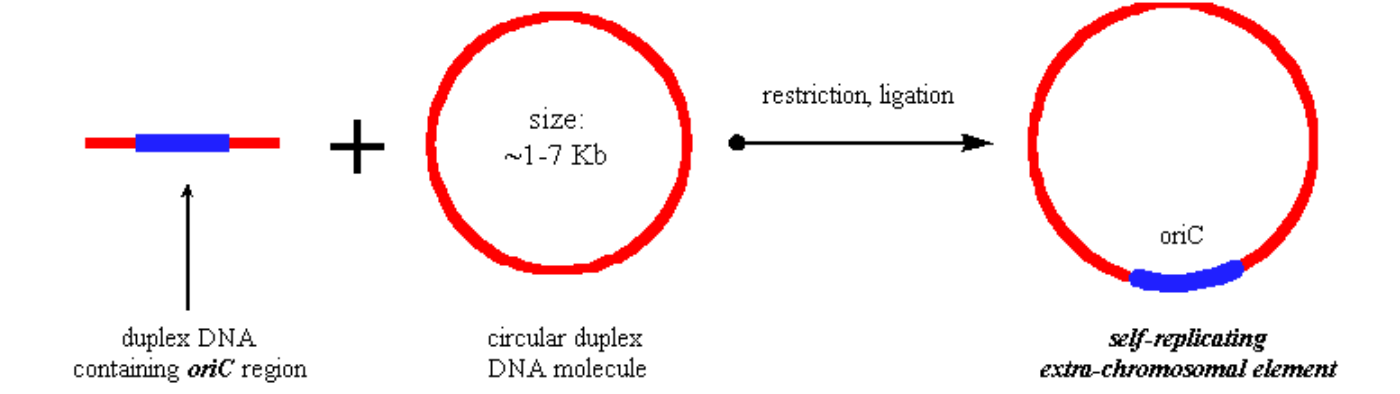
- Including an origin of replication (i.e. the E. coli oriC region) into a circular DNA molecule is a mechanism to have an extrachromosomal element in the prokaryotic cell.
.png?revision=1&size=bestfit&width=684&height=192)
Figure 2.3.1: oriC Extrachromosomal element
- Such an extrachromosomal element is called a plasmid, or vector
- The plasmid uses the host cell machinery (i.e. polymerases, helicases, dNTP's etc.) to direct replication.
.png?revision=1&size=bestfit&width=413&height=359)
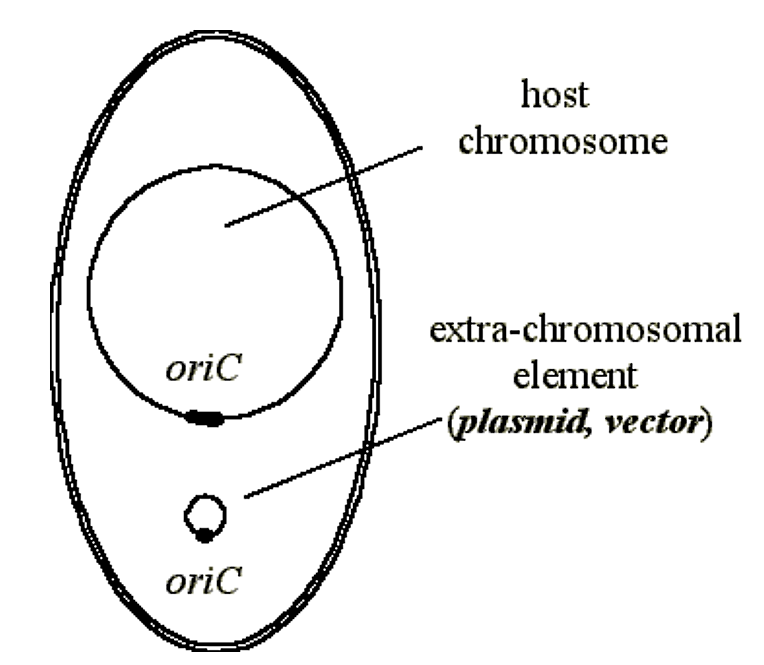
Figure 2.3.2: Vector/Plasmid
- However, since the added work of replicating the extrachromosomal element is a load on a cell, it will be out-competed by other cells which do not contain the plasmid.
- Since in prokaryotic cells the segregation of plasmids is a random event, daughter cells can arise which do not contain the plasmid and these grow faster (out-compete) the parent cell.
- In other words, in the absence of other pressures, after a period of time the population of cells in a culture will be those which have "lost" the plasmid.
.png?revision=1&size=bestfit&width=683&height=265)
Figure 2.3.3: Loss of plasmid
- In organisms with more than one chromosome (eukaryotes) there are a variety of mechanisms to ensure that proper segregation of chromosomes occurs, i.e. to make sure that daughter cells contain equal numbers of all the chromosomes.
- One basic mechanism is that each chromosome contains essential genes, and if these are lost, the cell cannot survive.
Drug resistance
- By far the most common approach to the maintenance of plasmids is through the incorporation of drug resistance genes.
- These are also known as selectable markers, i.e. we can select for their presence by including antibiotics in the growth media.
Ampicillin
- Ampicillin binds to and inhibits a number of enzymes in the bacterial membrane that are involved in the synthesis of the gram negative cell wall.
- Therefore, proper cell replication cannot occur in the presence of ampicillin.
- The ampicillin resistance gene (ampr) codes for an enzyme (b-lactamase) that is secreted into the periplasmic space of the bacterium where it catalyzes hydrolysis of the b-lactam ring of the ampicillin.
- Thus, the gene product of the ampr gene destroys the antibiotic.
- Over time the ampicillin in a culture medium or petri plate may be substantially destroyed by b-lactamase.
- When this occurs, cell populations can arise which have "lost" the plasmid.
.png?revision=1&size=bestfit&width=646&height=297)
Figure 2.3.4: Ampicillin resistance
Tetracycline
- Tetracycline binds to a protein of the 30S subunit of the ribosome and inhibits ribosomal translocation along the messenger RNA which codes for the protein (i.e. the drug interferes with normal translation or production of proteins).
- The tetracycline resistance gene (tetr) encodes a 399 amino acid outer membrane associated protein of gram negative cells that prevents the antibiotic from entering the cell.
- Thus, this drug resistance gene does not destroy the antibiotic. Pressure will be maintained throughout the cell culture process to keep the plasmid containing the drug resistant gene.
Chloramphenicol
- Chloramphenicol binds to the ribosomal 50S subunit and inhibits protein synthesis.
- The chloramphenicol resistance gene (Cmr) codes for a protein known at the cat protein.
- The cat protein is a tetrameric cytosolic protein that, in the presence of acetyl coenzyme A, catalyzes the formation of hydroxyl acetoxy derivatives of chloramphenicol that are unable to bind to the ribosome.
- Like with ampicillin, the Cmr gene product destroys the antibiotic.
- Additionally, the expression of cat protein is influenced (down regulated) by the presence of glucose in the media.
Kanamycin and neomycin
- Bind to ribosomal components and inhibits protein synthesis.
- The Kanr gene codes for a protein which is secreted into the periplasmic space and interferes with the transport of these antibiotics into the cell.
- Like tetracycline resistance, the Kanr gene does not destroy the antibiotic.
Colicin E1
- This is a member of a general class of substances known as bacteriocins.
- Colicin E1 causes lethal membrane changes in bacteria.
- The drug resistance gene (cea) codes for a protein that interferes with the action of colicin in an unknown manner.
Prokaryotic plasmids
- In addition to making a plasmid by using the E. coli OriC region, there is a naturally occuring E. coli plasmid called the ColE1 plasmid
- The ColE1 origin of replication is uni-directional (unlike oriC)
- The replication from the ColE1 ori region does not require the associated proteins (e.g. dnaA protein) like oriC, (but it does require specific RNA molecules).
- Depending on the exact region of the ColE1 origin which is inserted into a circular dna molecule, the extrachromosomal element will be maintained with either a "low" or "high" copy number
- The rop gene near the ColE1 origin is involved in the regulation of replication.
- If the ColE1 ori region includes this gene then the plasmid is maintained with an average copy number of 10-30 plasmids/cell. This is considered low copy number.
- If the ColE1 ori region does not have the rop gene, then the resulting plasmid is maintained with an average copy number of 100-200 plasmids/cell. This is considered a high copy number type of plasmid.
- If the plasmid contains a gene coding for a protein (like drug resistance genes) copy number can influence the amount of such protein in the cell.
pBR322 (4.36 Kb)
- One of the original cloning plasmids.
- Constructed by ligating together:
- the tetracycline resistance gene from plasmid pSC101
- ColE1 and rop region from the ColE1 plasmid
- the ampicillin resistance gene from the transposon Tn3
- Contains both ampicillin and tetracycline resistance genes (markers).
- Contains unique restriction sites inside and outside of these markers.
- Contains rop region near CoE1 ori , therefore, it has a low copy number (10-30)
- Numbering begins at the unique EcoR I restriction site (GAATTC). The first 'T' in this sequence is base number "1".
.png?revision=1&size=bestfit&width=509&height=446)
Figure 2.3.5: pBR322
The plasmid diagram:
- In the center is the plasmid name (usually starts with a lower case 'p') and the size in basepairs
- the inner ring provides ticks in 1 Kilobase (Kb) intervals to give an idea of the general location of parts of the plasmid
- the arrows indicate genes, markers, ori or replication, promoters, polylinkers, transcription terminators, and other important or functional items
- the outer ring usually indicates the location of unique, or limited number (usually <3), restriction endonuclease sites. Restriction enzymes which have more than three sites will not be indicated. Be aware that enzymes which do not cut at all will also not be listed!
PUC18/19 (2.69 Kb)
- Lacks the rop gene near the ColE1 ori region. Thus, this plasmid tends to accumulate in high copy number (100-200).
- This vector contains only the ampicillin resistance marker.
- This vector contains a polylinker region
- A synthetic DNA sequence which contains a clustering of unique restriction enzyme recognition sites
- Allows DNA fragments, generated by a variety of restriction endonuclease cleavages, to be inserted into the plasmid
- pUC18 has the polylinker in one orientation
- pUC19 has the same polylinker, but in the opposite orientation
- Thus, fragments with unique restriction sites on each end can be inserted in a specific orientation
- The Pst I site in the ampr gene was mutated to remove it. The EcoR I site at position (1) was mutated to remove it. This was done to make the restriction sites in the polylinker region unique
- This vector also contains a transcription promoter region from the lac operon, which allows foreign genes to be inserted and transcribed/translated.
- The polylinker region is just downstream (3') to the lac promoter
- Inserted genes can be transcribed from this promoter
.png?revision=1&size=bestfit&width=535&height=426)
Figure 2.3.6: pUC18/19


