2.6: Chapter 2 Questions
- Page ID
- 83788
Section 1 Questions:
Q1) Based on the interactive figure 2.1.1 in the default view, do you hypothesize that water could enter the core of the micelle? Now, let's test your hypothesis! Open the interactive figure, and click Style → Surface Type → Solvent Accessibility. Based on the space-filled solvent accessibility map, do you see any openings for water molecules to enter through? Explain your answer.
A1) Hypothesis can range from no water is accessible, or water could freely occupy the "empty" space in the default view of the micelle. However, once the solvent accessibility filter is applied, it is clear that the micelle will exclude water from entering the hydrophobic core. Some areas of the micelle (the polar head groups) are able to for interactions with the water and therefore show up as red/yellow in this view, but it is key to note there are no pores or solvent-accessible spaces on the surface of the micelle.
Q2) The structure for Limonene, the compound that gives citrus fruits their classic smell is shown below. Examine the structure and answer the following questions.
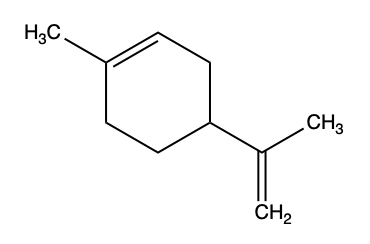
a) Will Limonene form any associations with water molecules? Explain.
b) For our brain to register the citrus aroma, Limonene needs to bind to a surface protein receptor, what type of interactions could be taking place? Explain.
c) When Limonene binds to its receptor are the interactions from b) stabilized by enthalpy ΔH or entropy ΔS?
A2)
a) No, there are no dipoles or polar functional groups on limonene.
b) The methyl and ethyl functional groups would likely form hydrophobic bonds with nonpolar amino acids in the protein. Because there are no polar/dipole residues, Limonene cannot make hydrogen or ionic bonds.
c) Binding of Limonene to its surface receptor will likely be stabilized by entropy. When the nonpolar Limonene binds to a nonpolar region of the surface receptor, order water will be released, thus creating a +ΔS.
Q3) Consider the following reaction of a polar substance and water at room temperature (22°C):
Glucose(s) + H2O(l) → Glucose(aq)
a) Estimate the enthalpy ΔH and entropy ΔS (+,-,≈0) for the reactants and product. Consider the order/disorder of the over all reaction as well as the net charge of the bond enthalpy.
b) Based on the Gibbs free energy equation ΔG = ΔH - TΔS, will this reaction be spontaneous, non-spontaneous, or at equilibrium?
A3)
a) Glucose(s) + H2O(aq) → Glucose(aq)
ΔH: H-bonds H-bonds H-bonds ΔH ≈ 0
ΔS: Solid Liquid Solution ΔS = +
b) ΔG = ΔH - TΔS
= 0 - (+)
= -
This dissolution of polar substances into water is spontaneous.
Q4) Consider the following reaction of a nonpolar substance and water at room temperature (22°C):
Methane (CH4)(g) + H2O(l) → Methane (CH4)(aq)
a) Estimate the enthalpy ΔH and entropy ΔS (+,-,≈0) for the reactants and product. Consider the order/disorder of the overall reaction as well as the net charge of the bond enthalpy.
b) Based on the Gibbs free energy equation ΔG = ΔH - TΔS, will this reaction be spontaneous, non-spontaneous, or at equilibrium?
A4)
a) Methane (CH4)(g) + H2O(l) → Methane (CH4)(aq)
ΔH: None H-bonds H-bonds ΔH ≈ 0 or slightly -
ΔS: Gas Liquid Solution ΔS = -
b) ΔG = ΔH - TΔS
= 0/- - (-)
= +
This dissolution of nonpolar substances into water is non-spontaneous. This is primarily due to water forming an ordered cage around the nonpolar methane gas. In terms of enthalpy, the water cage creates a more favorable environment for methane than the pure gas in liquid water, thus the enthalpy can be considered slightly negative. However, the cage structure the water molecules form around the methane gas makes the system ordered. Therefore the overall ΔG for nonpolar substances solubilized with water is positive and therefore non-spontaneous.
Need to add in some reactions? (last chunk of section 1)
Section 2 Questions
Q1) Tyrosine is commonly found in the active sites of enzymes, as the unique structure of its R-group can act as either an acid or base.
a) The enzyme DcpS, an mRNA capping enzyme, utilizes a tyrosine residue as an active site acid, and measurements show that the tyrosine is 75% ionized. What must the local pH need to be for this to occur?
b) Another way for tyrosine to be used as an acid is to lower its pKa by creating weak interactions with the oxygen of its R-group using neighboring amino acids. What type of interaction(s) could achieve this goal?
c) One enzyme utilizing the strategy in b) is Glutathione-S-Transferase. Measurements show that the active site pH is 7.5, and the tyrosine residue is 95% ionized. What must the pKa of tyrosine be for this to occur?
A1)
a) \[pH = pK_a + log \dfrac{[A^{-}]}{[HA]} \nonumber \]
\[pH = 10.1+ log \dfrac{[75]}{[25]} \nonumber \]
pH = 10.1 + (0.477)
pH = 10.58
b) Hydrogen bonding can create a change in the net dipole of the oxygen in tyrosine by making it more acidic and decreasing the pKa.
c) \[pH = pK_a + log \dfrac{[A^{-}]}{[HA]} \nonumber \]
\[7.5 = pK_a + log \dfrac{[95]}{[5]} \nonumber \]
7.5 = pKa + 1.28
6.22 = pKa
Section 3 Questions
Q1) Your lab wants to study an enzyme that catalyzes a reaction inside the chloroplast stroma, which has a pH of 8.0 due to the proton gradient that pumps H+ from the stroma to the thylakoid lumen.
a) Which of the buffers below would be the best choice to study this enzyme, in vitro? Explain your choice. Are there any other buffers that could work?
-Tricine: pKa 8.05
-TAPS: pKa 8.40
-MES: pKa 6.1
-Citrate: pKa 6.40
-HEPES: pKa 7.48
b) How many moles of the conjugate base form of HEPES would there be in 2.5 L of a 175 mM solution at pH 8.0?
A1)
a) Tricine would be the best choice as buffers work best at a pH closest to their pKa. TAPS or HEPES could work in a pinch if Tricine was not available, but remember, pH and pKa are log scales! So, while the pKa values might not seem that far from the intended pH of 8.0, on the log scale that is quite a difference in H+ concentration.
b) \[2.5 L × \dfrac{175 mmoles}{L}\ × \dfrac{1 mole}{1000 mmoles}\ = 0.4375 mmoles \nonumber \]
Now we want to determine what fraction of those moles are in the conjugate base form:
\[[A^{-}] + [HA] = 0.4375 \nonumber \]
\[[A^{-}] = 0.4375 - [HA] \nonumber \]
\[pH = pK_a + log \dfrac{[A^{-}]}{[HA]} \nonumber \]
\[8.0 = 7.48 + log \dfrac{[A^{-}]}{[HA]} \nonumber \]
\[0.52 = log \dfrac{[A^{-}]}{[HA]} \nonumber \]
\[3.31 = \dfrac{0.4375 - [HA]}{[HA]} \nonumber \]
At pH 8.0, [HA] = 0.101 mmoles, which is 23% of the total moles of HEPES in the solution.
Q2) As discussed in Section 2.3, when CO2 is inhaled, it reacts with water to form the weak acid carbonic acid, acidifying the blood. The reaction is given below for reference.
CO2 + H2O → H+ = HCO3-
Currently, the air you breathe contains about 0.04% CO2. This number has risen from 0.03% in the 1960s and is projected to increase to 0.08% by 2100 if fossil fuel consumption remains at its current rate. (PCO2 =0.0003 atm, Keeling, 1960, PCO2 =0.0008 atm MIT System Dynamics Group, 2015).
Using the Ideal Gas Laws, there were 0.44 μM CO2 in the 1960s, 0.60 μM CO2 today, and as much as 1.2 μM CO2 in 2100. The pKa of carbonic acid is 6.35 and the pH of your blood is 7.60.
a) What is the change in blood pH due to the increase in atmospheric CO2 from the 1960s to today?
b) If you were to do the calculations for what the pH of the blood would rise to in 2100, you'd find it to be pH 7.3. Do you think the body would be able to compensate for this? Use the pKa of histidine to explain your answer.
A2)
a)
\[pH = pK_a + log \dfrac{[A^{-}]}{[HA]} \nonumber \]
\[7.60 = 6.35 + log \dfrac{[A^{-}]}{0.6} \nonumber \]
\[1.25 = log \dfrac{[A^{-}]}{0.6} \nonumber \]
\[17.8 = \dfrac{[A^{-}]}{0.6} \nonumber \]
[A-] = 10.7 μM
\[pH_1960 = 6.35 + log \dfrac{10.7}{0.44} \nonumber \]
pH1960 = 6.35 + 1.39
pH1960 = 7.74
Now knowing the pH of the blood from 1960, the change is 7.74 - 7.6 = 0.14 pH units.
b) Yes the body would compensate by increasing the amount of carbonic acid in the blood to buffer the increase in free H+. If the body did not do this, the protonation state of histidine would change and affect every histidine-containing protein in the blood.
Section 4 Questions
Q1) Categorize the following bonds as ion-ion, ion-dipole, dipole-dipole, H-bond, or hydrophobic, (some maybe be used more than once, or not at all).
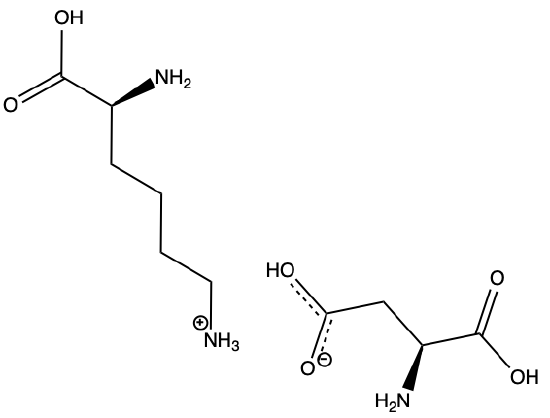
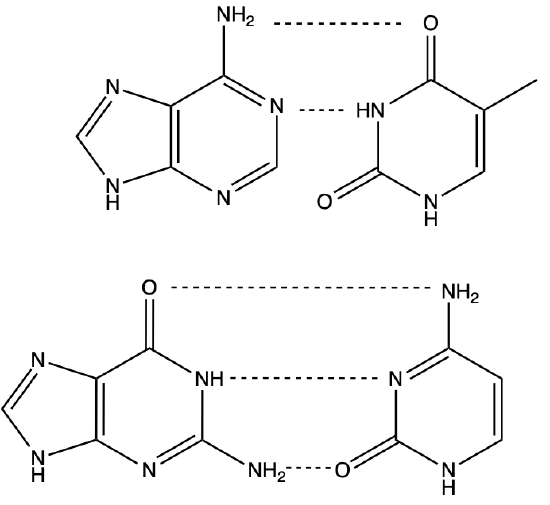
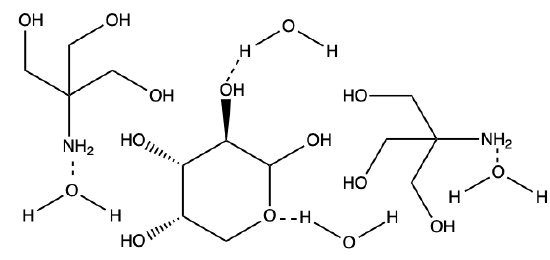
A1)
Q2) You discover a new enzyme, "biochemase" and decide to crystalize the protein to determine if you can determine the role from its structure. You hypothesize the protein forms a homodimer when two identical subunits of a protein come together to form one functional structure. You identify two regions that you believe could be the dimer interface. The first region looks to contain several polar amino acids, whose R-groups are less than 5 Å apart, while the second section contains a large cluster of hydrophobic amino acids.
a) You perform some biochemical experiments and discover that when the polar region is removed, the enzyme is not able to self-assemble, but maintains two intact separate structures. What can you conclude about this region and the type of interactions that are stabilizing the dimer interface?
b) When you remove the nonpolar region, you notice the protein aggregates or crashes out of the solution due to an inability to fold correctly. What role can you hypothesize the nonpolar amino acids have in keeping the protein properly folded?
A2)
a) When the polar region is removed, the protein is still able to fold into two stable subunits, but unable to form its homodimer. Therefore we can conclude that the polar region is necessary for the dimer interface, but the protein can still form subunits without it present.
b Now, when the nonpolar region is removed the protein cannot form subunits or dimers. So, we can conclude that the nonpolar region is necessary for protein stability, and without this region present, the protein cannot fold properly. Most likely, the hydrophobic region forces the protein to fold in the correct way by forcing the nonpolar amino acids to the core, and the polar to the surface. This would then allow for a proper dimer interface to form.
Section 5 Questions
Q1) Acetaminophen (Tylenol) and Ibuprofen (Aspirin) are both common pain-relieving/fever-reducing drugs. However, their chemical properties differ, making acetaminophen more suitable for relieving headaches and fever, while ibuprofen can more effectively reduce pain. The pKpart for acetaminophen is 0.91, while the pKpart is 3.97. (DrugBank) The chemical structures for each are given below:
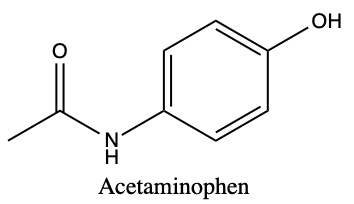
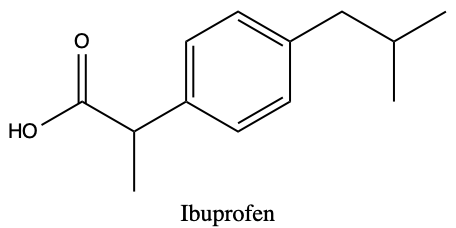
a) Using equation 2.5.4, determine the ΔG° for each compound, and hypothesize which is more soluble based on your answer and explain. Assume a normal body temperature of T = 37°C
b) With your answer from a) and the information reviews in chapter 1 on functional groups, and types of non-polar interactions in chapter 2, identity the regions of both acetaminophen and ibuprofen that can facilitate interactions.
c) Using your ΔG° calculations and functional group analysis, predict which compound is more likely to stay in the blood and bound to red blood cells, and which can rapidly diffuse across the cell membrane.
Q2) You are tasked with creating a solution of nonpolar and polar solvents to use for thin layer chromatography (a technique we will cover later), to separate lipids. Consider the following enthalpic and entropic values for these nonpolar and polar solvents. Using the information discussed in section 5, which of the following combinations of solvents will yield a homo



