9.7: Histidine Tagging
- Page ID
- 3084
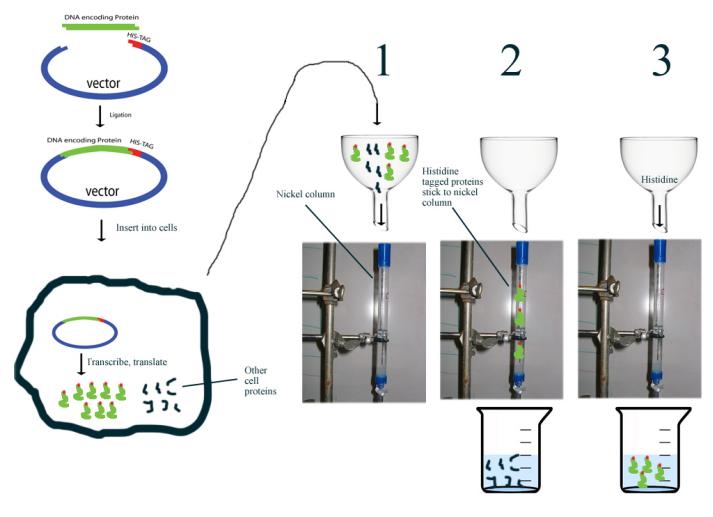
Histidine tagging is a powerful tool for isolating a recombinant protein from a cell lysate. It relies on using recombinant DNA techniques to add codons specifying a series of histidines (usually six) to the coding sequence for a protein. The protein produced when this gene is expressed has a run of histidine residues fused at either the carboxyl or amino terminus to the amino acids in the remainder of the protein. The histidine side chains of this “tag" have an affinity for nickel or cobalt ions, making separation of histidine tagged proteins from a cell lysate is relatively easy. Simply passing the sample through a column that has immobilized nickel or cobalt ions allows the histidine- tagged proteins to “stick," while the remaining cell proteins all pass quickly through. The histidine-tagged proteins are then eluted by addition of imidazole (which is chemically identical to the histidine side chain) to the column. Histidine tags can be cleaved off using endopeptidases.