6.5: Citric Acid Cycle
- Page ID
- 2987
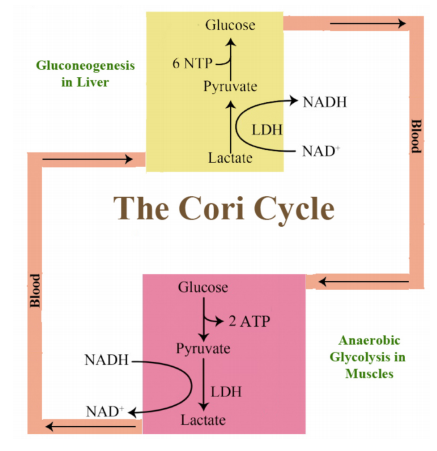
Cori Cycle
With respect to energy, the liver and muscles act complementarily. The liver is the major organ in the body for the synthesis of glucose. Muscles are major users of ATP. Actively exercising muscles generate lactate as a result of running glycolysis faster than the blood can deliver oxygen during periods of heavy exercise. As a consequence, the muscles go anaerobic and produce lactate. This lactate is of no use to muscle cells, so they dump it into the blood. Lactate travels in the blood to the liver, which takes it up and reoxidizes it back to pyruvate, catalyzed by the enzyme lactate dehydrogenase. Pyruvate in the liver is then converted to glucose by gluconeogenesis. The glucose thus made by the liver is dumped into the bloodstream where it is taken up by muscles and used for energy, completing a very important intercellular pathway known as the Cori cycle.
Citric Acid Cycle
The primary catabolic pathway in the body is the citric acid cycle because it is here that oxidation to carbon dioxide occurs for breakdown products of the cell’s major building blocks - sugars, fatty acids, amino acids. The pathway is cyclic (Figure 6.5.2) and thus, doesn’t really have a starting or ending point. All of the reactions occur in the mitochondrion, though one enzyme is embedded in the organelle’s membrane. As needs change, cells may use a subset of the reactions of the cycle to produce a desired molecule rather than to run the entire cycle (Figure 6.5.2).
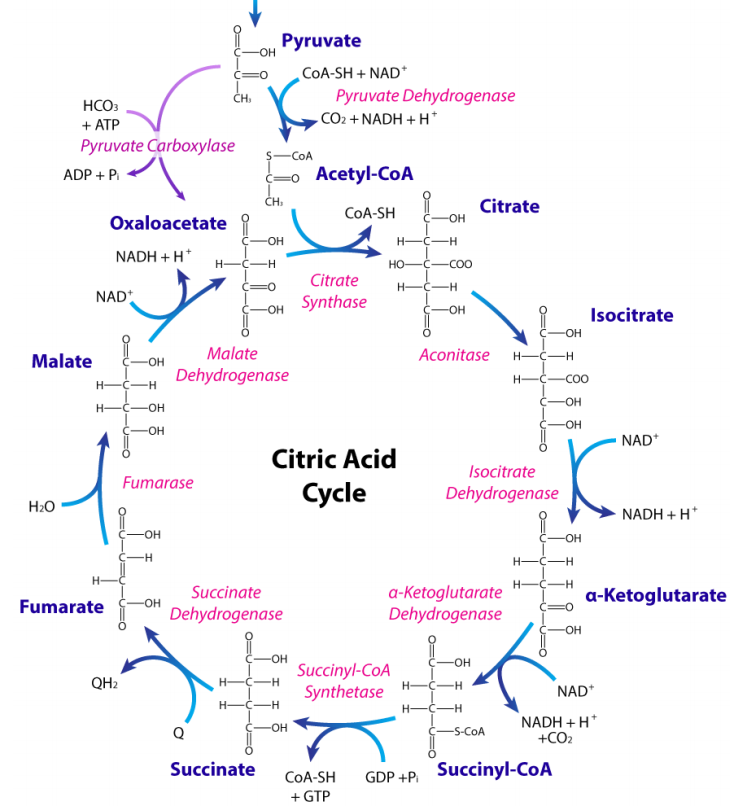
Focusing on the pathway itself, the traditional point to start discussion is addition of acetyl-CoA to oxaloacetate (OAA) to form citrate. Acetyl-CoA for the pathway can come from a variety of sources. They include pyruvate oxidation (from glycolysis and amino acid metabolism), fatty acid oxidation, and amino acid metabolism. The reaction joining it to OAA is catalyzed by citrate synthase and the \(\Delta\)G°’ is fairly negative. This, in turn, helps to “pull" the reaction preceding it in the cycle (catalyzed by malate dehydrogenase).
In the next reaction, citrate is isomerized to isocitrate by action of the enzyme called aconitase. Isocitrate is a branch point in plants and bacteria for the glyoxylate cycle. Oxidative decarboxylation of isocitrate by isocitrate dehydrogenase produces the first NADH and yields alpha-ketoglutarate. This five carbon intermediate is a branch point for synthesis of glutamate. In addition, glutamate can also be made easily into this citric acid cycle intermediate. Decarboxylation of alpha-ketoglutarate yields succinyl-CoA and is catalyzed by alpha- ketoglutarate dehydrogenase. This enzyme is structurally very similar to pyruvate dehydrogenase and employs the same five coenzymes – NAD, FAD, CoASH, TPP, and lipoic acid.
The remainder of the citric acid cycle involves conversion of the four carbon succinyl-CoA into oxaloacetate. Succinyl-CoA is a branch point for the synthesis of heme. Succinyl-CoA is converted to succinate in a reaction catalyzed by succinyl-CoA synthetase (named for the reverse reaction) and a GTP is produced, as well – the only substrate level phosphorylation in the cycle. The energy for the synthesis of the GTP comes from hydrolysis of the high energy thioester bond between succinate and the CoA. Evidence for the high energy of a thioester bond is also evident in the citrate synthase reaction, which is also very energetically favorable. Succinate is also produced by metabolism of odd-chain fatty acids (see below).
Oxidation of succinate occurs in the next step, catalyzed by succinate dehydrogenase. This interesting enzyme both catalyzes this reaction and participates in the electron transport system, funneling electrons from the \(\text{FADH}_2\) it gains in the reaction to coenzyme Q. The product of the reaction, fumarate gains a water across its trans double bond in the next reaction, catalyzed by fumarase to form malate . Fumarate is also a byproduct of nucleotide metabolism and of the urea cycle . Malate is important also for transporting electrons across membranes in the malate aspartate shuttle and in ferrying carbon dioxide in C4 plants.
Conversion of malate to OAA is a rare biological oxidation that has a \(\Delta\)G°’ with a positive value. The reaction product includes NADH and the reaction is ‘pulled’ by the energetically favorable conversion of OAA to citrate in what was described above as the first reaction of the cycle. OAA intersects other important pathways, including amino acid metabolism (readily converted to aspartic acid), transamination (nitrogen movement) and gluconeogenesis.


