2.1: Oxidative Energy
- Page ID
- 1702
Living organisms are made up of cells, and cells contain many biochemical components such as proteins, lipids, and carbohydrates. But, living cells are not random collections of these molecules. They are extraordinarily organized or "ordered". By contrast, in the nonliving world, there is a universal tendency to increasing disorder. Maintaining and creating order in cells takes the input of energy.
Without energy, life is not possible. It is therefore important that we consider energy first in our attempt to understand biochemistry. Where does energy come from? Photosynthetic organisms can capture energy from the sun, converting it to chemical forms usable by cells. Heterotrophic organisms like ourselves get our energy from the food we eat. How do we extract the energy from the food we eat?
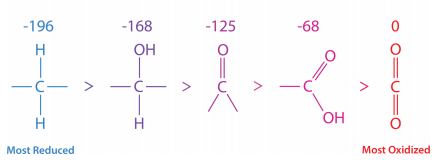
In this series, the most reduced form of carbon is on the left. The energy of oxidation of each form is shown above it. Fatty acids are more reduced overall than sugars. This can also be seen by their formulas.
Palmitic acid = \(C_{16}H_{34}O_2\)
Glucose = \(C_6H_{12}O_6\)
Palmitic acid only contains two oxygens per sixteen carbons, whereas glucose has six oxygen atoms per six carbons.
Consequently, when palmitic acid is fully oxidized, it generates more ATP per carbon (128/16) than glucose (38/6). It is because of this that we use fat as our primary energy storage material.


