Lab 3: Simple, Negative, and Gram Stain
- Page ID
- 23983
PREPARING AN EMULSION:
1. Working in pairs, label each slide and draw a circle on the center of the slide with a wax pencil which is provided at your table, DO NOT use a sharpie, this will keep the bacteria concentrated in one area on the slide.
2. Prepare an emulsion on each slide:
- If you are taking a bacteria from a plate, place a small drop of water on a slide and aseptically add bacteria.
- If you are taking a bacteria from a broth, place 3-6 loopfuls of bacteria onto a slide with no water added.
3. You and your lab partner will need to prepare the following slides:
- 3 slides of Staphylococcus epidermidis (one for simple stain, one for gram stain, one as back-up)
- 3 slides of Pseudomonas aeruginosa (one for simple stain, one for gram stain, one as back-up)
- 2 slides of S. epidermidis and P. aeruginosa mixed together (one for gram stain, one as back-up)
4. Mix gently until you get an even cloudy mixture (should look like skim milk)
Note
If you mix too aggressively, you will lose the bacterial morphology.
5. Allow the slide(s) to air dry on the slide warmer. While the slides are drying, start your negative stain.
6. Once the liquid has completely evaporated, heat fix by passing the slide through a flame three times.
Note
If you heat fix too little, the bacteria will wash off the slide. If you heat fix too much, you will cook the bacteria and denature them.
7. Allow the slide to cool and then continue with your staining protocol.
NEGATIVE STAIN:
Definition
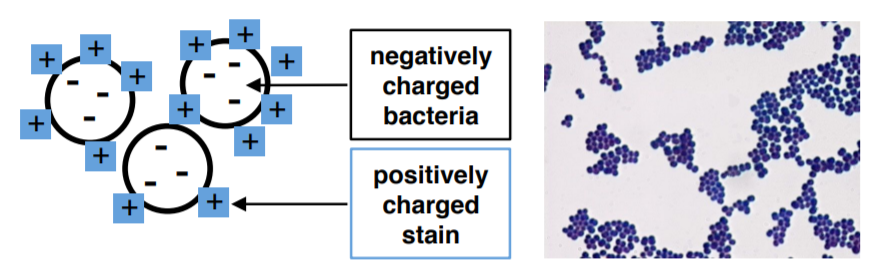
Nigrosin is a simple and indirect stain used for determining bacterial morphology. The shapes and sizes of the organisms are seen as color-free outlines against the dark background. An advantage of using this method is that prior fixation by heat is not needed, so the organisms are seen in more lifelike shapes. Nigrosin is an acidic stain which becomes negatively charged. Since the surface of most bacterial cells is negatively charged, the cell surface repels the stain. The glass of the slide will stain, but the bacterial cells will not.

8. If you need to, step outside and watch this video to make sure you understand how to do the procedure: www.youtube.com/watch?v=avveXgPWVJ8 (you can also google “negative stain video”)
9. Add a small drop of nigrosin to the slide.
10. Aseptically transfer one loopful of your NEGATIVE STAIN MIX bacteria into the drop of nigrosin and mix gently.
11. Use a second slide, held at a 45-degree angle to smear across your slide.
12. Allow the slides to air dry on the counter. Do not use slide warmer!
13. Repeat this procedure to make a slide of S. epidermidis and a slide of P. aeruginosa 14. Examine all three slides under oil immersion and record your results on your worksheet.
SIMPLE STAIN:
Definition
Methylene blue is a simple and direct stain used for determining bacterial morphology (shape and arrangement). It is a cationic dye (positive charge) which stains the cell a blue color. The presence of negatively charged molecules in the cell (like DNA & RNA) causes the cell to stain blue.
15. Use the slides that you already prepared.
16. Add Methylene blue stain to heat-fixed slide and leave for 2 minutes.
17. Hold the slide in an angle and rinse gently with water from the squirt bottle.
18. Blot gently with bibulous paper to get rid of excess water.
19. Examine under oil immersion and record your results on your worksheet.
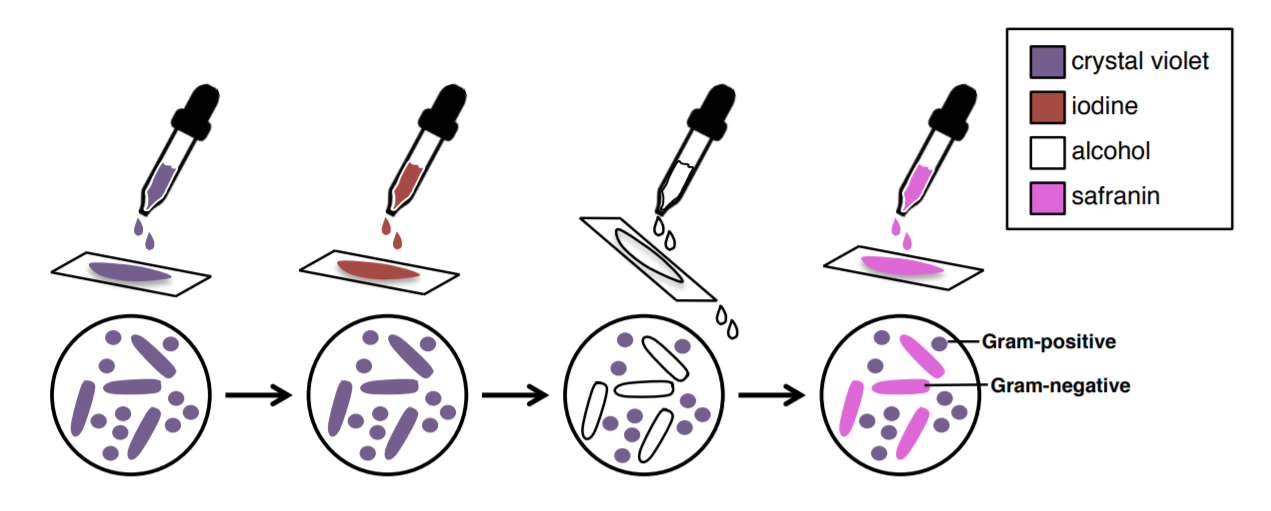
GRAM STAIN:
Definition
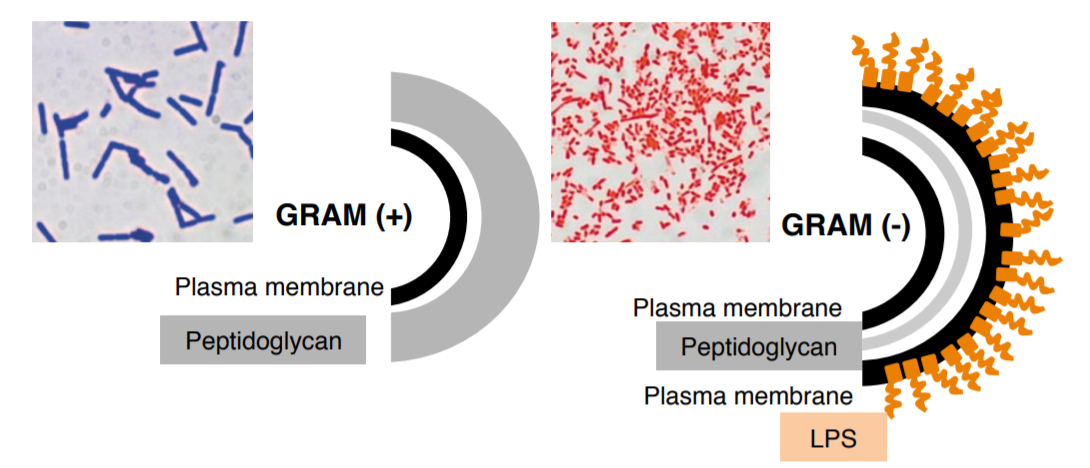
The Gram stain is the most important and universally used staining technique in the bacteriology laboratory. It is used to distinguish between gram (+) and gram (-) bacteria. The difference between gram (+) and gram (-) bacteria lies in the ability of the cell wall of the organism to retain the crystal violet.
18. Use the slides that you already prepared (1) S. epidermidis (2) P. aeruginosa (3) S. epidermidis and P. aeruginosa mixed together.
19. Stain with crystal violet for 1 minute, then rinse gently with water.
20. Treat with iodine mixture for 1 minute, then rinse gently with water.
21. Rinse with ~3-6 drops of 95% alcohol to decolorize, then rinse gently with water.
22. Counterstain with safranin for 30 seconds, then rinse with water.
Note
Proper gram stains of the mixed culture are worth 2 points extra credit!