B6. Ligand-Gated Acetylcholine Receptor of the Neuromuscular Junction
- Page ID
- 5594
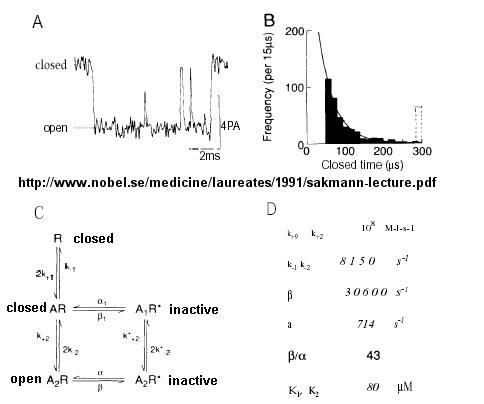
Kinetic modeling based on patch-clamp recordings shows that the protein can exist in multiple conformations, including a closed (C), open (O), and inactive (I) form. The binding of two acetylcholine (A) molecules are required to open the channel. When two are bound, the protein exist in two interconvertible forms: \( A_2C \rightleftharpoons A_2O\). Through another conformation change, the open form O can inactivate in a slow step to \(A_2I\), where I is an inactive form.
The channel consists of five helices which are constricted by hydrophobic side chains which point inward into the pore. A rotation and sliding of the helices relative to each other probably occurs when ligand binds the protein, allowing the channel opening to increase. Rings of negatively changed side chains line the pore at the top and bottom accounting for the selectivity of the channel for positive ions.
Much of our understanding of the mechanism of the acetylcholine channel opening comes from a patch clamp technique in which a micropipette is used to remove a small section of a membrane containing a channel.
Figure: patch clamp technique
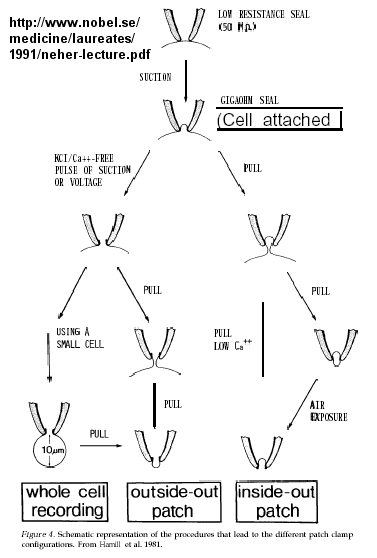
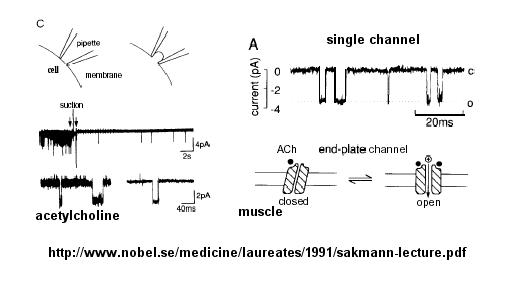
Ion flow, measured as picoamp current, can be measured across the membrane patch. The opening and closing of single channels can be recorded using this technique, for which Nehert and Sakman won the Nobel Prize.
Figure: opening and closing of single channels
From this, they deduced a mechanism for gating of the acetylcholine channel.
Figure: mechanism for gating of the acetylcholine channel
In general, transmitter-gated channels are similar to the acetylcholine receptor. Mechanisms to generate an excitatory synaptic potential (with a transmitter-gated receptor/channel) are similar to that of generating an action potential (with voltage-gated channels) in that both involve movement of sodium and potassium ions across the membrane. They differ in several ways, however:
- In the generation of action potentials, sodium and potassium ions move through different voltage-gated channels - i.e. the channels are selective for the ion. Transmitter-gated channels usually are not selective for the ions since the pore is too large.
- ion flux through voltage-gated channels is "regenerative" in that increased depolarization caused by the channel opening leads to more influx of the ion. In transmitter-gated channel, the amount of ion passing through the membrane depends on the local concentration of neurotransmitter released at the synapse.
- When acetylcholine causes the opening of the acetylcholine receptor/pore, sodium can flow in and potasium can flow out (down their respective concentration gradients). However, more sodium flows in initially than potassium flows out since potassium efflux is resisted by the negative transmembrane potential. This leads to an inward flow of sodium enough to cause the activation of potential-gated sodium channels. Since these are voltage gated, much more sodium can enter the cell, leading to an action potential and the ultimate opening of vesicular calcium ion channels. This leads to a rise in intracellular calcium (through opening of a Ca channel in membranes of intracellular organelles with high concentrations of Ca ions), which activates muscle contraction.
![]() Electron Micrograph of Junction-End Plate
Electron Micrograph of Junction-End Plate
![]() Animation: Acetylcholine receptor
Animation: Acetylcholine receptor
![]() Updated Acetylcholine Receptor Pore - JSMol (HTML5) (1OED)
Updated Acetylcholine Receptor Pore - JSMol (HTML5) (1OED)
![]() How does ligand binding lead to channel opening? This has been studied in detail by Douherty and Lester for binding of acetylcholine, a positively charged ligand to its neural receptor, the neuronal nicotinic nACh receptor, a ligand-gated ion channel. It, along with the neural serotonin (HT3) and GABA receptors belong to the Cys-Loop superfamily of receptors which consists of 5 protein chains. Douherty and Lester have reconstituted the AChR into Xenopus oocyte (egg) membranes and have done a combination of electrophysiological and chemical studies to probe the conformational changes in the receptor. In the absence of ligand the channel pore is blocked by 5 leucine side chains, which move on ligand binding. The neurotransmitter binds to a tryptophan side chain in an "aromatic box" that contains five aromatic amino acids), with the positive charge interacting with the aromatic cloud by cation-π interactions. Serotonin and benzodiazepines (a ligand that interacts with the GABA channel - see below) bind to different aromatic side chains in the aromatic boxes in their receptors, indicating much less specific interactions compared to enzyme-substrate interactions. This explains how nicotine can bind to neural AChR and activate it even though their structures differ significantly. They found that the conformational changes allowing channel opening requires a trans-cis isomerization of a Pro side chain. Site-specific mutants that favor the trans form favor closing. However, not all members of the Cys-Loop superfamily have a Pro at this position.
How does ligand binding lead to channel opening? This has been studied in detail by Douherty and Lester for binding of acetylcholine, a positively charged ligand to its neural receptor, the neuronal nicotinic nACh receptor, a ligand-gated ion channel. It, along with the neural serotonin (HT3) and GABA receptors belong to the Cys-Loop superfamily of receptors which consists of 5 protein chains. Douherty and Lester have reconstituted the AChR into Xenopus oocyte (egg) membranes and have done a combination of electrophysiological and chemical studies to probe the conformational changes in the receptor. In the absence of ligand the channel pore is blocked by 5 leucine side chains, which move on ligand binding. The neurotransmitter binds to a tryptophan side chain in an "aromatic box" that contains five aromatic amino acids), with the positive charge interacting with the aromatic cloud by cation-π interactions. Serotonin and benzodiazepines (a ligand that interacts with the GABA channel - see below) bind to different aromatic side chains in the aromatic boxes in their receptors, indicating much less specific interactions compared to enzyme-substrate interactions. This explains how nicotine can bind to neural AChR and activate it even though their structures differ significantly. They found that the conformational changes allowing channel opening requires a trans-cis isomerization of a Pro side chain. Site-specific mutants that favor the trans form favor closing. However, not all members of the Cys-Loop superfamily have a Pro at this position.
In a very recent finding, Dougherthy's group discovered the mechanism underlying the fact that nicotine does not bind with high affinity to the acetylcholine receptor in muscles. Apparently it can not make the cation-π interactions necessary for interaction with the receptor. A H-bond between a H attached to the positive charged nicotine to the Trp carbonyl in the aromatic box allows the cation-π interaction in the neural form. The H bond is not apparently not present in the muscle form, which prevents strong cation-π interaction with nicotine. If it could bind to the muscle acetylcholine receptor, nicotine would be so poisonous that no one would smoke.
Contributors and Attributions


